Glycolysis is a central metabolic pathway responsible for the breakdown of glucose Glucose A primary source of energy for living organisms. It is naturally occurring and is found in fruits and other parts of plants in its free state. It is used therapeutically in fluid and nutrient replacement. Lactose Intolerance and plays a vital role in generating free energy Free energy Enzyme Kinetics for the cell and metabolites for further oxidative degradation. Glucose Glucose A primary source of energy for living organisms. It is naturally occurring and is found in fruits and other parts of plants in its free state. It is used therapeutically in fluid and nutrient replacement. Lactose Intolerance primarily becomes available in the blood as a result of glycogen breakdown or from its synthesis Synthesis Polymerase Chain Reaction (PCR) from noncarbohydrate precursors ( gluconeogenesis Gluconeogenesis Gluconeogenesis is the process of making glucose from noncarbohydrate precursors. This metabolic pathway is more than just a reversal of glycolysis. Gluconeogenesis provides the body with glucose not obtained from food, such as during a fasting period. The production of glucose is critical for organs and cells that cannot use fat for fuel. Gluconeogenesis) and is imported into cells by specific transport proteins Transport proteins Proteins and Peptides. Glycolysis occurs in the cytoplasm and consists of 10 reactions, the net result of which is the conversion of 1 C6 glucose Glucose A primary source of energy for living organisms. It is naturally occurring and is found in fruits and other parts of plants in its free state. It is used therapeutically in fluid and nutrient replacement. Lactose Intolerance to 2 C3 pyruvate molecules. The free energy Free energy Enzyme Kinetics of this process is harvested to produce adenosine Adenosine A nucleoside that is composed of adenine and d-ribose. Adenosine or adenosine derivatives play many important biological roles in addition to being components of DNA and RNA. Adenosine itself is a neurotransmitter. Class 5 Antiarrhythmic Drugs triphosphate (ATP) and nicotinamide adenine dinucleotide Nicotinamide adenine dinucleotide A coenzyme composed of ribosylnicotinamide 5'-diphosphate coupled to adenosine 5'-phosphate by pyrophosphate linkage. It is found widely in nature and is involved in numerous enzymatic reactions in which it serves as an electron carrier by being alternately oxidized (NAD+) and reduced (NADH). Pentose Phosphate Pathway hydride (NADH), key energy-yielding metabolites. The overall stoichiometry of the pathway is: glucose Glucose A primary source of energy for living organisms. It is naturally occurring and is found in fruits and other parts of plants in its free state. It is used therapeutically in fluid and nutrient replacement. Lactose Intolerance + 2 Pi + 2 ADP + 2 NAD NAD+ A coenzyme composed of ribosylnicotinamide 5'-diphosphate coupled to adenosine 5'-phosphate by pyrophosphate linkage. It is found widely in nature and is involved in numerous enzymatic reactions in which it serves as an electron carrier by being alternately oxidized (NAD+) and reduced (NADH). Pentose Phosphate Pathway+ > 2 pyruvate + 2 ATP + 2 NADH + 2 H+ + 2 H2O (H+: hydrogen ion, Pi: phosphate Phosphate Inorganic salts of phosphoric acid. Electrolytes ion, NAD NAD+ A coenzyme composed of ribosylnicotinamide 5'-diphosphate coupled to adenosine 5'-phosphate by pyrophosphate linkage. It is found widely in nature and is involved in numerous enzymatic reactions in which it serves as an electron carrier by being alternately oxidized (NAD+) and reduced (NADH). Pentose Phosphate Pathway+: nicotinamide adenine dinucleotide Nicotinamide adenine dinucleotide A coenzyme composed of ribosylnicotinamide 5'-diphosphate coupled to adenosine 5'-phosphate by pyrophosphate linkage. It is found widely in nature and is involved in numerous enzymatic reactions in which it serves as an electron carrier by being alternately oxidized (NAD+) and reduced (NADH). Pentose Phosphate Pathway).
Last updated: Apr 25, 2025
The 1st half of glycolysis requires an energy investment of 2 adenosine Adenosine A nucleoside that is composed of adenine and d-ribose. Adenosine or adenosine derivatives play many important biological roles in addition to being components of DNA and RNA. Adenosine itself is a neurotransmitter. Class 5 Antiarrhythmic Drugs triphosphate (ATP) molecules and serves to convert the hexose glucose Glucose A primary source of energy for living organisms. It is naturally occurring and is found in fruits and other parts of plants in its free state. It is used therapeutically in fluid and nutrient replacement. Lactose Intolerance into 2 trioses. The process consists of 5 steps:
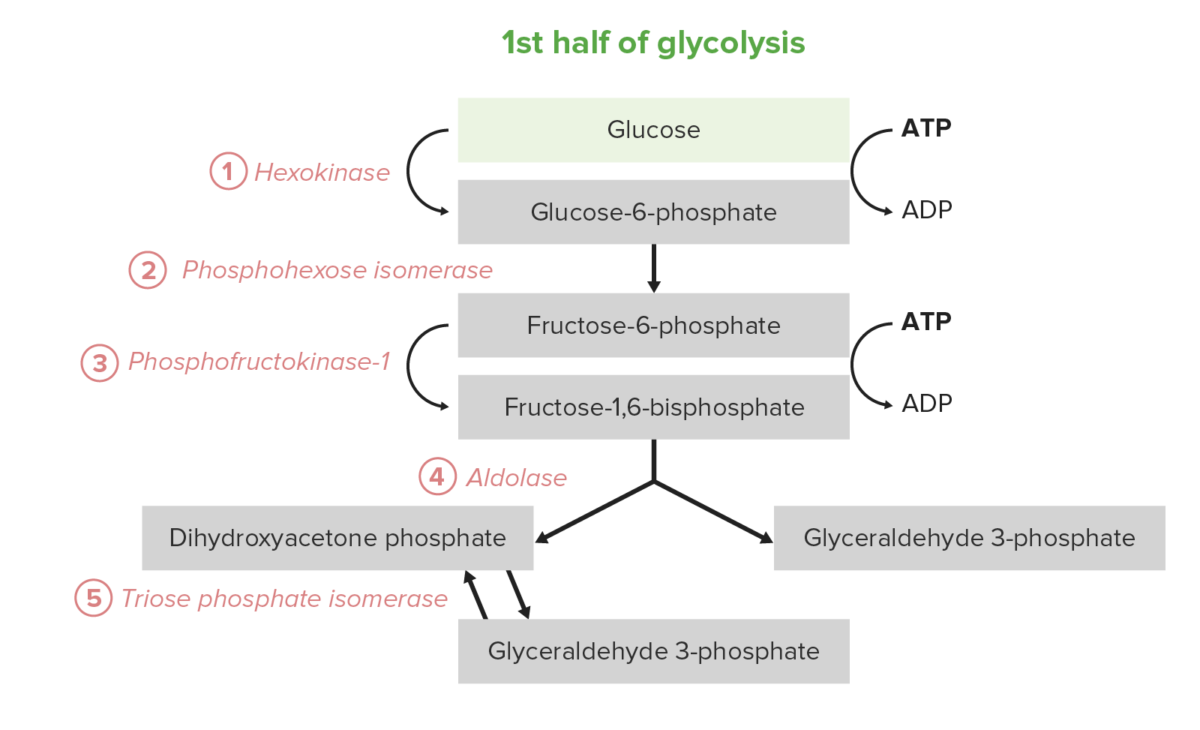
The first 5 steps (first half) of the glycolysis pathway
Image by Lecturio.The 2nd half of glycolysis converts the triose GAP to pyruvate, with the concomitant generation of 4 ATP and 2 nicotinamide adenine dinucleotide Nicotinamide adenine dinucleotide A coenzyme composed of ribosylnicotinamide 5′-diphosphate coupled to adenosine 5′-phosphate by pyrophosphate linkage. It is found widely in nature and is involved in numerous enzymatic reactions in which it serves as an electron carrier by being alternately oxidized (NAD+) and reduced (NADH). Pentose Phosphate Pathway hydride (NADH) per 2 GAP. Thus, the energy investment of steps 1–5 is paid back twice here. In certain cell types and conditions, these 5 steps are the predominant source of ATP:
Net reaction: glucose Glucose A primary source of energy for living organisms. It is naturally occurring and is found in fruits and other parts of plants in its free state. It is used therapeutically in fluid and nutrient replacement. Lactose Intolerance + 2 Pi + 2 ADP + 2 NAD NAD+ A coenzyme composed of ribosylnicotinamide 5′-diphosphate coupled to adenosine 5′-phosphate by pyrophosphate linkage. It is found widely in nature and is involved in numerous enzymatic reactions in which it serves as an electron carrier by being alternately oxidized (NAD+) and reduced (NADH). Pentose Phosphate Pathway+ → 2 pyruvate + 2 ATP + 2 NADH + 2 H+ + 2 H2O
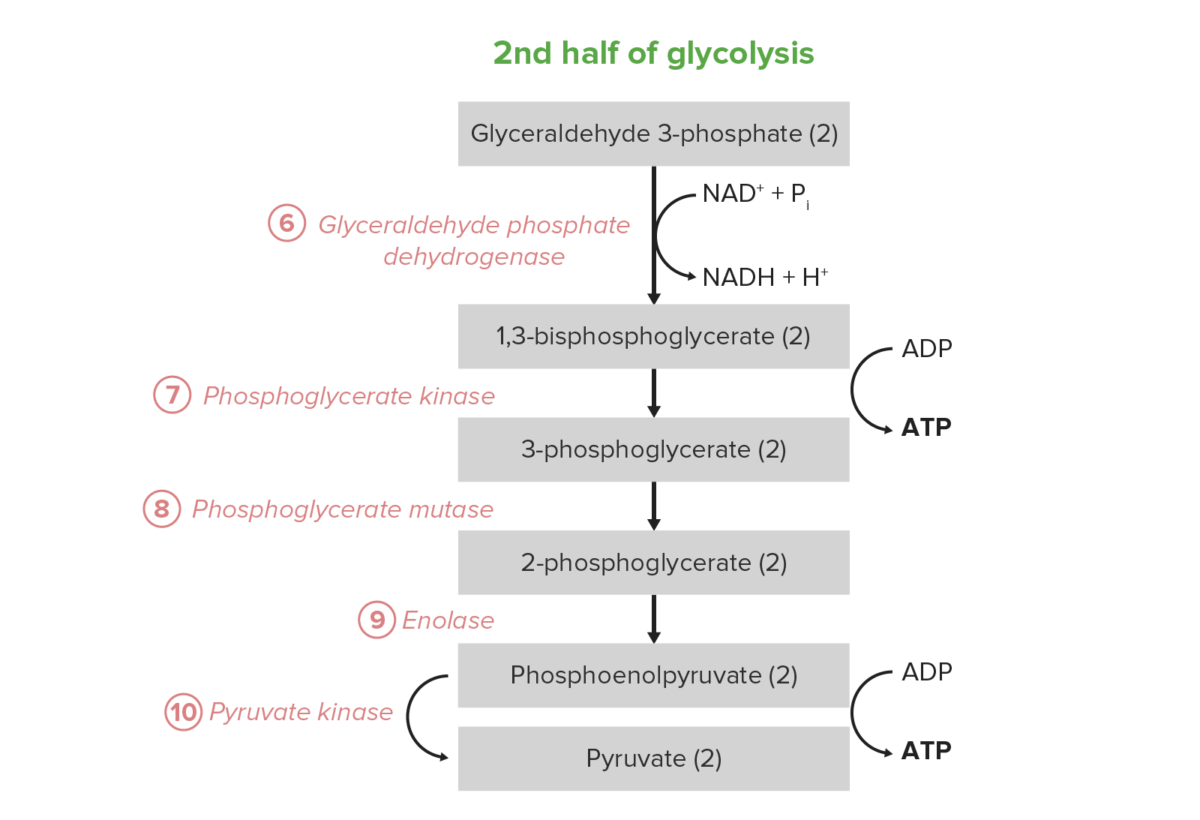
The last 5 steps (last half) of the glycolysis pathway.
Image by Lecturio.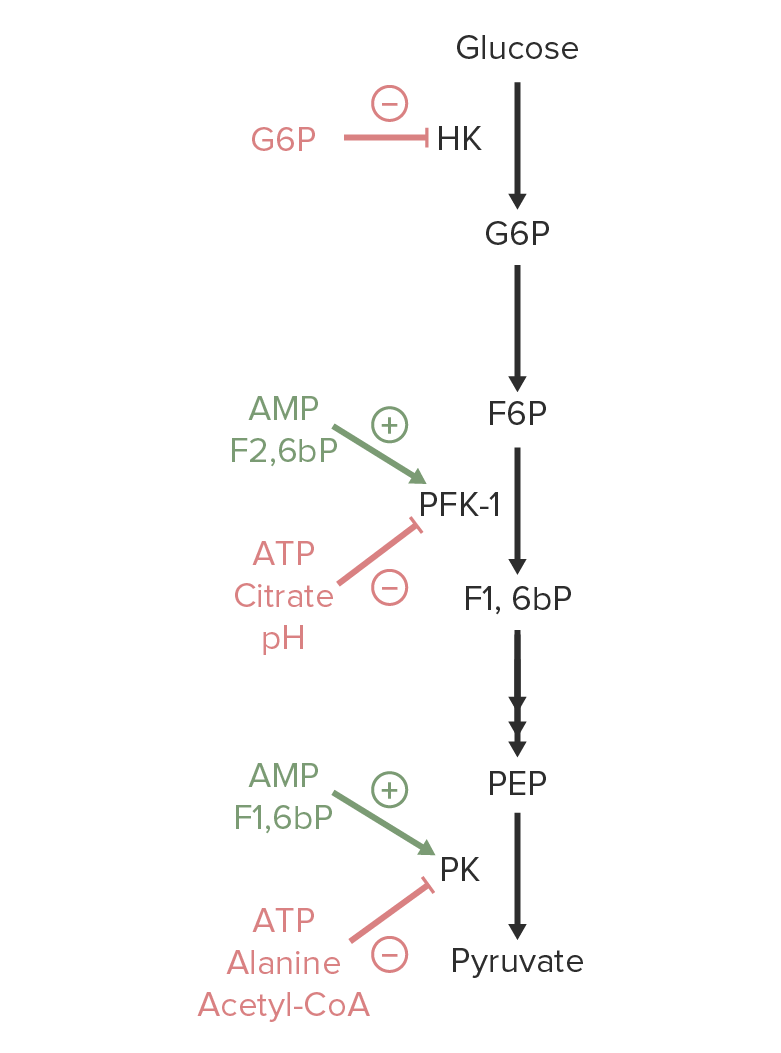
An overview of the regulation of glycolysis. Activators of hexokinase (HK), phosphofructokinase-1 (PFK-1), or pyruvate kinase (PK) are marked in green. Metabolites that inhibit these enzymes are marked in red.
Image by Lecturio.The following are enzymes Enzymes Enzymes are complex protein biocatalysts that accelerate chemical reactions without being consumed by them. Due to the body’s constant metabolic needs, the absence of enzymes would make life unsustainable, as reactions would occur too slowly without these molecules. Basics of Enzymes of the glycolysis pathway that may be involved in congenital enzymatic defects:
These congenital enzymatic defects produce hemolytic anemia Hemolytic Anemia Hemolytic anemia (HA) is the term given to a large group of anemias that are caused by the premature destruction/hemolysis of circulating red blood cells (RBCs). Hemolysis can occur within (intravascular hemolysis) or outside the blood vessels (extravascular hemolysis). Hemolytic Anemia.
Hemolytic anemia Hemolytic Anemia Hemolytic anemia (HA) is the term given to a large group of anemias that are caused by the premature destruction/hemolysis of circulating red blood cells (RBCs). Hemolysis can occur within (intravascular hemolysis) or outside the blood vessels (extravascular hemolysis). Hemolytic Anemia: a group of anemias that are due to destruction or premature Premature Childbirth before 37 weeks of pregnancy (259 days from the first day of the mother’s last menstrual period, or 245 days after fertilization). Necrotizing Enterocolitis clearance of RBCs RBCs Erythrocytes, or red blood cells (RBCs), are the most abundant cells in the blood. While erythrocytes in the fetus are initially produced in the yolk sac then the liver, the bone marrow eventually becomes the main site of production. Erythrocytes: Histology. Intrinsic abnormalities of the RBC lead to splenic clearance ( extravascular hemolysis Extravascular hemolysis Hemolytic Anemia). The chronic destruction of RBCs RBCs Erythrocytes, or red blood cells (RBCs), are the most abundant cells in the blood. While erythrocytes in the fetus are initially produced in the yolk sac then the liver, the bone marrow eventually becomes the main site of production. Erythrocytes: Histology can present as jaundice Jaundice Jaundice is the abnormal yellowing of the skin and/or sclera caused by the accumulation of bilirubin. Hyperbilirubinemia is caused by either an increase in bilirubin production or a decrease in the hepatic uptake, conjugation, or excretion of bilirubin. Jaundice, splenomegaly Splenomegaly Splenomegaly is pathologic enlargement of the spleen that is attributable to numerous causes, including infections, hemoglobinopathies, infiltrative processes, and outflow obstruction of the portal vein. Splenomegaly, cholelithiasis Cholelithiasis Cholelithiasis (gallstones) is the presence of stones in the gallbladder. Most gallstones are cholesterol stones, while the rest are composed of bilirubin (pigment stones) and other mixed components. Patients are commonly asymptomatic but may present with biliary colic (intermittent pain in the right upper quadrant). Cholelithiasis, hematuria Hematuria Presence of blood in the urine. Renal Cell Carcinoma, and symptoms of anemia Anemia Anemia is a condition in which individuals have low Hb levels, which can arise from various causes. Anemia is accompanied by a reduced number of RBCs and may manifest with fatigue, shortness of breath, pallor, and weakness. Subtypes are classified by the size of RBCs, chronicity, and etiology. Anemia: Overview and Types ( shortness of breath Shortness of breath Dyspnea is the subjective sensation of breathing discomfort. Dyspnea is a normal manifestation of heavy physical or psychological exertion, but also may be caused by underlying conditions (both pulmonary and extrapulmonary). Dyspnea, fatigue Fatigue The state of weariness following a period of exertion, mental or physical, characterized by a decreased capacity for work and reduced efficiency to respond to stimuli. Fibromyalgia, syncope Syncope Syncope is a short-term loss of consciousness and loss of postural stability followed by spontaneous return of consciousness to the previous neurologic baseline without the need for resuscitation. The condition is caused by transient interruption of cerebral blood flow that may be benign or related to a underlying life-threatening condition. Syncope, and tachycardia Tachycardia Abnormally rapid heartbeat, usually with a heart rate above 100 beats per minute for adults. Tachycardia accompanied by disturbance in the cardiac depolarization (cardiac arrhythmia) is called tachyarrhythmia. Sepsis in Children).