Neuroleptic malignant syndrome (NMS) is a rare, idiosyncratic, and potentially life-threatening reaction to antipsychotic Antipsychotic Antipsychotics, also called neuroleptics, are used to treat psychotic disorders and alleviate agitation, mania, and aggression. Antipsychotics are notable for their use in treating schizophrenia and bipolar disorder and are divided into 1st-generation antipsychotics (FGAs) and atypical or 2nd-generation antipsychotics. First-Generation Antipsychotics drugs. Neuroleptic malignant syndrome presents with ≥ 2 of the following cardinal symptoms: fever Fever Fever is defined as a measured body temperature of at least 38°C (100.4°F). Fever is caused by circulating endogenous and/or exogenous pyrogens that increase levels of prostaglandin E2 in the hypothalamus. Fever is commonly associated with chills, rigors, sweating, and flushing of the skin. Fever, altered mental status Altered Mental Status Sepsis in Children, muscle rigidity Muscle rigidity Continuous involuntary sustained muscle contraction which is often a manifestation of basal ganglia diseases. When an affected muscle is passively stretched, the degree of resistance remains constant regardless of the rate at which the muscle is stretched. This feature helps to distinguish rigidity from muscle spasticity. Motor Neuron Lesions, and autonomic dysfunction Autonomic Dysfunction Anterior Cord Syndrome. It is important to promptly identify this condition because of its high morbidity Morbidity The proportion of patients with a particular disease during a given year per given unit of population. Measures of Health Status and mortality Mortality All deaths reported in a given population. Measures of Health Status rates. Diagnosis is based on positive clinical and laboratory findings, along with the exclusion of other possible causes. Management involves discontinuing antipsychotic Antipsychotic Antipsychotics, also called neuroleptics, are used to treat psychotic disorders and alleviate agitation, mania, and aggression. Antipsychotics are notable for their use in treating schizophrenia and bipolar disorder and are divided into 1st-generation antipsychotics (FGAs) and atypical or 2nd-generation antipsychotics. First-Generation Antipsychotics medication, supportive care, and pharmacologic options.
Last updated: Jun 15, 2025
Neuroleptic malignant syndrome (NMS) is a life-threatening neurologic disorder associated with the use of antipsychotics that presents as a classic tetrad:
Cardinal features of the neuroleptic malignant syndrome include:
Other signs and symptoms are as follows:
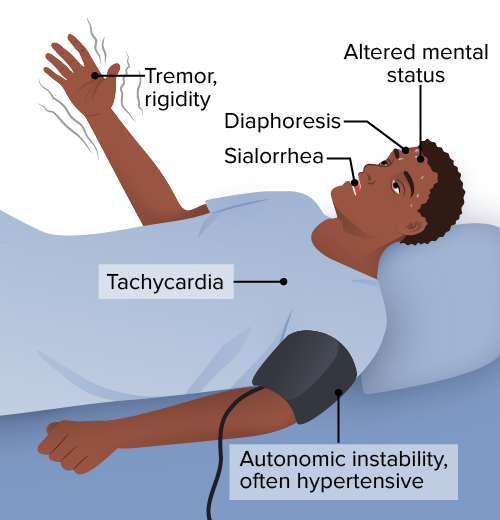
Symptoms of neuroleptic malignant syndrome
Image by Lecturio.The diagnosis of NMS is mainly clinical. Laboratory investigations are performed to rule out other conditions or complications.
Laboratory findings associated with NMS:
NMS is an acute medical emergency and should be treated as such.
The 1st step should be to stop the antipsychotic Antipsychotic Antipsychotics, also called neuroleptics, are used to treat psychotic disorders and alleviate agitation, mania, and aggression. Antipsychotics are notable for their use in treating schizophrenia and bipolar disorder and are divided into 1st-generation antipsychotics (FGAs) and atypical or 2nd-generation antipsychotics. First-Generation Antipsychotics agent or restart the dopamine Dopamine One of the catecholamine neurotransmitters in the brain. It is derived from tyrosine and is the precursor to norepinephrine and epinephrine. Dopamine is a major transmitter in the extrapyramidal system of the brain, and important in regulating movement. Receptors and Neurotransmitters of the CNS agent.
The most relevant differential diagnoses for NMS are serotonin Serotonin A biochemical messenger and regulator, synthesized from the essential amino acid l-tryptophan. In humans it is found primarily in the central nervous system, gastrointestinal tract, and blood platelets. Serotonin mediates several important physiological functions including neurotransmission, gastrointestinal motility, hemostasis, and cardiovascular integrity. Receptors and Neurotransmitters of the CNS syndrome and malignant hyperthermia Malignant hyperthermia An important complication of anesthesia is malignant hyperthermia, an autosomal dominant disorder of the regulation of calcium transport in the skeletal muscles resulting in a hypermetabolic crisis. Malignant hyperthermia is marked by high fever, muscle rigidity, rhabdomyolysis, and respiratory and metabolic acidosis. Malignant Hyperthermia, and it is important to distinguish between them.
Other conditions that present with altered mental status Altered Mental Status Sepsis in Children, autonomic dysfunction Autonomic Dysfunction Anterior Cord Syndrome, and fever Fever Fever is defined as a measured body temperature of at least 38°C (100.4°F). Fever is caused by circulating endogenous and/or exogenous pyrogens that increase levels of prostaglandin E2 in the hypothalamus. Fever is commonly associated with chills, rigors, sweating, and flushing of the skin. Fever: