Cancer is the 2nd leading cause of death in the US after cardiovascular disease. Many malignancies are treatable or curable, but some may recur. Thus, all malignancies must be assigned a grade and stage in order to guide management and determine prognosis Prognosis A prediction of the probable outcome of a disease based on a individual's condition and the usual course of the disease as seen in similar situations. Non-Hodgkin Lymphomas. The tumor Tumor Inflammation grade classifies a tumor Tumor Inflammation by its histology and is part of the internationally accepted TNM ( tumor Tumor Inflammation, node, metastasis) staging system, which is used to characterize the extent of the disease. Metastatic disease refers to cancer that has spread beyond its primary tumor Tumor Inflammation site.
Last updated: Jan 21, 2026
Contents
Grading is the histologic assessment of tumor Tumor Inflammation cells according to their state of differentiation.
Grading is performed for most types of tumors, but there are specific exceptions.
Tumor Tumor Inflammation grading:
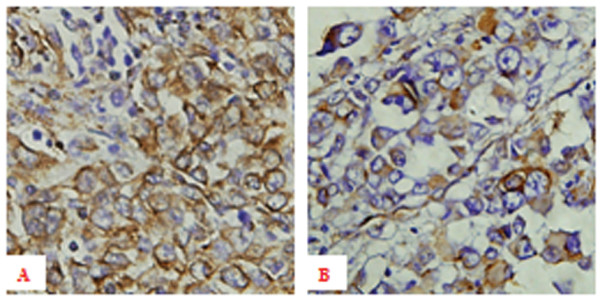
Immunohistochemical expression of vimentin in individuals with melanoma:
A: High expression of vimentin in primary melanoma tissue with hematogenous metastasis ×400
B: Low expression of vimentin in primary melanoma tissue without hematogenous metastasis ×400
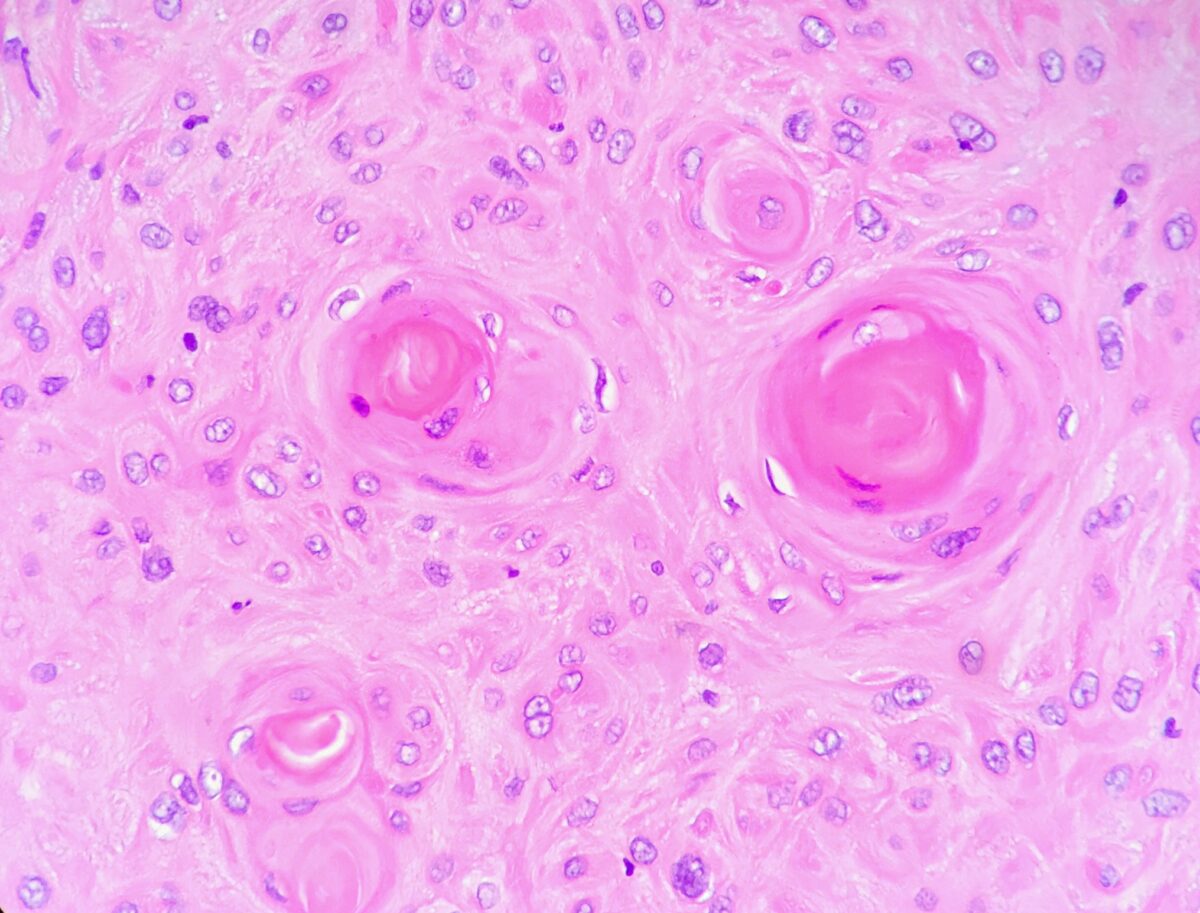
Keratin pearls visualized in a well-differentiated (low grade) squamous cell carcinoma of the larynx
Image: “Keratin pearls in squamous cell carcinoma of the larynx” by Ed Uthman. License: CC BY 2.0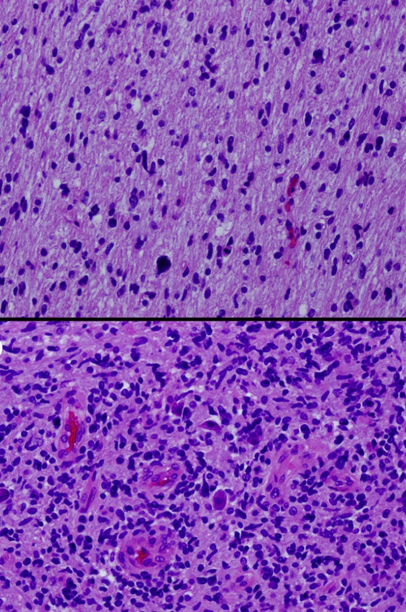
Glioma histology showing hematoxylin and eosin stains from different sections of a single tumor showing low-grade (top) and high-grade (bottom) areas
Image: “Histologic geographic variability of DIPG” by Katherine E. Warren. License: CC BY 3.0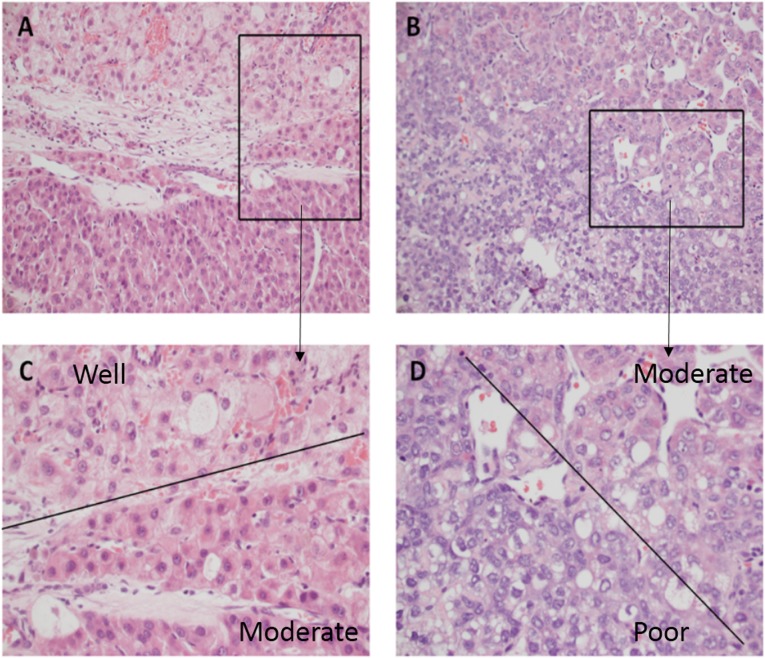
Hepatocellular carcinoma tumor grading: These tumors are commonly very heterogeneous.
C: A portion of the tumor is grade 1 (the morphology of tumor cells were close to that of normal liver cells, with augmentation of nucleus/cytoplasm), while an adjacent area is grade 2 (which contains markedly enlarged oncocytic hepatocytes with some nuclear pleomorphism and angulation).
D: An area of grade 2 is adjacent to an area of grade 3 (tcell shapes are quite irregular, tumor cells show marked pleomorphism).
Staging for cancer describes the extent of the disease and is used to help communicate with other members of the medical and surgical team for treatment decisions and prognosis Prognosis A prediction of the probable outcome of a disease based on a individual’s condition and the usual course of the disease as seen in similar situations. Non-Hodgkin Lymphomas. For example, with colon Colon The large intestines constitute the last portion of the digestive system. The large intestine consists of the cecum, appendix, colon (with ascending, transverse, descending, and sigmoid segments), rectum, and anal canal. The primary function of the colon is to remove water and compact the stool prior to expulsion from the body via the rectum and anal canal. Colon, Cecum, and Appendix: Anatomy cancer, after surgery and pathologic staging, chemotherapy Chemotherapy Osteosarcoma given to individuals with stage III disease eradicates micrometastases, reduces the likelihood of disease recurrence, and increases cure rates.
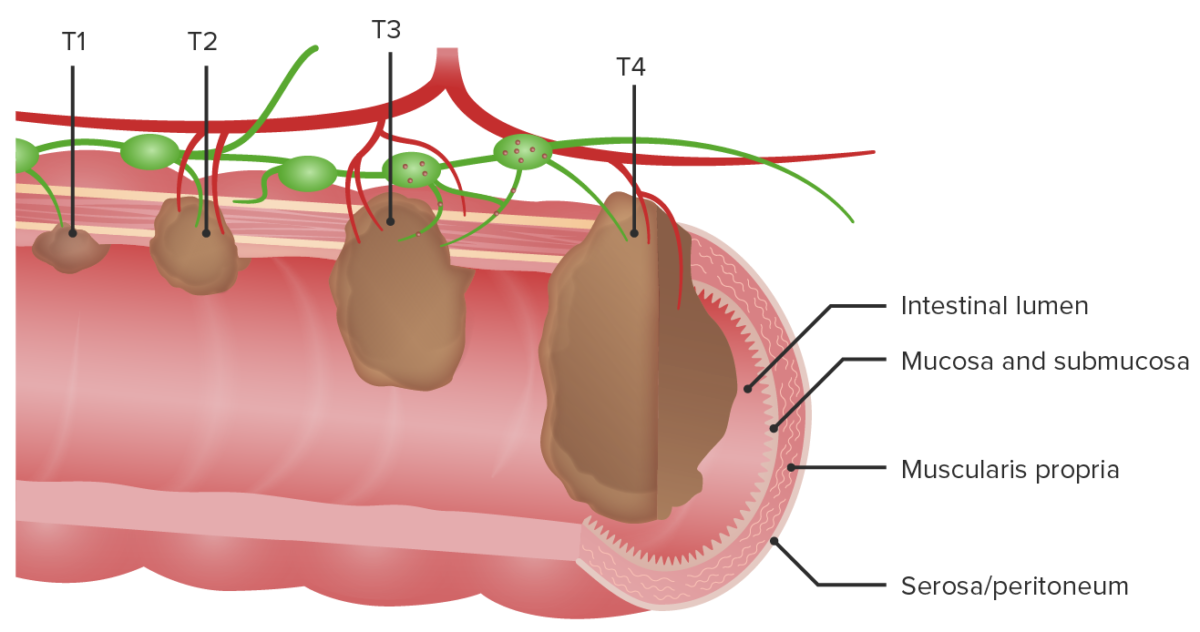
Example of T1–T4 staging for tumor in the colon
T1: Tumor invades the submucosa (through the muscularis mucosa but not into the muscularis propria).
T2: Tumor invades the muscularis propria.
T3: Tumor invades through the muscularis propria into the surrounding tissues.
T4: Direct extension through the serosa, invading the visceral peritoneum or adhering to adjacent organs
Metastasis is the spread of a tumor Tumor Inflammation from its primary site to nearby or distant places.
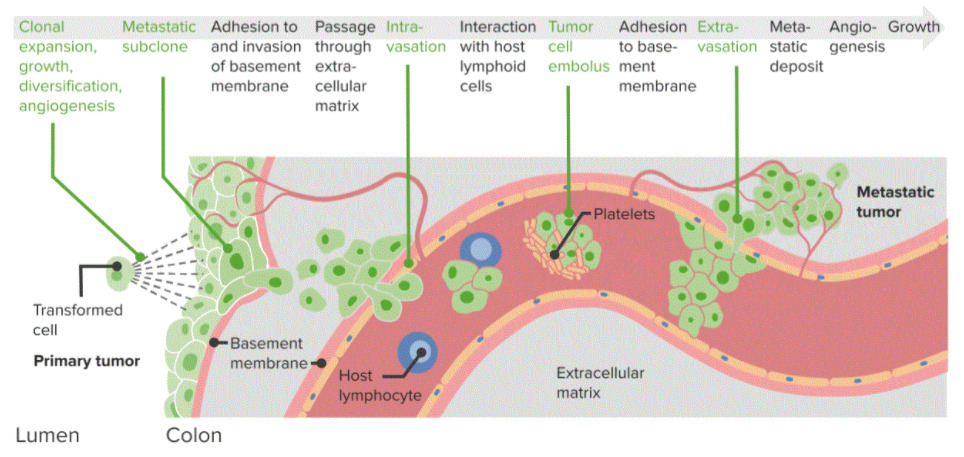
Diagram of cancer metastasis:
Step-by-step process driving tumor spread from a primary tumor to a metastatic tumor. These steps rely upon adhesion and spread through the basement membrane, interaction with lymphoid cells, and extravasation and metastatic deposition.
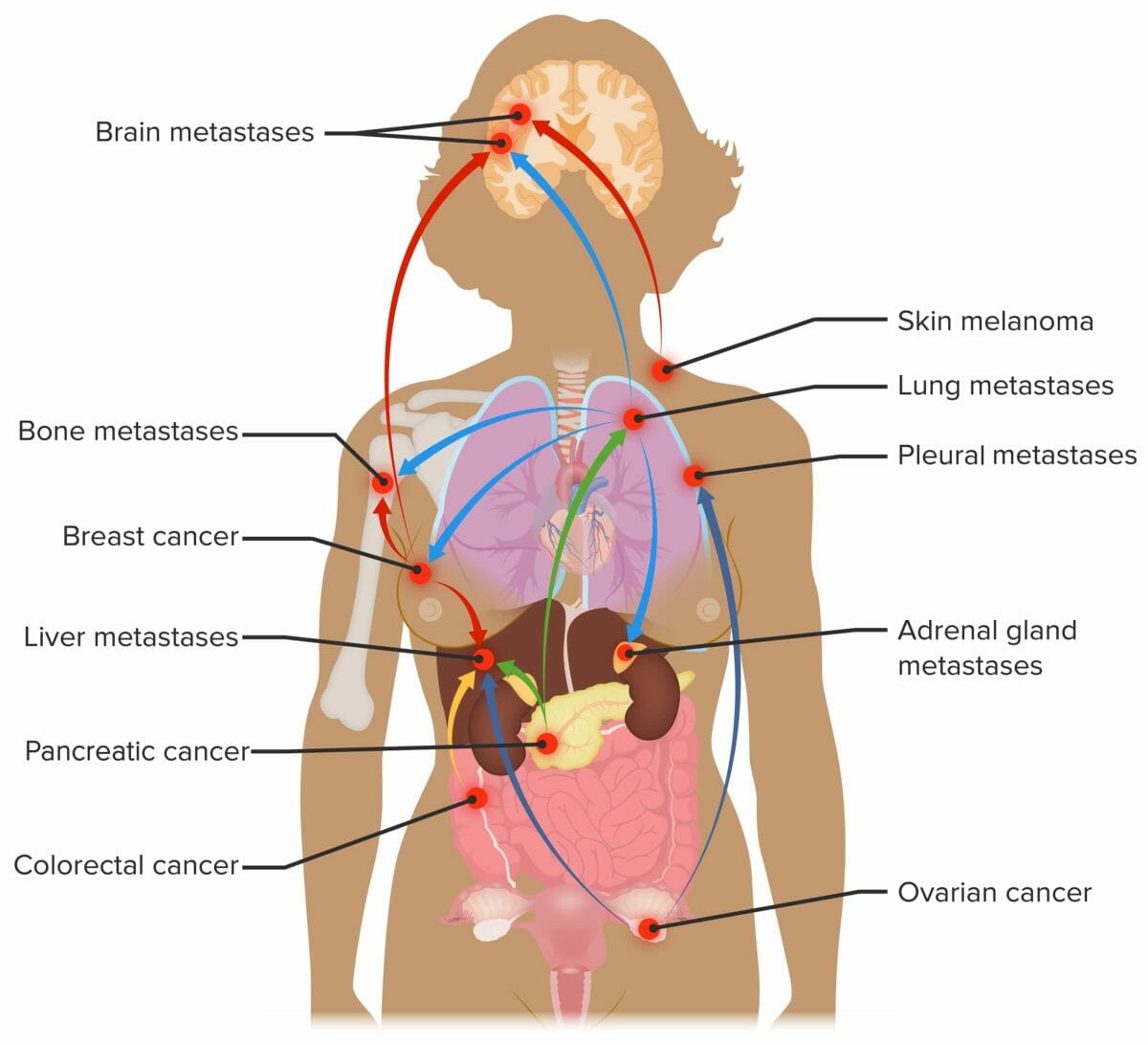
Sites of metastasis:
Common sites of metastasis for cancers with high incidences such as breast, lung, and colorectal cancer