Necrotizing enterocolitis Enterocolitis Inflammation of the mucosa of both the small intestine and the large intestine. Etiology includes ischemia, infections, allergic, and immune responses. Yersinia spp./Yersiniosis (NEC) is an intestinal inflammatory process that can lead to mucosal injury and necrosis Necrosis The death of cells in an organ or tissue due to disease, injury or failure of the blood supply. Ischemic Cell Damage. The condition is multifactorial, with underlying risk factors that include prematurity Prematurity Neonatal Respiratory Distress Syndrome and formula feeding Formula feeding Formulations for the nutrition of infants that are substituted for breast milk. Infant Care. The clinical presentation varies in severity from feeding intolerance, acute findings on abdominal exam, and systemic symptoms. The diagnosis is based on a clinical suspicion, abnormal abdominal radiographs, and supporting abnormal laboratory results. Management consists of supportive medical care Medical care Conflict of Interest for milder stages and bowel rest and surgical intervention for more advanced stages. Necrotizing enterocolitis Enterocolitis Inflammation of the mucosa of both the small intestine and the large intestine. Etiology includes ischemia, infections, allergic, and immune responses. Yersinia spp./Yersiniosis and its complications carry a high risk of morbidity Morbidity The proportion of patients with a particular disease during a given year per given unit of population. Measures of Health Status and mortality Mortality All deaths reported in a given population. Measures of Health Status.
Last updated: Dec 1, 2022
Necrotizing enterocolitis Enterocolitis Inflammation of the mucosa of both the small intestine and the large intestine. Etiology includes ischemia, infections, allergic, and immune responses. Yersinia spp./Yersiniosis (NEC) is an intestinal inflammatory condition associated with focal or diffuse ulceration Ulceration Corneal Abrasions, Erosion, and Ulcers and necrosis Necrosis The death of cells in an organ or tissue due to disease, injury or failure of the blood supply. Ischemic Cell Damage that primarily affects the terminal ileum Ileum The distal and narrowest portion of the small intestine, between the jejunum and the ileocecal valve of the large intestine. Small Intestine: Anatomy and colon Colon The large intestines constitute the last portion of the digestive system. The large intestine consists of the cecum, appendix, colon (with ascending, transverse, descending, and sigmoid segments), rectum, and anal canal. The primary function of the colon is to remove water and compact the stool prior to expulsion from the body via the rectum and anal canal. Colon, Cecum, and Appendix: Anatomy.
The diagnosis of NEC is made primarily by imaging.
Abdominal X-ray X-ray Penetrating electromagnetic radiation emitted when the inner orbital electrons of an atom are excited and release radiant energy. X-ray wavelengths range from 1 pm to 10 nm. Hard x-rays are the higher energy, shorter wavelength x-rays. Soft x-rays or grenz rays are less energetic and longer in wavelength. The short wavelength end of the x-ray spectrum overlaps the gamma rays wavelength range. The distinction between gamma rays and x-rays is based on their radiation source. Pulmonary Function Tests is the method of choice to establish the diagnosis of NEC.
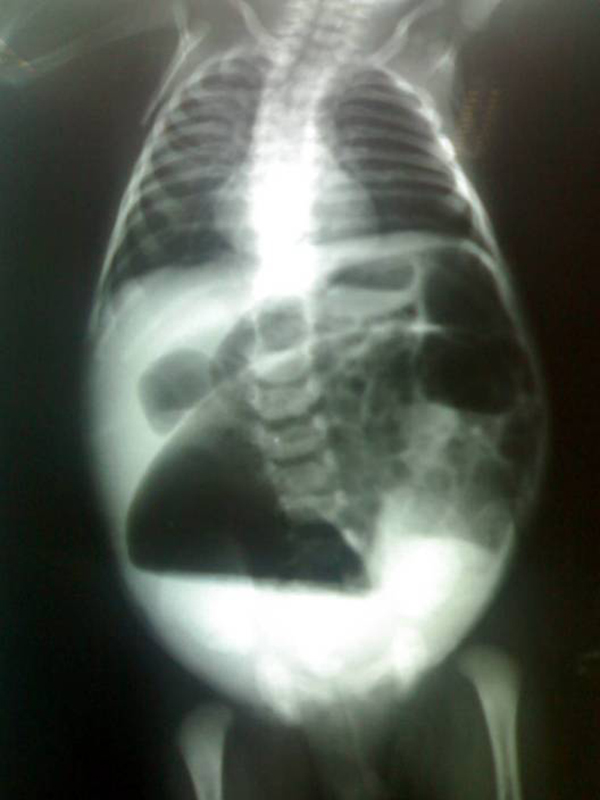
Dilated bowel loops on an abdominal radiograph in patient with necrotizing enterocolitis
Image: “A report of a rare congenital malformation in a Nepalese child with congenital pouch colon: a case report” by Shakya VC, Agrawal CS, Koirala R, Khaniya S, Poudel P, Adhikary S. License: CC BY 3.0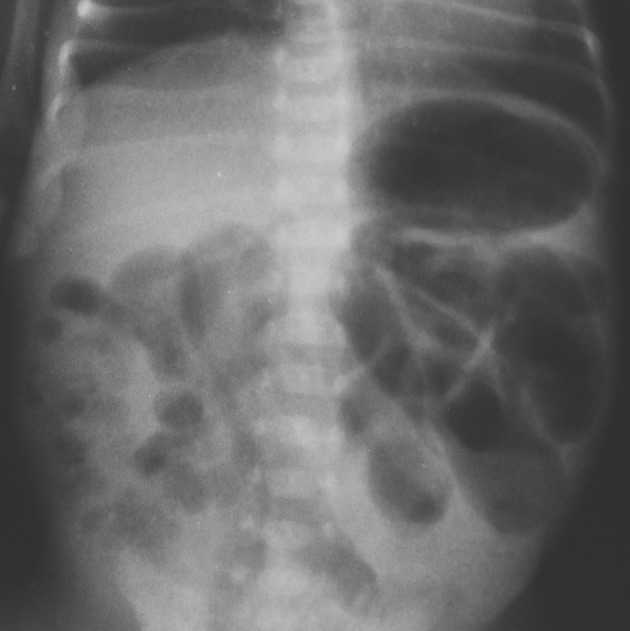
Abdominal radiograph of necrotizing enterocolitis:
This image shows diffuse, gaseous, intestinal distention with discrete signs of suspected pneumatosis in the lower right quadrant.
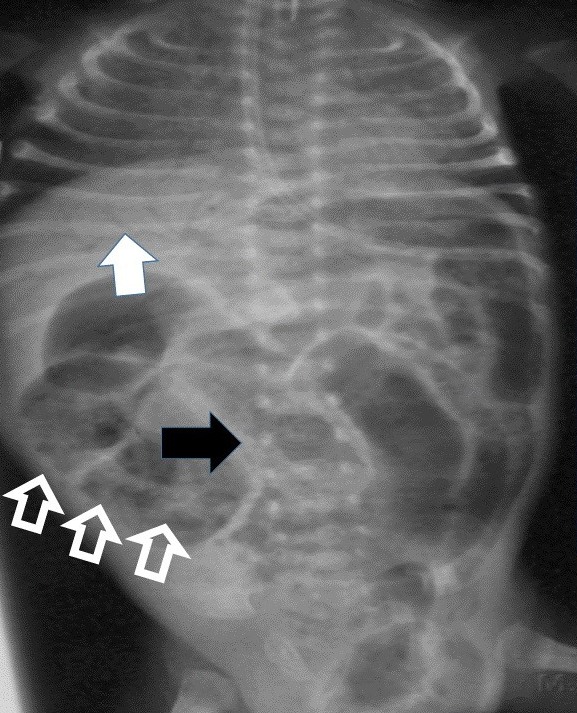
Radiograph of a patient with necrotizing enterocolitis:
Pneumatosis intestinalis appears as a linear lucency (black arrow) and as a “frothy” appearance over the bowel (hollow arrows). The white arrow indicates lucency due to portal venous gas.
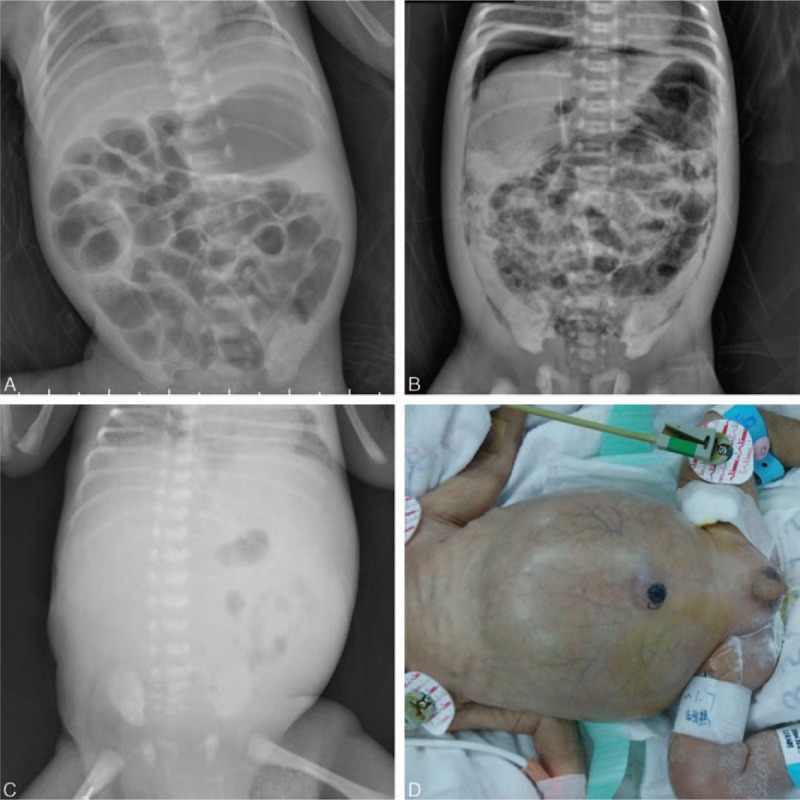
Radiographic findings of necrotizing enterocolitis:
A: Portal venous gas and pneumatosis intestinalis
B: Pneumoperitoneum (air below both diaphragms) and pneumatosis intestinalis
C: Gasless abdomen
D: An infant with a distended abdomen and periumbilical erythema
The following studies are nonspecific, but they may support the diagnosis and demonstrate the severity of the disease.
The following table provides staging Staging Methods which attempt to express in replicable terms the extent of the neoplasm in the patient. Grading, Staging, and Metastasis criteria used to enhance recognition of NEC and assess its severity.
| Stage | Classification | Clinical signs | Radiologic signs |
|---|---|---|---|
| IA | Suspected NEC |
|
Normal to mild intestinal dilation |
| IB | In addition to the above findings: hematochezia Hematochezia Gastrointestinal Bleeding | ||
| IIA |
|
In addition to the above findings:
|
|
| IIB |
|
In addition to the above findings:
|
In addition to the above findings:
|
| IIIA |
|
In addition to the above findings:
|
In addition to the above findings: ascites Ascites Ascites is the pathologic accumulation of fluid within the peritoneal cavity that occurs due to an osmotic and/or hydrostatic pressure imbalance secondary to portal hypertension (cirrhosis, heart failure) or non-portal hypertension (hypoalbuminemia, malignancy, infection). Ascites |
| IIIB |
|
In addition to the above findings: pneumoperitoneum Pneumoperitoneum A condition with trapped gas or air in the peritoneal cavity, usually secondary to perforation of the internal organs such as the lung and the gastrointestinal tract, or to recent surgery. Pneumoperitoneum may be purposely introduced to aid radiological examination. Perforated Viscus |
Early and aggressive treatment is necessary.
Medical management:
Surgical intervention: