The immune system Immune system The body's defense mechanism against foreign organisms or substances and deviant native cells. It includes the humoral immune response and the cell-mediated response and consists of a complex of interrelated cellular, molecular, and genetic components. Primary Lymphatic Organs is equipped with a varied repertoire of defense mechanisms Defense mechanisms Defense mechanisms are normal subconscious means of resolving inner conflicts between an individual's subjective moral sense and their thoughts, feelings, or actions. Defense mechanisms serve to protect the self from unpleasant feelings (anxiety, shame, and/or guilt) and are divided into pathologic, immature, mature, neurotic, and other types. Defense Mechanisms against pathogens. Functionally, the immune system Immune system The body's defense mechanism against foreign organisms or substances and deviant native cells. It includes the humoral immune response and the cell-mediated response and consists of a complex of interrelated cellular, molecular, and genetic components. Primary Lymphatic Organs is differentiated into the innate and adaptive components. Innate immunity, the 1st protective layer of defense, is a system that recognizes threatening microbes, distinguishes self-tissues from pathogens, and subsequently eliminates the foreign invaders. The response is nonspecific and uses different layers of protection: barriers such as the skin Skin The skin, also referred to as the integumentary system, is the largest organ of the body. The skin is primarily composed of the epidermis (outer layer) and dermis (deep layer). The epidermis is primarily composed of keratinocytes that undergo rapid turnover, while the dermis contains dense layers of connective tissue. Skin: Structure and Functions, pattern recognition receptors Receptors Receptors are proteins located either on the surface of or within a cell that can bind to signaling molecules known as ligands (e.g., hormones) and cause some type of response within the cell. Receptors (PRRs) as well as circulating proteins Proteins Linear polypeptides that are synthesized on ribosomes and may be further modified, crosslinked, cleaved, or assembled into complex proteins with several subunits. The specific sequence of amino acids determines the shape the polypeptide will take, during protein folding, and the function of the protein. Energy Homeostasis (e.g., complement) that relay signals of a threat, and immune cells that help eliminate the microbe. Pathogen-associated molecular patterns Pathogen-Associated Molecular Patterns Sepsis and Septic Shock ( PAMPs PAMPs Sepsis and Septic Shock) in microorganisms and damage-associated molecular patterns Damage-Associated Molecular Patterns Sepsis and Septic Shock (DAMPs) from injured tissues are identified, and the appropriate cells are recruited. Involved cells include phagocytes and accessory cells. The offending pathogens are engulfed by phagocytes for destruction. In antigen-presenting cells Antigen-presenting cells A heterogeneous group of immunocompetent cells that mediate the cellular immune response by processing and presenting antigens to the T-cells. Traditional antigen-presenting cells include macrophages; dendritic cells; langerhans cells; and B-lymphocytes. Follicular dendritic cells are not traditional antigen-presenting cells, but because they hold antigen on their cell surface in the form of immune complexes for b-cell recognition they are considered so by some authors. Adaptive Immune Response (the most potent of which is the dendritic cell), parts of the pathogen material or peptides are transported to the cell surface. Through a unique antigen-loading mechanism specific to MHC I or II, the processed antigen Antigen Substances that are recognized by the immune system and induce an immune reaction. Vaccination peptides are then presented to the appropriate T cells T cells Lymphocytes responsible for cell-mediated immunity. Two types have been identified - cytotoxic (t-lymphocytes, cytotoxic) and helper T-lymphocytes (t-lymphocytes, helper-inducer). They are formed when lymphocytes circulate through the thymus gland and differentiate to thymocytes. When exposed to an antigen, they divide rapidly and produce large numbers of new T cells sensitized to that antigen. T cells: Types and Functions, leading to T-cell activation. This interaction links innate immunity with adaptive immunity.
Last updated: Apr 18, 2023
The immune system Immune system The body’s defense mechanism against foreign organisms or substances and deviant native cells. It includes the humoral immune response and the cell-mediated response and consists of a complex of interrelated cellular, molecular, and genetic components. Primary Lymphatic Organs provides defense (immunity) against invading pathogens ranging from viruses Viruses Minute infectious agents whose genomes are composed of DNA or RNA, but not both. They are characterized by a lack of independent metabolism and the inability to replicate outside living host cells. Virology to parasites, and components are interconnected by blood and the lymphatic circulation Circulation The movement of the blood as it is pumped through the cardiovascular system. ABCDE Assessment.
There are 2 lines of defense Lines of Defense Inflammation (that overlap):
| Innate immunity | Adaptive immunity | |
|---|---|---|
| Genetics Genetics Genetics is the study of genes and their functions and behaviors. Basic Terms of Genetics | Germline encoded | Gene Gene A category of nucleic acid sequences that function as units of heredity and which code for the basic instructions for the development, reproduction, and maintenance of organisms. Basic Terms of Genetics rearrangements involved in lymphocyte development |
| Immune response | Nonspecific | Highly specific |
| Timing of response | Immediate (minutes to hours) | Develops over a longer period of time |
| Memory Memory Complex mental function having four distinct phases: (1) memorizing or learning, (2) retention, (3) recall, and (4) recognition. Clinically, it is usually subdivided into immediate, recent, and remote memory. Psychiatric Assessment response | None | Responds quickly upon recognition of antigen Antigen Substances that are recognized by the immune system and induce an immune reaction. Vaccination with memory Memory Complex mental function having four distinct phases: (1) memorizing or learning, (2) retention, (3) recall, and (4) recognition. Clinically, it is usually subdivided into immediate, recent, and remote memory. Psychiatric Assessment response |
| Recognition of pathogen | Pattern recognition receptors Receptors Receptors are proteins located either on the surface of or within a cell that can bind to signaling molecules known as ligands (e.g., hormones) and cause some type of response within the cell. Receptors (PRRs) such as TLRs recognize pathogen-associated molecular patterns Pathogen-Associated Molecular Patterns Sepsis and Septic Shock ( PAMPs PAMPs Sepsis and Septic Shock) |
|
| Components |
|
|
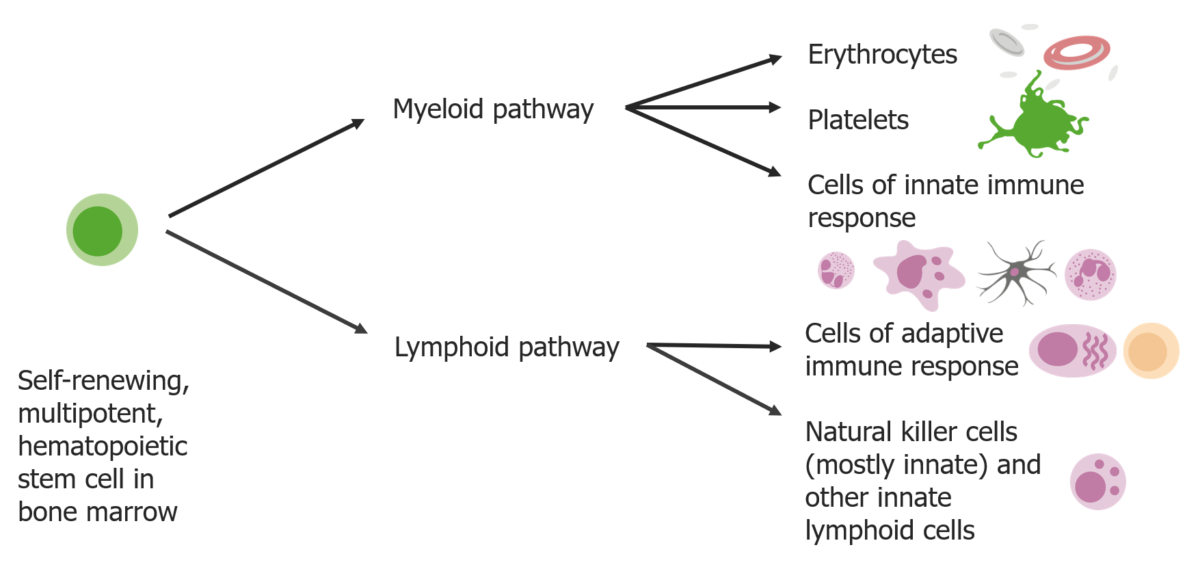
Stem cells differentiate into 2 pathways:
Myeloid pathways produce erythrocytes, platelets, and cells of the innate immune response. Lymphoid pathways produce the cells of adaptive response and natural killer cells.
Phagocytes “eat” the foreign material, and help detect, clear, and repair damaged tissue, recognizing pathogens via PRRs or opsonization (by complement or immunoglobulins Immunoglobulins Immunoglobulins (Igs), also known as antibodies, are glycoprotein molecules produced by plasma cells that act in immune responses by recognizing and binding particular antigens. The various Ig classes are IgG (the most abundant), IgM, IgE, IgD, and IgA, which differ in their biologic features, structure, target specificity, and distribution. Immunoglobulins: Types and Functions).
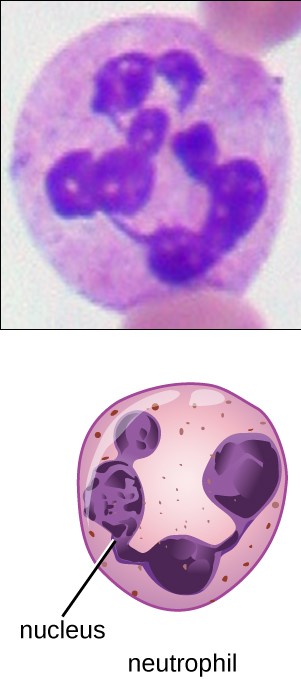
Neutrophil:
Granulocyte with multilobed nucleus and fine faintly pink granules
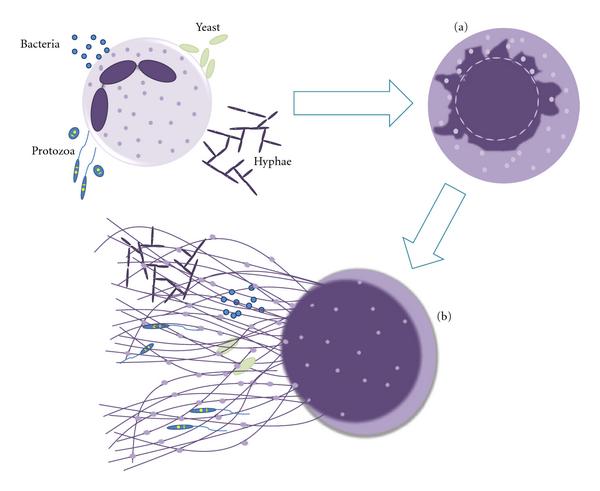
Mechanism of neutrophil extracellular trap (NET) release:
Neutrophils are stimulated by contact with bacteria, protozoa, fungi (yeast and hyphae forms), which leads to: (a) Ultrastructural alterations of nuclear shape with chromatin decondensation and a swollen and fragmented nuclear membrane, which allows the association of granules and cytoplasmic proteins with the chromatin, and (b) release of extracellular structures consisting of a DNA backbone, decorated with histones, neutrophil, granular, and cytoplasmic proteins (NETs), which ensnare and kill microorganisms.
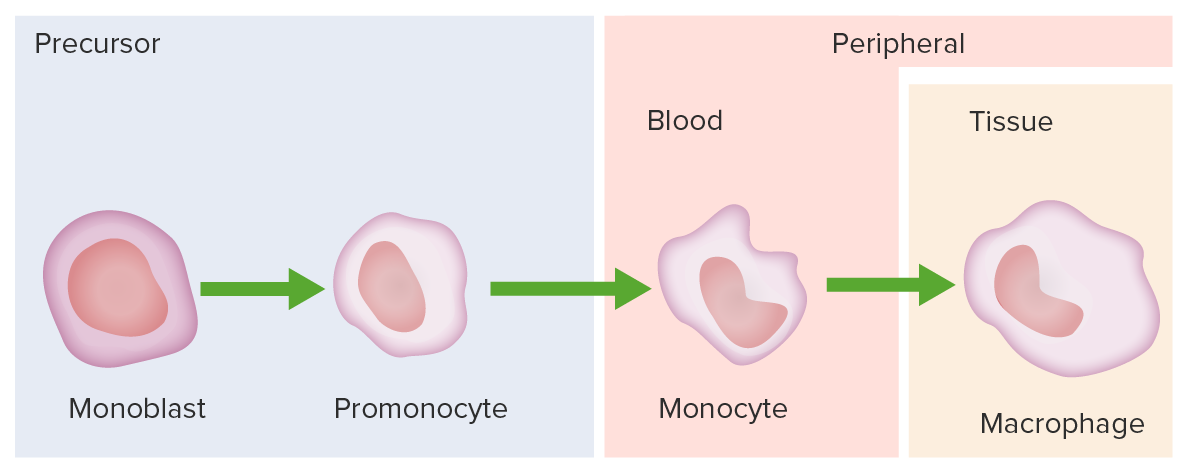
Monocyte development starts from hematopoietic stem cells (HSCs) and progresses through stages to the colony-forming unit granulocyte-macrophage (CFU-GM):
The 1st monocyte precursor is the monoblast, which has a round or oval nucleus.
The promonocyte follows and has a convoluted nucleus.
The monocyte arises with an indented nucleus and is released from the bone marrow to become a macrophage in the tissues.
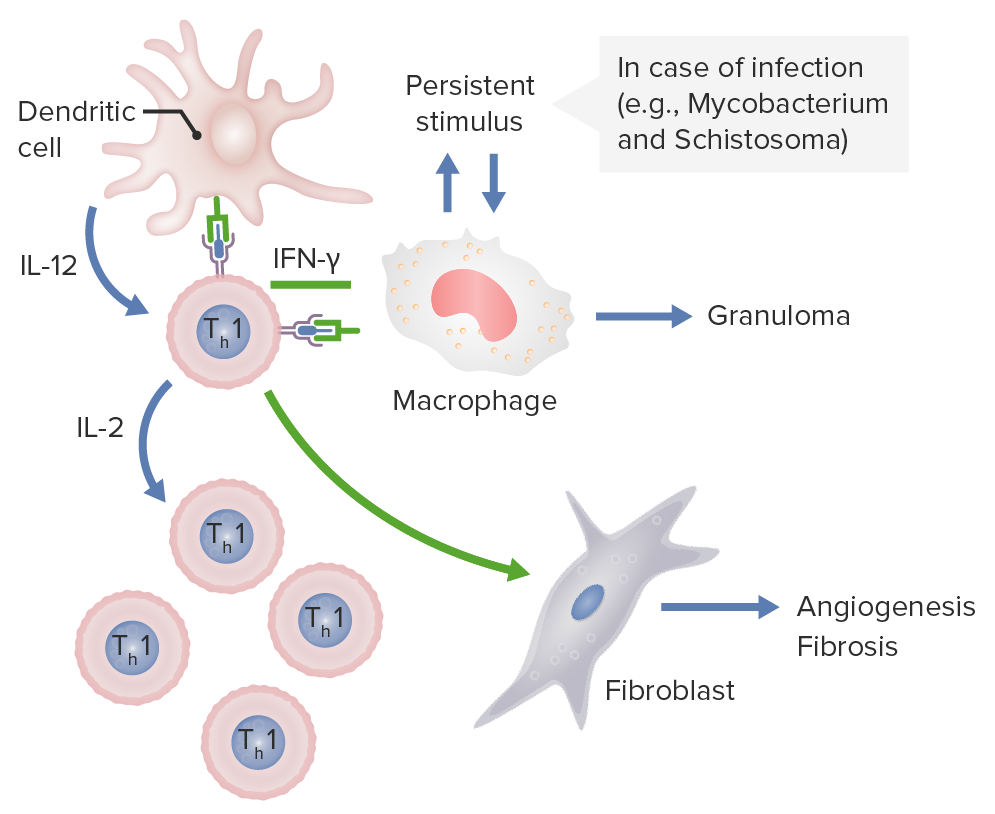
Dendritic cells release IL-12, which activates CD4 Th1 cells. These Th1 cells produce IL-2, stimulating production of more Th1 T-cell subsets. Th1 cells also release IFN-γ, which activates macrophages and activates fibroblasts to cause angiogenesis and fibrosis. If these macrophages are persistently stimulated by pathogens, such as Mycobacterium and Schistosoma, granulomas are formed.
Image by Lecturio.It is important to note that follicular dendritic cells Follicular dendritic cells Non-hematopoietic cells, with extensive dendritic processes, found in the primary and secondary follicles of lymphoid tissue (the B cell zones). They are different from conventional dendritic cells associated with T-cells. They are derived from mesenchymal stem cells and are negative for class II mhc antigen and do not process or present antigen like the conventional dendritic cells do. Instead, follicular dendritic cells have fc receptors and C3b receptors that hold antigen in the form of antigen-antibody complexes on their surfaces for long periods for recognition by B-cells. MALT Lymphoma are completely unrelated to dendritic cells Dendritic cells Specialized cells of the hematopoietic system that have branch-like extensions. They are found throughout the lymphatic system, and in non-lymphoid tissues such as skin and the epithelia of the intestinal, respiratory, and reproductive tracts. They trap and process antigens, and present them to T-cells, thereby stimulating cell-mediated immunity. They are different from the non-hematopoietic follicular dendritic cells, which have a similar morphology and immune system function, but with respect to humoral immunity (antibody production). Skin: Structure and Functions in lineage and function.
Follicular dendritic cells Follicular dendritic cells Non-hematopoietic cells, with extensive dendritic processes, found in the primary and secondary follicles of lymphoid tissue (the B cell zones). They are different from conventional dendritic cells associated with T-cells. They are derived from mesenchymal stem cells and are negative for class II mhc antigen and do not process or present antigen like the conventional dendritic cells do. Instead, follicular dendritic cells have fc receptors and C3b receptors that hold antigen in the form of antigen-antibody complexes on their surfaces for long periods for recognition by B-cells. MALT Lymphoma:
| Dendritic cells Dendritic cells Specialized cells of the hematopoietic system that have branch-like extensions. They are found throughout the lymphatic system, and in non-lymphoid tissues such as skin and the epithelia of the intestinal, respiratory, and reproductive tracts. They trap and process antigens, and present them to T-cells, thereby stimulating cell-mediated immunity. They are different from the non-hematopoietic follicular dendritic cells, which have a similar morphology and immune system function, but with respect to humoral immunity (antibody production). Skin: Structure and Functions | Follicular dendritic cells Follicular dendritic cells Non-hematopoietic cells, with extensive dendritic processes, found in the primary and secondary follicles of lymphoid tissue (the B cell zones). They are different from conventional dendritic cells associated with T-cells. They are derived from mesenchymal stem cells and are negative for class II mhc antigen and do not process or present antigen like the conventional dendritic cells do. Instead, follicular dendritic cells have fc receptors and C3b receptors that hold antigen in the form of antigen-antibody complexes on their surfaces for long periods for recognition by B-cells. MALT Lymphoma | |
|---|---|---|
| Origin | Derived from hematopoietic stem cells Hematopoietic stem cells Progenitor cells from which all blood cells derived. They are found primarily in the bone marrow and also in small numbers in the peripheral blood. Bone Marrow: Composition and Hematopoiesis | Derived from mesenchymal stem cells |
| Sites | Present throughout the body | Present only in germinal centers of secondary lymphoid tissues |
| MHC class and costimulatory molecules | Possess MHC II and costimulatory (e.g., B7) molecules | Lack MHC II and costimulatory molecules |
| Functions |
|
|
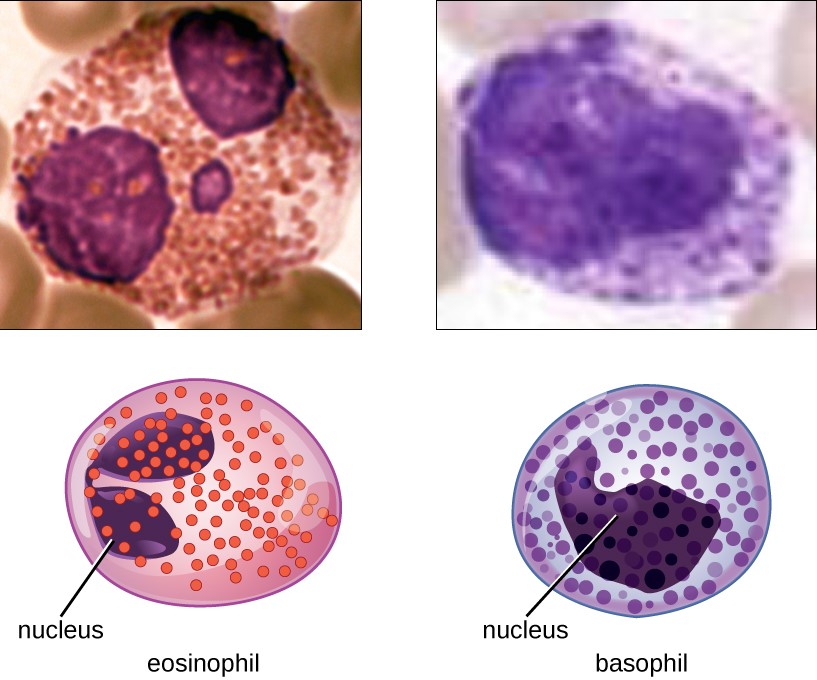
Eosinophil and basophil
Both are granulocytes, with eosinophils possessing a bilobed nucleus and dark pink granules and basophils having a bilobed or trilobed nucleus, and dark blue granules.
Antigen-presenting cells Antigen-presenting cells A heterogeneous group of immunocompetent cells that mediate the cellular immune response by processing and presenting antigens to the T-cells. Traditional antigen-presenting cells include macrophages; dendritic cells; langerhans cells; and B-lymphocytes. Follicular dendritic cells are not traditional antigen-presenting cells, but because they hold antigen on their cell surface in the form of immune complexes for b-cell recognition they are considered so by some authors. Adaptive Immune Response (such as dendritic cells Dendritic cells Specialized cells of the hematopoietic system that have branch-like extensions. They are found throughout the lymphatic system, and in non-lymphoid tissues such as skin and the epithelia of the intestinal, respiratory, and reproductive tracts. They trap and process antigens, and present them to T-cells, thereby stimulating cell-mediated immunity. They are different from the non-hematopoietic follicular dendritic cells, which have a similar morphology and immune system function, but with respect to humoral immunity (antibody production). Skin: Structure and Functions and macrophages) detect, process, and present the antigens to T cells T cells Lymphocytes responsible for cell-mediated immunity. Two types have been identified – cytotoxic (t-lymphocytes, cytotoxic) and helper T-lymphocytes (t-lymphocytes, helper-inducer). They are formed when lymphocytes circulate through the thymus gland and differentiate to thymocytes. When exposed to an antigen, they divide rapidly and produce large numbers of new T cells sensitized to that antigen. T cells: Types and Functions, allowing adaptive immunity to recognize and mount a response every time the pathogen is encountered (immunologic memory Memory Complex mental function having four distinct phases: (1) memorizing or learning, (2) retention, (3) recall, and (4) recognition. Clinically, it is usually subdivided into immediate, recent, and remote memory. Psychiatric Assessment).
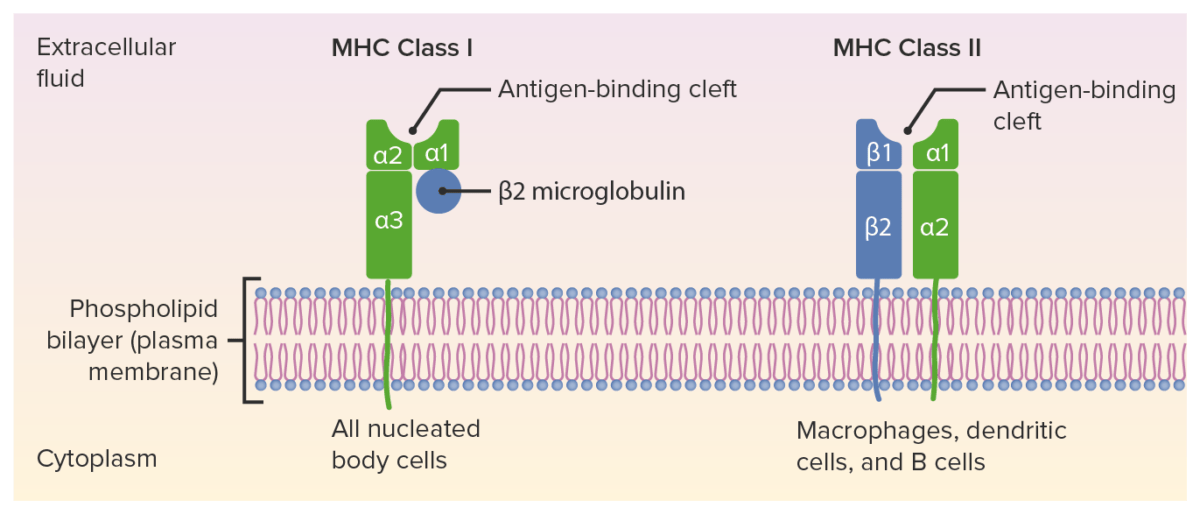
Structures of MHC I and MHC II:
MHC I has 1 short and 1 long chain (ɑ chain with 3 domains: ɑ1, ɑ2, and ɑ3), associated with the β₂-microglobulin. MHC II has 2 ɑ and 2 β chains. The peptide antigen goes to the antigen-binding cleft.
MHC: major histocompatibility complex
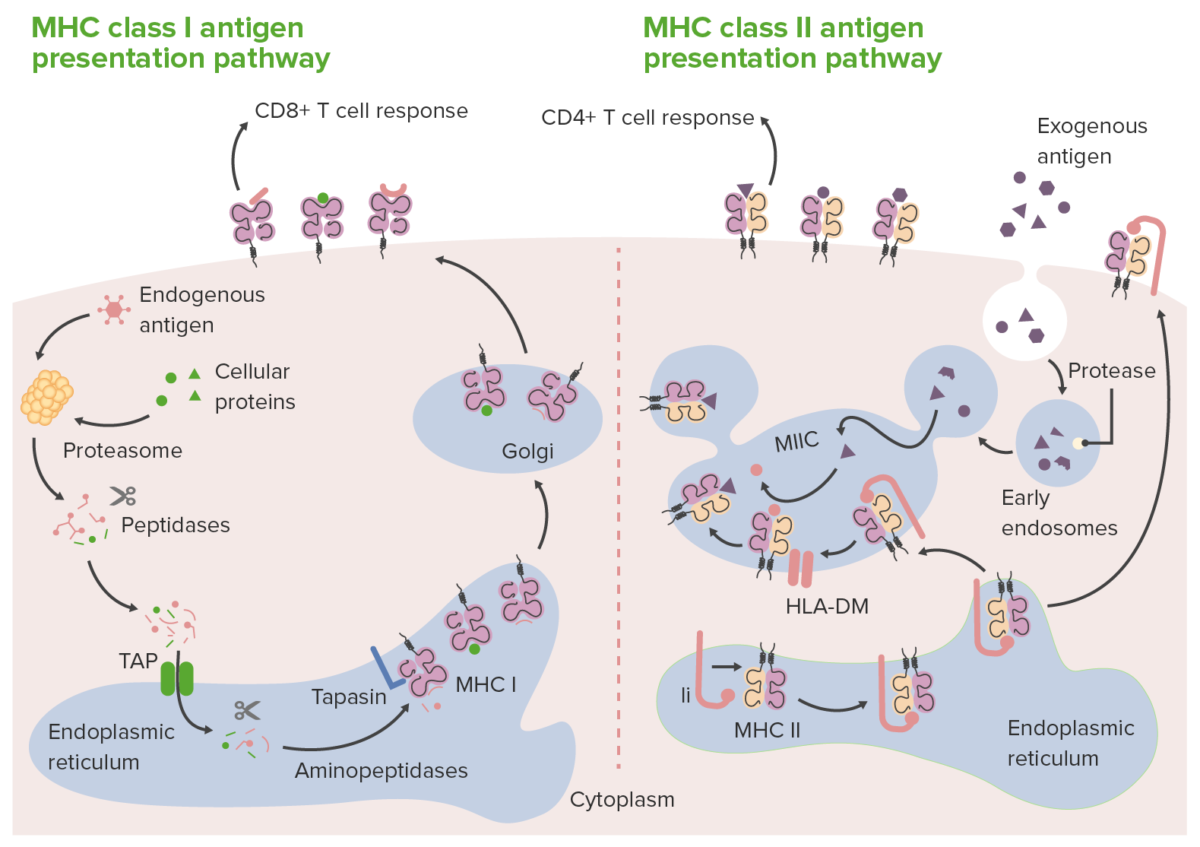
Routes of antigen presentation by MHC class I and II molecules:
In class I antigen presentation (left), proteasomes degrade endogenous antigens or proteins (within the cell) into peptides. Peptide fragments are transported (via transporter associated with antigen processing (TAP)) to the ER, where they are further trimmed by aminopeptidases and loaded onto the MHC class I molecule. MHC class I–loaded complexes go to the Golgi apparatus for posttranslational modification. Then the complexes are transported to the cell surface, where they are presented to CD8+ T cells.
IN class II antigen presentation (right), extracellular/exogenous antigens are taken up within phagosomes by antigen-presenting cells. The phagosomes then fuse with proteolytic enzyme-filled lysosomes. This results in the breakdown of phagocytosed proteins into small peptides. Meanwhile, in the endoplasmic reticulum (ER), new MHC class II molecules are synthesized. These molecules have the invariant chain (pink structure in the right image, marked Ii), which binds the antigen-binding cleft. With the cleft occluded (by the invariant chain), ER-resident peptides cannot bind. The invariant chain directs the MHC II complex to the acidified endosome (where antigen peptides are) as it exits from the ER. When MHC II complexes are delivered to the endosome, the invariant chain is released, allowing loading of antigen peptides (chaperoned by a protein, HLA-DM) onto the MHC class II molecules. Once loaded, the formed antigen peptide-MHC class II complexes are brought to the cell surface, ready to present the antigen to CD4+ T cells.
Ii: MHC class II–associated invariant chain
MIIC: MHC class II compartment
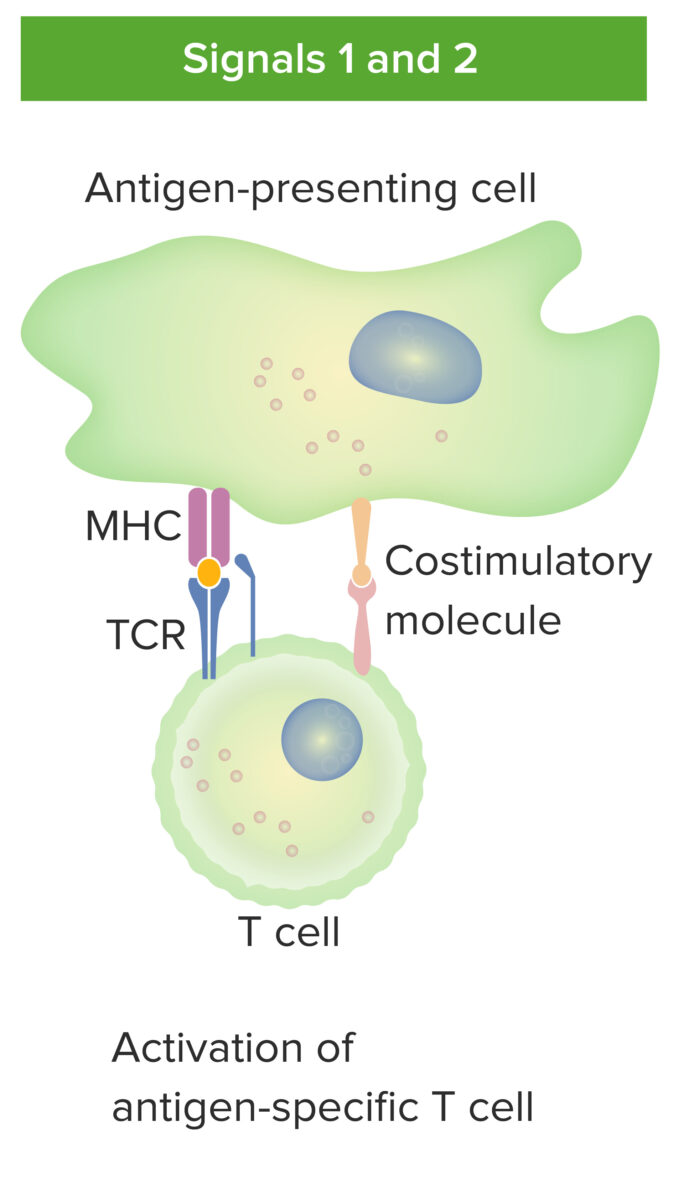
Antigen-presenting–cell and T-cell interaction:
Antigen-presenting cell interacts with T cell via signal 1 (T-cell receptor binding the cognate antigen presented by MHC molecule in the APC) and signal 2 (costimulatory molecule interaction between APC and T cell). With the proper antigen presentation, the mature T cell becomes activated.
| MHC I | MHC II | |
|---|---|---|
| Loci | HLA-A HLA-A Polymorphic class I human histocompatibility (HLA) surface antigens present on almost all nucleated cells. At least 20 antigens have been identified which are encoded by the a locus of multiple alleles on chromosome 6. They serve as targets for t-cell cytolytic responses and are involved with acceptance or rejection of tissue/organ grafts. Organ Transplantation, HLA-B HLA-B Class I human histocompatibility (HLA) surface antigens encoded by more than 30 detectable alleles on locus B of the HLA complex, the most polymorphic of all the HLA specificities. Several of these antigens (e.g., hla-b27, -b7, -b8) are strongly associated with predisposition to rheumatoid and other autoimmune disorders. Like other class I HLA determinants, they are involved in the cellular immune reactivity of cytolytic T lymphocytes. Organ Transplantation, HLA-C HLA-C Class I human histocompatibility (HLA) antigens encoded by a small cluster of structural genes at the c locus on chromosome 6. They have significantly lower immunogenicity than the hla-a and -b determinants and are therefore of minor importance in donor/recipient crossmatching. Their primary role is their high-risk association with certain disease manifestations (e.g., spondyloarthritis, psoriasis, multiple myeloma). Organ Transplantation | HLA-DP, HLA-DQ, HLA-DR |
| Binding | CD8 T cell | CD4 T cell |
| Distribution | All nucleated cells (none on RBCs RBCs Erythrocytes, or red blood cells (RBCs), are the most abundant cells in the blood. While erythrocytes in the fetus are initially produced in the yolk sac then the liver, the bone marrow eventually becomes the main site of production. Erythrocytes: Histology) | Antigen-presenting cells Antigen-presenting cells A heterogeneous group of immunocompetent cells that mediate the cellular immune response by processing and presenting antigens to the T-cells. Traditional antigen-presenting cells include macrophages; dendritic cells; langerhans cells; and B-lymphocytes. Follicular dendritic cells are not traditional antigen-presenting cells, but because they hold antigen on their cell surface in the form of immune complexes for b-cell recognition they are considered so by some authors. Adaptive Immune Response |
| Role | Present endogenous antigens to CD8+ T cells T cells Lymphocytes responsible for cell-mediated immunity. Two types have been identified – cytotoxic (t-lymphocytes, cytotoxic) and helper T-lymphocytes (t-lymphocytes, helper-inducer). They are formed when lymphocytes circulate through the thymus gland and differentiate to thymocytes. When exposed to an antigen, they divide rapidly and produce large numbers of new T cells sensitized to that antigen. T cells: Types and Functions (cytolytic) | Present exogenous antigens to CD4+ T cells T cells Lymphocytes responsible for cell-mediated immunity. Two types have been identified – cytotoxic (t-lymphocytes, cytotoxic) and helper T-lymphocytes (t-lymphocytes, helper-inducer). They are formed when lymphocytes circulate through the thymus gland and differentiate to thymocytes. When exposed to an antigen, they divide rapidly and produce large numbers of new T cells sensitized to that antigen. T cells: Types and Functions |
| Structure |
|
2 chains of equal length (2 ɑ, 2 β) |
| Associated protein | β₂-microglobulin | Invariant chain |
| Antigen Antigen Substances that are recognized by the immune system and induce an immune reaction. Vaccination loading | Loading of antigen Antigen Substances that are recognized by the immune system and induce an immune reaction. Vaccination peptide onto MHC I in ER (delivered via TAP) | Loading of antigen Antigen Substances that are recognized by the immune system and induce an immune reaction. Vaccination peptide onto MHC II in the acidified phagolysosome Phagolysosome Chédiak-Higashi Syndrome after release of invariant chain |
The HLA region encodes several molecules that perform key functions in the immune system Immune system The body’s defense mechanism against foreign organisms or substances and deviant native cells. It includes the humoral immune response and the cell-mediated response and consists of a complex of interrelated cellular, molecular, and genetic components. Primary Lymphatic Organs. There is a robust association between the HLA region and several diseases.
| HLA subtype | Condition(s) | Mnemonics |
|---|---|---|
| A3 | Hemochromatosis Hemochromatosis A disorder of iron metabolism characterized by a triad of hemosiderosis; liver cirrhosis; and diabetes mellitus. It is caused by massive iron deposits in parenchymal cells that may develop after a prolonged increase of iron absorption. Hereditary Hemochromatosis | HA3mochromatosis Fe3 ( iron Iron A metallic element with atomic symbol fe, atomic number 26, and atomic weight 55. 85. It is an essential constituent of hemoglobins; cytochromes; and iron-binding proteins. It plays a role in cellular redox reactions and in the transport of oxygen. Trace Elements = hemochromatosis Hemochromatosis A disorder of iron metabolism characterized by a triad of hemosiderosis; liver cirrhosis; and diabetes mellitus. It is caused by massive iron deposits in parenchymal cells that may develop after a prolonged increase of iron absorption. Hereditary Hemochromatosis), A3 |
| B8 |
|
Don’t B l8, Dr. Addison, or you’ll send my patient to the grave! |
| B27 |
|
PAIR |
| C | Psoriasis Psoriasis Psoriasis is a common T-cell-mediated inflammatory skin condition. The etiology is unknown, but is thought to be due to genetic inheritance and environmental triggers. There are 4 major subtypes, with the most common form being chronic plaque psoriasis. Psoriasis | “C-riasis” |
| DQ2/DQ8 | Celiac disease Celiac disease Celiac disease (also known as celiac sprue or gluten enteropathy) is an autoimmune reaction to gliadin, which is a component of gluten. Celiac disease is closely associated with HLA-DQ2 and HLA-DQ8. The immune response is localized to the proximal small intestine and causes the characteristic histologic findings of villous atrophy, crypt hyperplasia, and intraepithelial lymphocytosis. Celiac Disease | I 8 2 much gluten Gluten Prolamins in the endosperm of seeds from the triticeae tribe which includes species of wheat; barley; and rye. Celiac Disease at Dairy Queen (DQ2/8; gluten Gluten Prolamins in the endosperm of seeds from the triticeae tribe which includes species of wheat; barley; and rye. Celiac Disease = celiac disease Celiac disease Celiac disease (also known as celiac sprue or gluten enteropathy) is an autoimmune reaction to gliadin, which is a component of gluten. Celiac disease is closely associated with HLA-DQ2 and HLA-DQ8. The immune response is localized to the proximal small intestine and causes the characteristic histologic findings of villous atrophy, crypt hyperplasia, and intraepithelial lymphocytosis. Celiac Disease) |
| DR2 |
|
|
| DR3 |
|
|
| DR4 |
|
|
| DR5 | Hashimoto’s thyroiditis Thyroiditis Thyroiditis is a catchall term used to describe a variety of conditions that have inflammation of the thyroid gland in common. It includes pathologies that cause an acute illness with severe thyroid pain (e.g., subacute thyroiditis and infectious thyroiditis) as well as conditions in which there is no clinically evident inflammation and the manifestations primarily reflect thyroid dysfunction or a goiter (e.g., painless thyroiditis and fibrous Riedel’s thyroiditis). Thyroiditis | Dr. Hashimoto is odd (odd numbers 3, 5) |
| DR7 | Steroid-responsive nephrotic syndrome Nephrotic syndrome Nephrotic syndrome is characterized by severe proteinuria, hypoalbuminemia, and peripheral edema. In contrast, the nephritic syndromes present with hematuria, variable loss of renal function, and hypertension, although there is sometimes overlap of > 1 glomerular disease in the same individual. Nephrotic Syndrome | 7, “pee in heaven” (nephrotic) |