Familial juvenile polyposis, also known as juvenile polyposis syndrome, is an autosomal dominant Autosomal dominant Autosomal inheritance, both dominant and recessive, refers to the transmission of genes from the 22 autosomal chromosomes. Autosomal dominant diseases are expressed when only 1 copy of the dominant allele is inherited. Autosomal Recessive and Autosomal Dominant Inheritance condition characterized by the growth of hamartomatous polyps (juvenile type) in the gastrointestinal tract, mostly in the colon Colon The large intestines constitute the last portion of the digestive system. The large intestine consists of the cecum, appendix, colon (with ascending, transverse, descending, and sigmoid segments), rectum, and anal canal. The primary function of the colon is to remove water and compact the stool prior to expulsion from the body via the rectum and anal canal. Colon, Cecum, and Appendix: Anatomy. Polyposis syndromes are a group of inherited or acquired conditions characterized by the growth of polyps in the GI tract, and are associated with other extracolonic features. These syndromes are caused by mutations in specific genes Genes A category of nucleic acid sequences that function as units of heredity and which code for the basic instructions for the development, reproduction, and maintenance of organisms. DNA Types and Structure associated with tumor Tumor Inflammation suppression Suppression Defense Mechanisms or cell cycle Cell cycle The phases of the cell cycle include interphase (G1, S, and G2) and mitosis (prophase, metaphase, anaphase, and telophase). The cell's progression through these phases is punctuated by checkpoints regulated by cyclins, cyclin-dependent kinases, tumor suppressors, and their antagonists. Cell Cycle regulation. Familial juvenile polyposis is commonly associated with mutations in the SMAD4 ( chromosome Chromosome In a prokaryotic cell or in the nucleus of a eukaryotic cell, a structure consisting of or containing DNA which carries the genetic information essential to the cell. Basic Terms of Genetics 18q) and BMPR1A ( chromosome Chromosome In a prokaryotic cell or in the nucleus of a eukaryotic cell, a structure consisting of or containing DNA which carries the genetic information essential to the cell. Basic Terms of Genetics 10q) genes Genes A category of nucleic acid sequences that function as units of heredity and which code for the basic instructions for the development, reproduction, and maintenance of organisms. DNA Types and Structure. Diagnosis is made by visualizing 5 or more juvenile polyps on colonoscopy Colonoscopy Endoscopic examination, therapy or surgery of the luminal surface of the colon. Colorectal Cancer Screening, multiple juvenile polyps in other areas of the GI tract, or any number of juvenile polyps plus a positive family history Family History Adult Health Maintenance. Management is surgical in order to reduce the likelihood of GI bleeding and obstruction.
Last updated: Jan 16, 2024
Familial juvenile polyposis, or juvenile polyposis syndrome (JPS), is an autosomal dominant Autosomal dominant Autosomal inheritance, both dominant and recessive, refers to the transmission of genes from the 22 autosomal chromosomes. Autosomal dominant diseases are expressed when only 1 copy of the dominant allele is inherited. Autosomal Recessive and Autosomal Dominant Inheritance condition characterized by the growth of hamartomatous polyps (juvenile type) in the colon Colon The large intestines constitute the last portion of the digestive system. The large intestine consists of the cecum, appendix, colon (with ascending, transverse, descending, and sigmoid segments), rectum, and anal canal. The primary function of the colon is to remove water and compact the stool prior to expulsion from the body via the rectum and anal canal. Colon, Cecum, and Appendix: Anatomy.
Other manifestations can include:
Diagnosis is made by visualizing > 5 polyps on colonoscopy Colonoscopy Endoscopic examination, therapy or surgery of the luminal surface of the colon. Colorectal Cancer Screening, multiple juvenile polyps in other areas of the GI tract, or any number of polyps with a positive family history Family History Adult Health Maintenance.
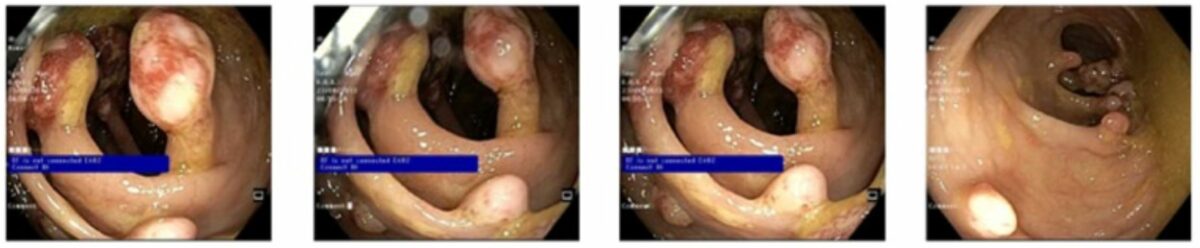
Multiple pedunculated and sessile, nonbleeding juvenile polyps
Image: “Multiple pedunculated and sessile, nonbleeding polyps” by Amna Ahmed and Badr Alsaleem. License: CC BY 4.0