Sarcoidosis is a multisystem inflammatory disease that causes noncaseating granulomas Noncaseating granulomas Crohn Disease. The exact etiology is unknown. Sarcoidosis usually affects the lungs Lungs Lungs are the main organs of the respiratory system. Lungs are paired viscera located in the thoracic cavity and are composed of spongy tissue. The primary function of the lungs is to oxygenate blood and eliminate CO2. Lungs: Anatomy and thoracic lymph nodes Lymph Nodes They are oval or bean shaped bodies (1 - 30 mm in diameter) located along the lymphatic system. Lymphatic Drainage System: Anatomy, but it can also affect almost every system in the body, including the skin Skin The skin, also referred to as the integumentary system, is the largest organ of the body. The skin is primarily composed of the epidermis (outer layer) and dermis (deep layer). The epidermis is primarily composed of keratinocytes that undergo rapid turnover, while the dermis contains dense layers of connective tissue. Skin: Structure and Functions, heart, and eyes, most commonly. Acutely, sarcoidosis presents with lymphadenopathy Lymphadenopathy Lymphadenopathy is lymph node enlargement (> 1 cm) and is benign and self-limited in most patients. Etiologies include malignancy, infection, and autoimmune disorders, as well as iatrogenic causes such as the use of certain medications. Generalized lymphadenopathy often indicates underlying systemic disease. Lymphadenopathy, fever Fever Fever is defined as a measured body temperature of at least 38°C (100.4°F). Fever is caused by circulating endogenous and/or exogenous pyrogens that increase levels of prostaglandin E2 in the hypothalamus. Fever is commonly associated with chills, rigors, sweating, and flushing of the skin. Fever, malaise Malaise Tick-borne Encephalitis Virus, joint pains, a panniculitis Panniculitis General term for inflammation of adipose tissue, usually of the skin, characterized by reddened subcutaneous nodules. Erythema Nodosum on the shins Shins Erythema Nodosum known as erythema Erythema Redness of the skin produced by congestion of the capillaries. This condition may result from a variety of disease processes. Chalazion nodosum, and occasionally cough and shortness of breath Shortness of breath Dyspnea is the subjective sensation of breathing discomfort. Dyspnea is a normal manifestation of heavy physical or psychological exertion, but also may be caused by underlying conditions (both pulmonary and extrapulmonary). Dyspnea. Chronic pulmonary sarcoidosis presents with an insidious onset of dyspnea Dyspnea Dyspnea is the subjective sensation of breathing discomfort. Dyspnea is a normal manifestation of heavy physical or psychological exertion, but also may be caused by underlying conditions (both pulmonary and extrapulmonary). Dyspnea, cough, chest pain Pain An unpleasant sensation induced by noxious stimuli which are detected by nerve endings of nociceptive neurons. Pain: Types and Pathways, and a variety of other symptoms depending on the organ systems involved. Diagnosis often requires a biopsy Biopsy Removal and pathologic examination of specimens from the living body. Ewing Sarcoma of the granulomas. Management includes observation, NSAIDs NSAIDS Primary vs Secondary Headaches, glucocorticoids Glucocorticoids Glucocorticoids are a class within the corticosteroid family. Glucocorticoids are chemically and functionally similar to endogenous cortisol. There are a wide array of indications, which primarily benefit from the antiinflammatory and immunosuppressive effects of this class of drugs. Glucocorticoids, and potentially one of several steroid-sparing agents. Acute sarcoidosis is usually self-limiting Self-Limiting Meningitis in Children with an excellent prognosis Prognosis A prediction of the probable outcome of a disease based on a individual's condition and the usual course of the disease as seen in similar situations. Non-Hodgkin Lymphomas, but chronic sarcoidosis can lead to severe pulmonary fibrosis Fibrosis Any pathological condition where fibrous connective tissue invades any organ, usually as a consequence of inflammation or other injury. Bronchiolitis Obliterans.
Last updated: Dec 15, 2025
Sarcoidosis is a multisystem inflammatory disease characterized by the formation of noncaseating granulomas Noncaseating granulomas Crohn Disease that are most likely caused by a cell-mediated immune reaction of unknown etiology.
Pulmonary sarcoidosis is a restrictive interstitial lung disease with granuloma formation in the:
Extrapulmonary sarcoidosis is characterized by granuloma formation in:
Sarcoidosis may be acute or chronic:
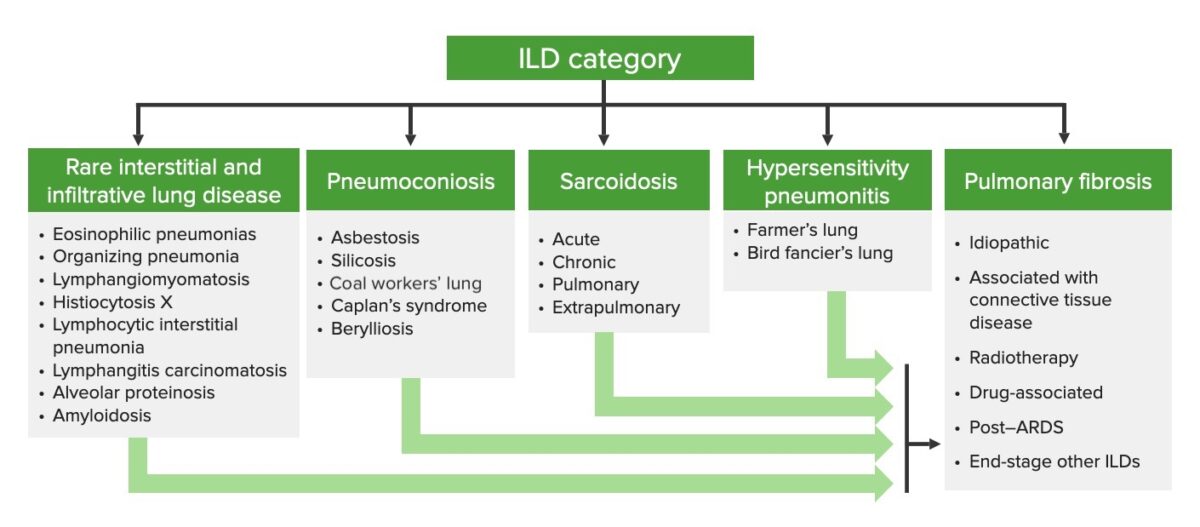
Categorization of interstitial lung diseases
ILD: interstitial lung disease
Etiology is undetermined but is most likely multifactorial.
Theory: An infectious or environmental agent triggers a cell-mediated inflammatory immune response in a genetically susceptible host, leading to granuloma formation.
Potential triggering exposures:
Genetic predispositions:
Sarcoidosis is the result of a cell-mediated immune reaction.
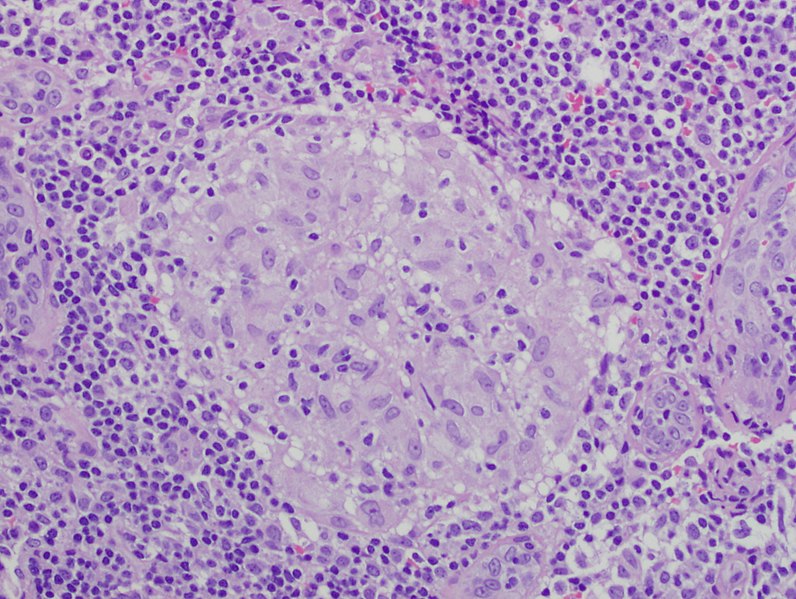
Noncaseating granuloma
Image: “Granuloma 20x” by Mutleysmith. License: Public DomainSarcoidosis usually progresses slowly, with few symptoms initially. Symptoms appear as an increasing number of granulomas begin to affect organ function.
| Organ system | Clinical presentation of sarcoidosis |
|---|---|
| Systemic symptoms (typically in acute presentations) |
|
| Pulmonary |
|
| Lymph Lymph The interstitial fluid that is in the lymphatic system. Secondary Lymphatic Organs |
|
| Ocular |
|
| Cutaneous |
|
| Musculoskeletal |
|
| Cardiac |
|
| Nervous system Nervous system The nervous system is a small and complex system that consists of an intricate network of neural cells (or neurons) and even more glial cells (for support and insulation). It is divided according to its anatomical components as well as its functional characteristics. The brain and spinal cord are referred to as the central nervous system, and the branches of nerves from these structures are referred to as the peripheral nervous system. Nervous System: Anatomy, Structure, and Classification |
|
| Other potential sites of involvement |
|
An acute presentation of sarcoidosis with a classic triad of symptoms:

Erythema nodosum
Image: “Bilateral shin lesions” by MRC Clinical Trials Unit at UCL, Aviation House, 125 Kingsway, London, WC2B 6NH, UK. License: CC BY 4.0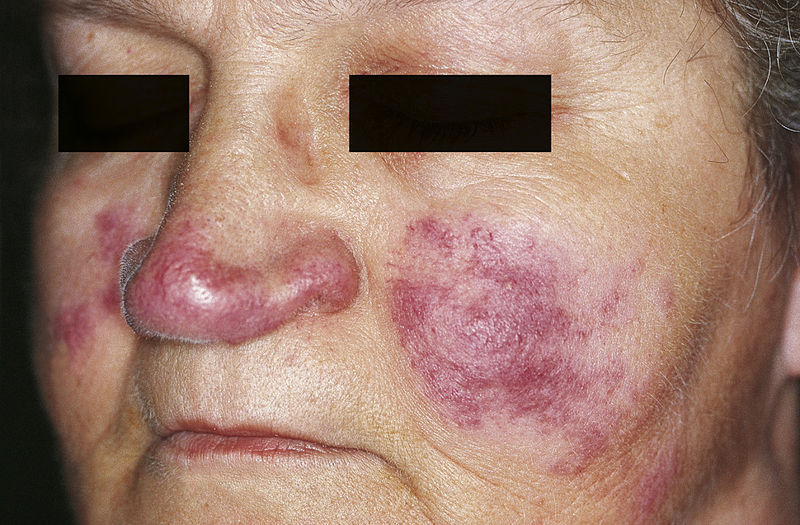
Lupus pernio: cutaneous lesions of sarcoidosis
Image: “Cutaneous lesions of sarcoidosis” by M. Sand et al. License: CC BY 2.0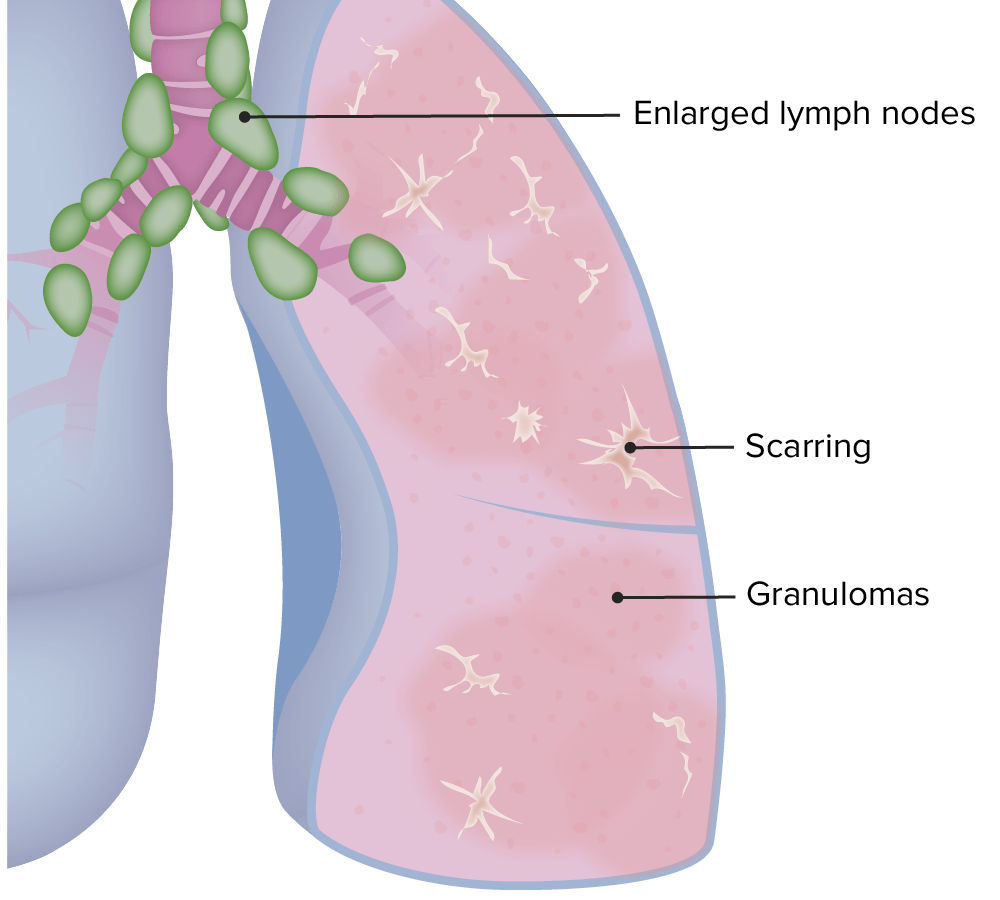
Pulmonary manifestations of sarcoidosis
Image by Lecturio.Diagnosing sarcoidosis typically requires:
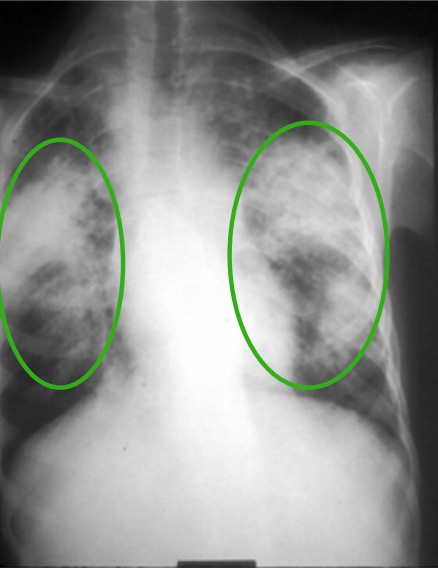
Chest X-ray in a patient with chronic sarcoidosis:
Green circles denote areas of upper lobe interstitial infiltrates. There is also distortion of the hilum.
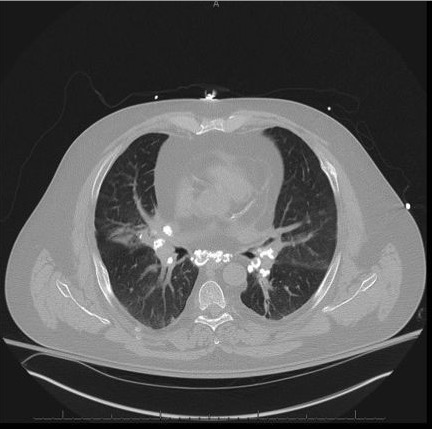
Non–contrast-enhanced chest CT scan demonstrating pulmonary nodules and calcified mediastinal and hilar lymphadenopathy
Image: “Noncontrast-computed tomography scan” by Penn State/Milton S, Hershey Medical Center Department of Medicine, 500 University Drive, Hershey PA 17033, Pennsylvania, USA. License: CC BY 4.0
Noncaseating granuloma
Image: “Granuloma 20x” by Mutleysmith. License: Public DomainObtained to assess severity of respiratory involvement and monitor disease; less helpful in actually establishing diagnosis
These tests are important in assessing and monitoring the extent of disease rather than making the diagnosis. The following findings are often seen in sarcoidosis.
| Stage | Chest X-ray X-ray Penetrating electromagnetic radiation emitted when the inner orbital electrons of an atom are excited and release radiant energy. X-ray wavelengths range from 1 pm to 10 nm. Hard x-rays are the higher energy, shorter wavelength x-rays. Soft x-rays or grenz rays are less energetic and longer in wavelength. The short wavelength end of the x-ray spectrum overlaps the gamma rays wavelength range. The distinction between gamma rays and x-rays is based on their radiation source. Pulmonary Function Tests | Symptoms | Frequency |
|---|---|---|---|
| 0 | Extrapulmonary disease Extrapulmonary disease Blastomyces/Blastomycosis only | None | 10% |
| 1 | BHL only | None or mild cough | 50% |
| 2 | BHL plus parenchymal infiltrates | None, dyspnea Dyspnea Dyspnea is the subjective sensation of breathing discomfort. Dyspnea is a normal manifestation of heavy physical or psychological exertion, but also may be caused by underlying conditions (both pulmonary and extrapulmonary). Dyspnea, or cough | 25% |
| 3 | Parenchymal infiltrates only | Dyspnea Dyspnea Dyspnea is the subjective sensation of breathing discomfort. Dyspnea is a normal manifestation of heavy physical or psychological exertion, but also may be caused by underlying conditions (both pulmonary and extrapulmonary). Dyspnea | 10% |
| 4 | Advanced pulmonary fibrosis Fibrosis Any pathological condition where fibrous connective tissue invades any organ, usually as a consequence of inflammation or other injury. Bronchiolitis Obliterans | Dyspnea Dyspnea Dyspnea is the subjective sensation of breathing discomfort. Dyspnea is a normal manifestation of heavy physical or psychological exertion, but also may be caused by underlying conditions (both pulmonary and extrapulmonary). Dyspnea | 5% |
Management is based on stage and location of disease and includes:
| Stage | Remission Remission A spontaneous diminution or abatement of a disease over time, without formal treatment. Cluster Headaches rate |
|---|---|
| 0 | – |
| 1 | 90% |
| 2 | 70% |
| 3 | 20% |
| 4 | < 5% |