Hypertrophic scars (HSs) and keloids are raised, red, and rigid (3 Rs) scars that develop during cutaneous wound healing Wound healing Wound healing is a physiological process involving tissue repair in response to injury. It involves a complex interaction of various cell types, cytokines, and inflammatory mediators. Wound healing stages include hemostasis, inflammation, granulation, and remodeling. Wound Healing and are characterized by a local abnormal proliferation of fibroblasts Fibroblasts Connective tissue cells which secrete an extracellular matrix rich in collagen and other macromolecules. Sarcoidosis with over-production of collagen Collagen A polypeptide substance comprising about one third of the total protein in mammalian organisms. It is the main constituent of skin; connective tissue; and the organic substance of bones (bone and bones) and teeth (tooth). Connective Tissue: Histology. Over-expression of growth factors, such as transforming growth factor-beta (TGF-β), and decreased production of molecules that promote matrix breakdown, such as matrix metalloproteinases, appear to be involved in the etiology in both an HS and a keloid. Genetics Genetics Genetics is the study of genes and their functions and behaviors. Basic Terms of Genetics has a strong influence on keloid predisposition, with people of darker skin Skin The skin, also referred to as the integumentary system, is the largest organ of the body. The skin is primarily composed of the epidermis (outer layer) and dermis (deep layer). The epidermis is primarily composed of keratinocytes that undergo rapid turnover, while the dermis contains dense layers of connective tissue. Skin: Structure and Functions complexion having a higher predisposition. Keloids occur mostly on the upper torso, shoulders, head, and neck Neck The part of a human or animal body connecting the head to the rest of the body. Peritonsillar Abscess. Treatment of keloids is often ineffective, but moderate success can be achieved with surgical excision, especially when combined with adjuvant Adjuvant Substances that augment, stimulate, activate, potentiate, or modulate the immune response at either the cellular or humoral level. The classical agents (freund's adjuvant, bcg, corynebacterium parvum, et al.) contain bacterial antigens. Some are endogenous (e.g., histamine, interferon, transfer factor, tuftsin, interleukin-1). Their mode of action is either non-specific, resulting in increased immune responsiveness to a wide variety of antigens, or antigen-specific, i.e., affecting a restricted type of immune response to a narrow group of antigens. The therapeutic efficacy of many biological response modifiers is related to their antigen-specific immunoadjuvanticity. Vaccination non-surgical management.
Last updated: Dec 15, 2025
A hypertrophic scar Scar Dermatologic Examination (HS) is a benign Benign Fibroadenoma exuberant overgrowth of fibro-collagenous tissue. An HS occurs in an area of injury and grows rapidly, but does not go beyond the boundaries of the wound and tends to regress.
Keloids are defined as a benign Benign Fibroadenoma overgrowth of fibro-collagenous tissue, occurring in an area of dermal injury or chronic inflammation Chronic Inflammation Inflammation (rarely spontaneously). Unlike HSs, keloids continue to enlarge and grow beyond the original boundaries of the injury.
HSs:
Keloids:
Altered wound healing Wound healing Wound healing is a physiological process involving tissue repair in response to injury. It involves a complex interaction of various cell types, cytokines, and inflammatory mediators. Wound healing stages include hemostasis, inflammation, granulation, and remodeling. Wound Healing causes an imbalance between an increased synthesis Synthesis Polymerase Chain Reaction (PCR) of collagen Collagen A polypeptide substance comprising about one third of the total protein in mammalian organisms. It is the main constituent of skin; connective tissue; and the organic substance of bones (bone and bones) and teeth (tooth). Connective Tissue: Histology and extracellular matrix (ECM) and decreased degradation of collagen Collagen A polypeptide substance comprising about one third of the total protein in mammalian organisms. It is the main constituent of skin; connective tissue; and the organic substance of bones (bone and bones) and teeth (tooth). Connective Tissue: Histology and ECM.
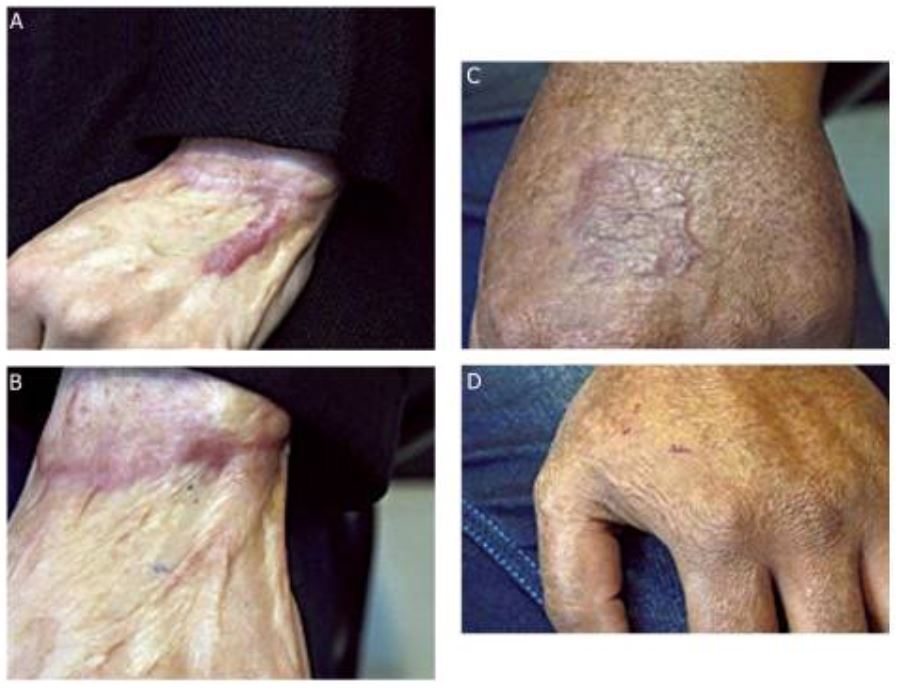
Hypertrophic scars, before and after treatment
The patients developed HSs after full-thickness burns (A and C). Note how the HSs are “raised, red, and rigid” like keloid scars, but unlike keloids, HSs do not extend beyond the area of injury into healthy surrounding skin. Images in B and D show each of the healing sites 2 months after surgical excision and application of autologous fibroblast and keratinocyte cultured cells in fibrin glue to the now-healing wounds.
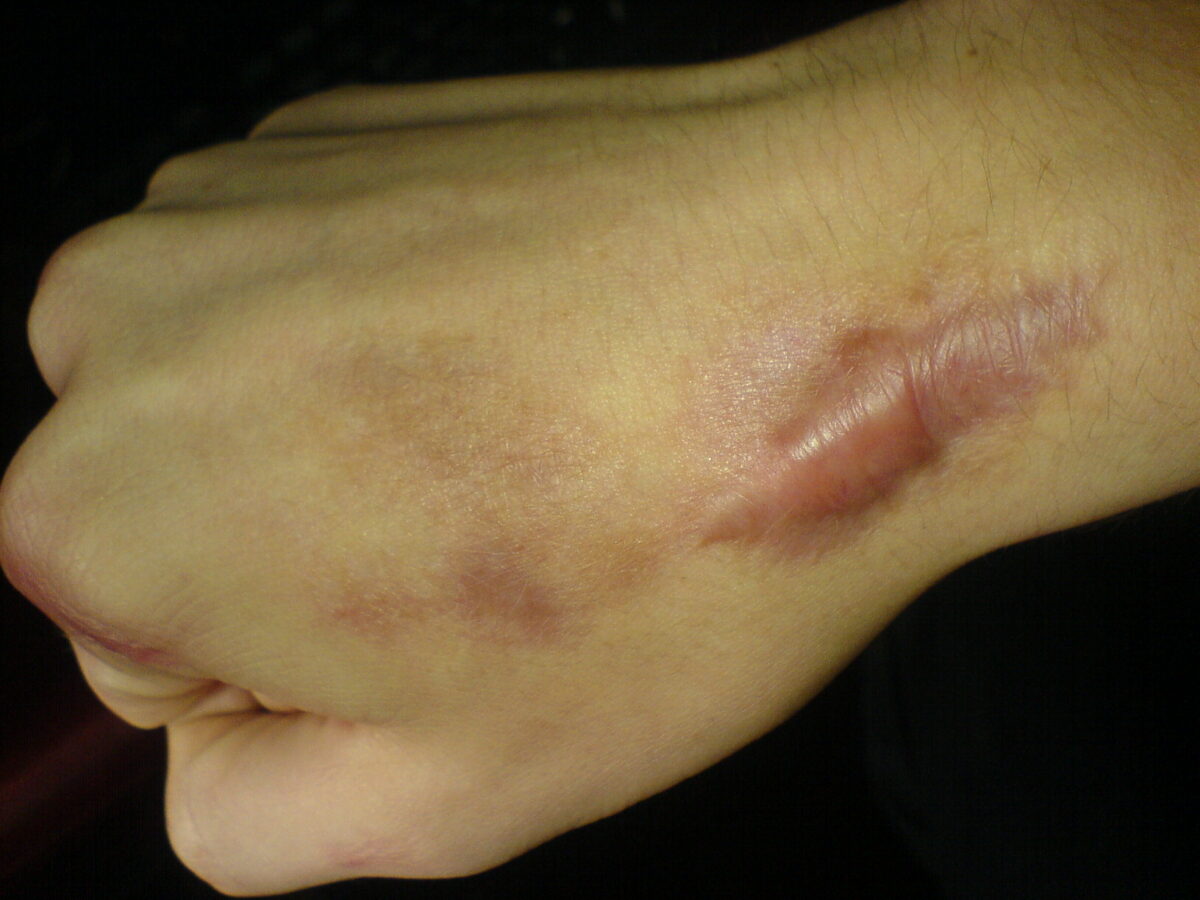
Post-operative keloid on the wrist, showing the classic 3 Rs
The classic 3 Rs common to both keloids and HSs are “raised, red, and rigid.” Unlike an HS, however, a keloid extends beyond the boundaries of the original wound, as demonstrated here. Note the smooth, dome-shaped, rounded surface.
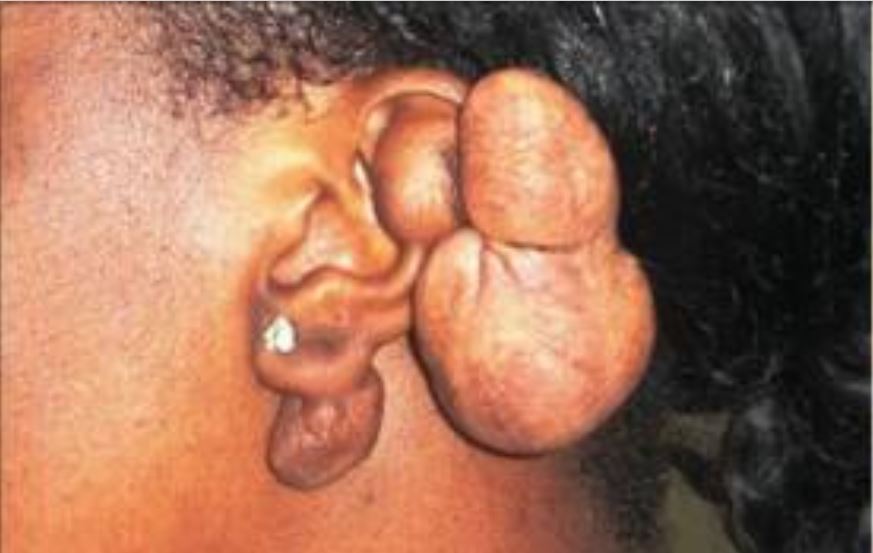
Keloid formed on the left ear
This 23-year-old woman had a history of ear piercing since the age of 16 years, followed by keloid formation. She underwent surgery in 2002 without post-operative radiation and did not follow up regularly with her treating surgeon. The lesion recurred soon after surgery.
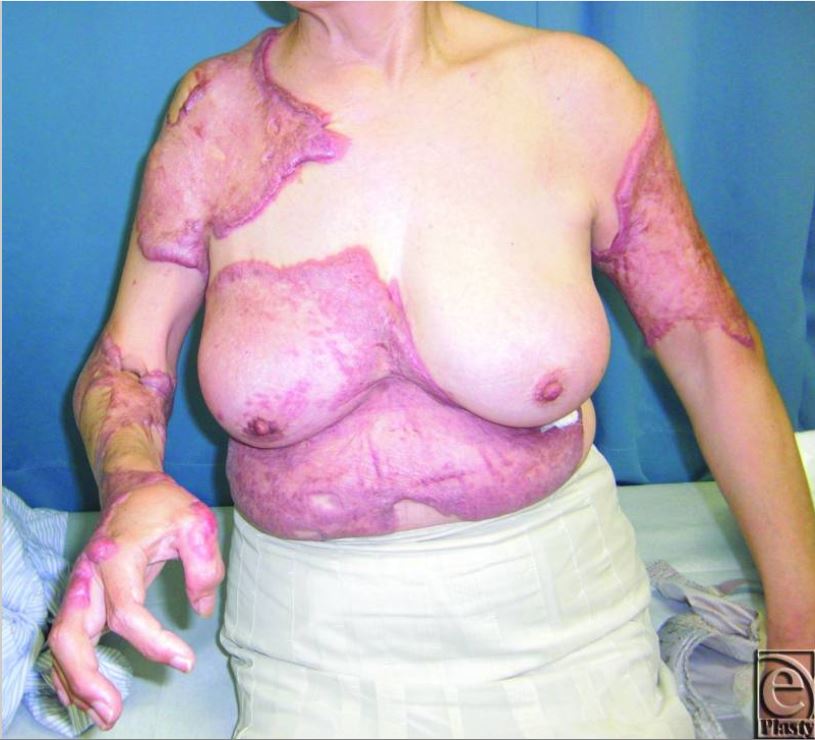
A case of severe spontaneously occurring widespread keloids with hypertension
This 63-year-old woman started to develop keloids on her trunk and upper limbs when she was an elementary school student. During her 1st examination in the clinic, her blood pressure was 150/95 mm Hg.
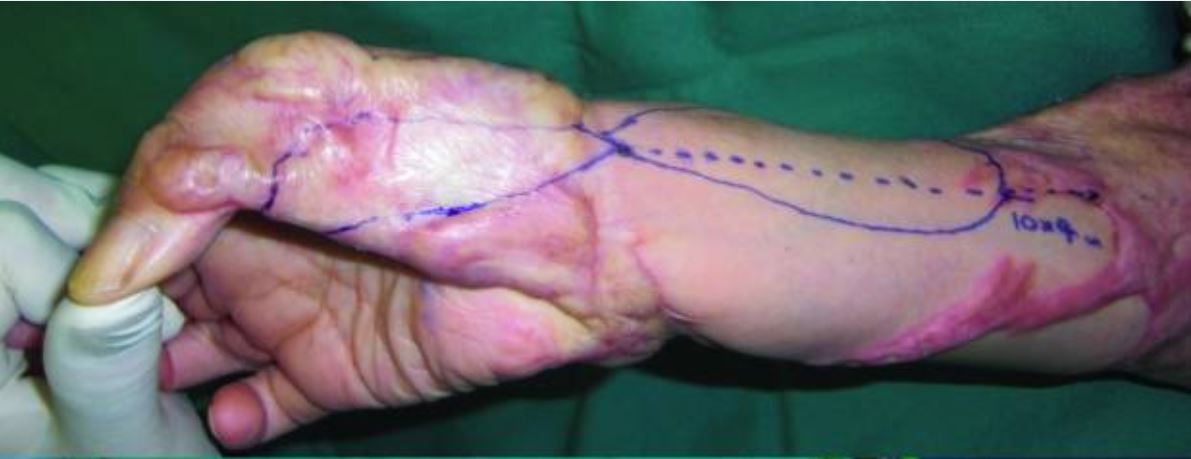
Preoperative views of severe keloids in the right arm and hand of the 63-year-old woman in the prior photograph
The woman underwent surgical treatment with radiation adjuvant therapy. A distally based radial artery flap was selected for the thumb and wrist. Starting on the day after surgery, both the donor and recipient sites were subjected to 4 MeV electron beam irradiation (15 Gy/3 fractions for 3 days). See the 2-year post-operative views in the next image.
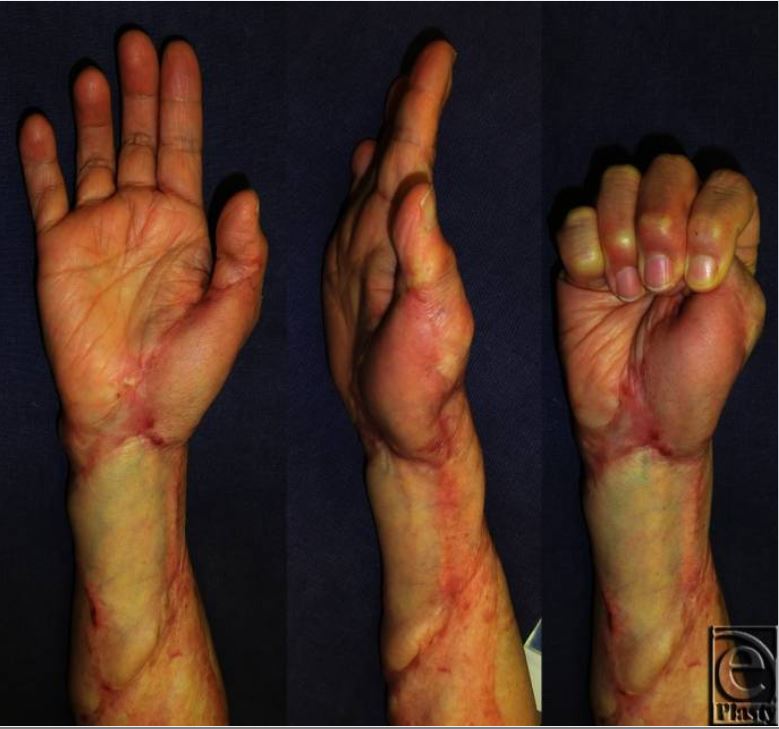
Two-year post-operative views of contractures noted in the prior image
The range of motion of all joints recovered fully, and there was no recurrence of the keloids or scar contractures.
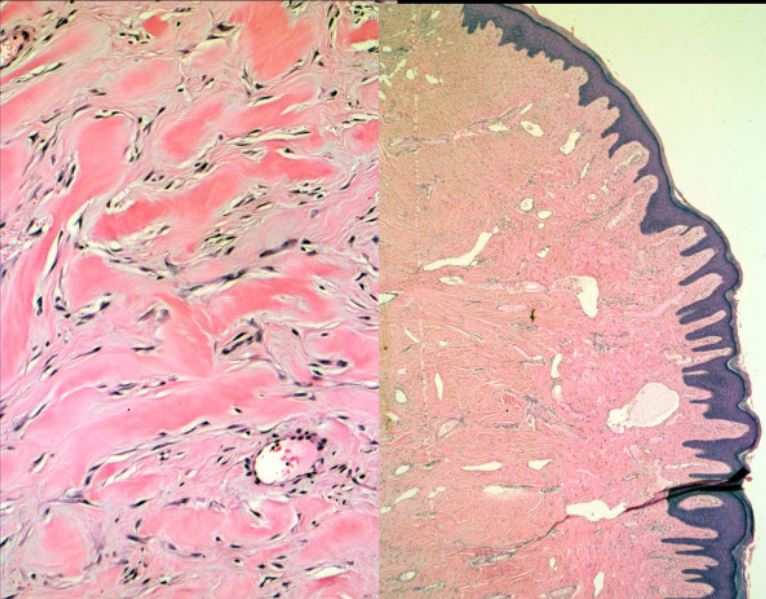
High-power and low-power microphotographs of an excised keloid
The high-power view shows disorganized, thick (“glassy”) collagen bundles containing a reduced number of myofibroblasts compared with normal or HS. The low-power view exemplifies the scant vascularity and dilated blood vessels of a keloid.
| Category | HSs | Keloids |
|---|---|---|
| Epidemiology |
|
|
| Predilection sites |
|
|
| Time course |
|
|
| Appearance |
|
|
| Histologic characteristics |
|
|
| Category | HSs | Keloids |
|---|---|---|
| Description |
|
|
| Predilection sites |
|
|
| Triggers |
|
|
| Growth history |
|
|
| Recurrence after excision |
|
|
For persons with a history of hypertrophic or keloid scarring Scarring Inflammation: