Tumor Tumor Inflammation necrosis Necrosis The death of cells in an organ or tissue due to disease, injury or failure of the blood supply. Ischemic Cell Damage factor (TNF) is a major cytokine, released primarily by macrophages Macrophages The relatively long-lived phagocytic cell of mammalian tissues that are derived from blood monocytes. Main types are peritoneal macrophages; alveolar macrophages; histiocytes; kupffer cells of the liver; and osteoclasts. They may further differentiate within chronic inflammatory lesions to epithelioid cells or may fuse to form foreign body giant cells or langhans giant cells. Innate Immunity: Phagocytes and Antigen Presentation in response to stimuli. The presence of microbial products and dead cells and injury are among the stimulating factors. This protein belongs to the TNF superfamily, a group of ligands and receptors Receptors Receptors are proteins located either on the surface of or within a cell that can bind to signaling molecules known as ligands (e.g., hormones) and cause some type of response within the cell. Receptors performing functions in inflammatory response, morphogenesis, and cell proliferation. Tumor Tumor Inflammation necrosis Necrosis The death of cells in an organ or tissue due to disease, injury or failure of the blood supply. Ischemic Cell Damage factor interacts with 2 receptors Receptors Receptors are proteins located either on the surface of or within a cell that can bind to signaling molecules known as ligands (e.g., hormones) and cause some type of response within the cell. Receptors, which initiate signal transduction Transduction The transfer of bacterial DNA by phages from an infected bacterium to another bacterium. This also refers to the transfer of genes into eukaryotic cells by viruses. This naturally occurring process is routinely employed as a gene transfer technique. Bacteriology pathways leading to different cellular responses ( inflammation Inflammation Inflammation is a complex set of responses to infection and injury involving leukocytes as the principal cellular mediators in the body's defense against pathogenic organisms. Inflammation is also seen as a response to tissue injury in the process of wound healing. The 5 cardinal signs of inflammation are pain, heat, redness, swelling, and loss of function. Inflammation, cell survival, or apoptosis Apoptosis A regulated cell death mechanism characterized by distinctive morphologic changes in the nucleus and cytoplasm, including the endonucleolytic cleavage of genomic DNA, at regularly spaced, internucleosomal sites, I.e., DNA fragmentation. It is genetically-programmed and serves as a balance to mitosis in regulating the size of animal tissues and in mediating pathologic processes associated with tumor growth. Ischemic Cell Damage). Inappropriate or unrestrained activation of TNF signaling produces chronic inflammation Chronic Inflammation Inflammation, as seen in autoimmune conditions (e.g., rheumatoid arthritis Arthritis Acute or chronic inflammation of joints. Osteoarthritis, psoriasis Psoriasis Psoriasis is a common T-cell-mediated inflammatory skin condition. The etiology is unknown, but is thought to be due to genetic inheritance and environmental triggers. There are 4 major subtypes, with the most common form being chronic plaque psoriasis. Psoriasis). The mechanism of TNF inhibition has been used in treating these inflammatory diseases.
Last updated: May 17, 2024
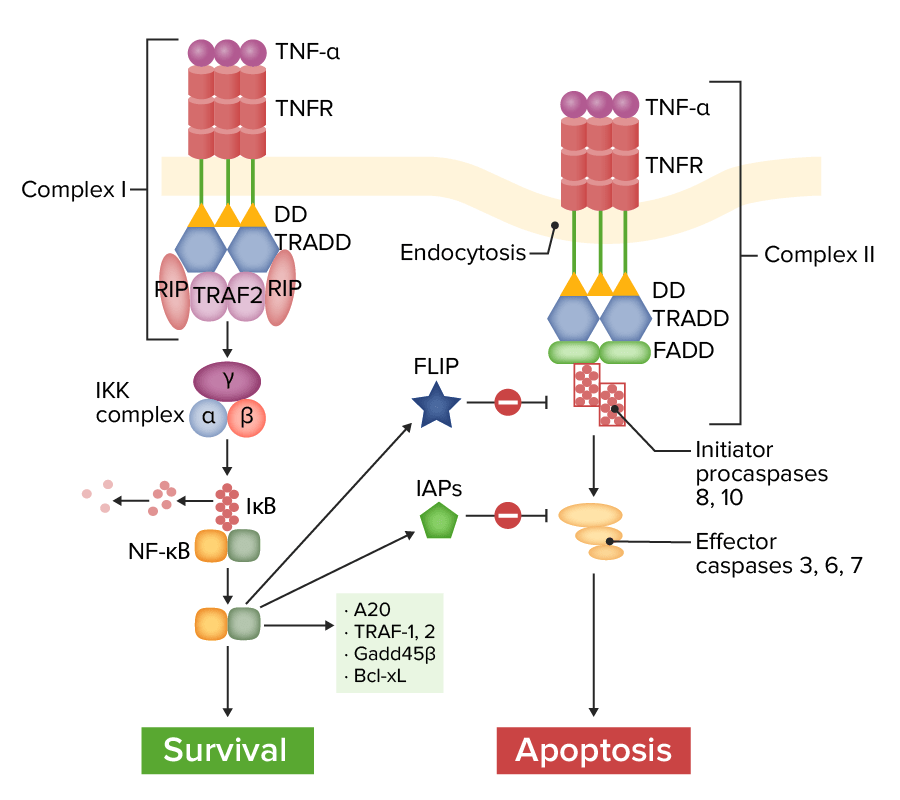
Tumor necrosis factor receptor 1 (TNFR1) pathway of signaling:
Complex I (on the left): With ligation of TNF-α, TNFR recruits various adapter molecules, resulting in the activation of nuclear factor kappa-B (NF-κB), which induces several anti-apoptotic genes and survival signals. FLICE inhibitory protein (FLIP) and inhibitor of apoptosis proteins (IAPs) modulate and inhibit the apoptosis pathway.
Complex II (on the right): Without certain adapter proteins (TRAF-2, RIP), TNFR leads to recruitment of Fas-associated protein with death domain (FADD). Caspase-8 is activated and released into the cytoplasm, activating effector caspases to induce apoptosis.
IκB: inhibitor of NF-κB
TRAF-2: TNF-receptor–associated factor.
RIP: receptor interacting protein
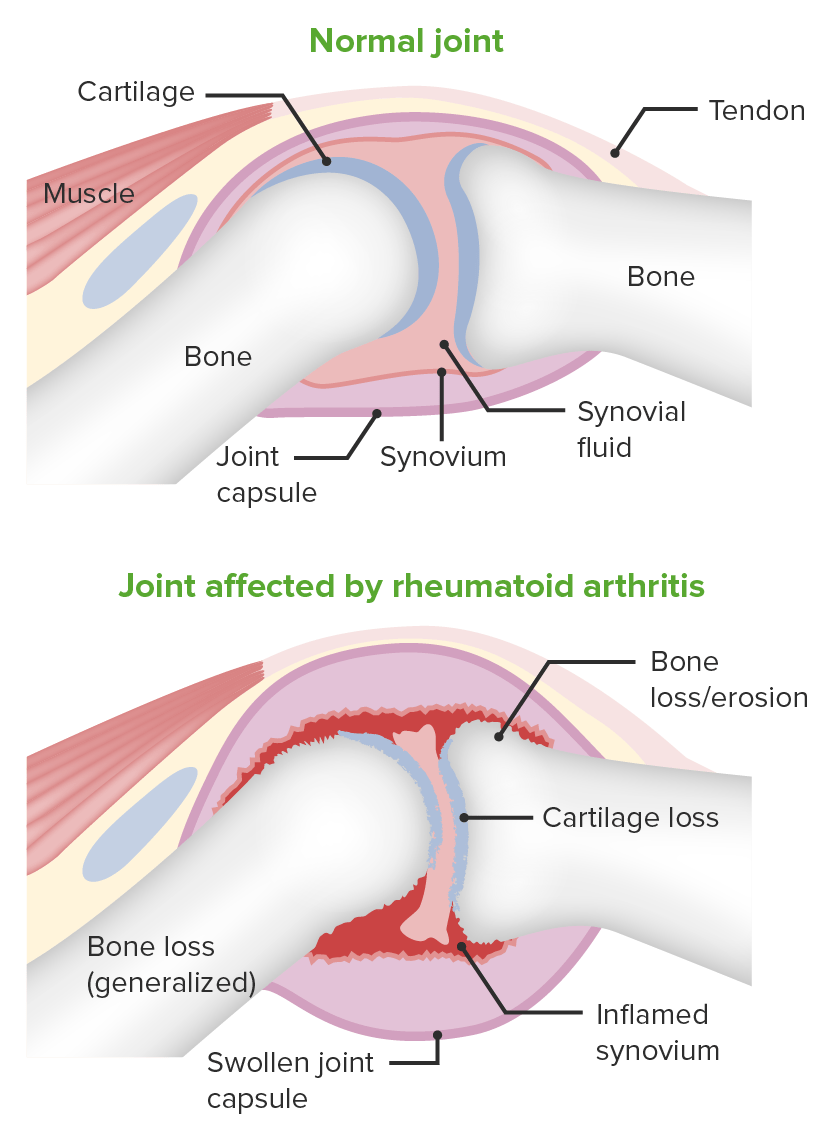
Image demonstrating the pathology of rheumatoid arthritis:
Changes in the joint space are shown.
Tumor Tumor Inflammation necrosis Necrosis The death of cells in an organ or tissue due to disease, injury or failure of the blood supply. Ischemic Cell Damage factor has multiple biologic effects, and in certain conditions (e.g., rheumatoid arthritis Arthritis Acute or chronic inflammation of joints. Osteoarthritis), elevated levels of TNF have been noted. The mechanism of TNF inhibition has been used in treating inflammatory diseases.
| Anti-TNF therapy | Mechanism of action | Indications |
|---|---|---|
| Infliximab Infliximab A chimeric monoclonal antibody to tnf-alpha that is used in the treatment of rheumatoid arthritis; ankylosing spondylitis; psoriatic arthritis and Crohn’s disease. Disease-Modifying Antirheumatic Drugs (DMARDs) | Recombinant chimeric antibody (having a murine variable region Variable region That region of the immunoglobulin molecule that varies in its amino acid sequence and composition, and comprises the binding site for a specific antigen. It is located at the n-terminus of the fab fragment of the immunoglobulin. It includes hypervariable regions (complementarity determining regions) and framework regions. Immunoglobulins: Types and Functions and a human IgG1 constant region Constant region The domains of the immunoglobulin molecules that are invariable in their amino acid sequence within any class or subclass of immunoglobulin. They confer biological as well as structural functions to immunoglobulins. One each on both the light chains and the heavy chains comprises the c-terminus half of the immunoglobulin fab fragment and two or three of them make up the rest of the heavy chains (all of the immunoglobulin Fc fragment). Immunoglobulins: Types and Functions) that binds to TNF, preventing receptor Receptor Receptors are proteins located either on the surface of or within a cell that can bind to signaling molecules known as ligands (e.g., hormones) and cause some type of response within the cell. Receptors interaction |
|
| Etanercept Etanercept A recombinant version of soluble human tnf receptor fused to an IgG Fc fragment that binds specifically to tumor necrosis factor and inhibits its binding with endogenous tnf receptors. It prevents the inflammatory effect of tnf and is used to treat rheumatoid arthritis; psoriatic arthritis and ankylosing spondylitis. Immunosuppressants | Fusion protein Fusion protein Proteins that catalyze membrane fusion. Measles Virus that binds and neutralizes TNF and lymphotoxin |
|
| Adalimumab Adalimumab A humanized monoclonal antibody that binds specifically to tnf-alpha and blocks its interaction with endogenous tnf receptors to modulate inflammation. It is used in the treatment of rheumatoid arthritis; psoriatic arthritis; Crohn’s disease and ulcerative colitis. Disease-Modifying Antirheumatic Drugs (DMARDs) | Fully humanized IgG1 monoclonal antibody that blocks TNF binding to receptors Receptors Receptors are proteins located either on the surface of or within a cell that can bind to signaling molecules known as ligands (e.g., hormones) and cause some type of response within the cell. Receptors |
|
| Golimumab Golimumab Disease-Modifying Antirheumatic Drugs (DMARDs) | Fully humanized IgG1 monoclonal antibody with high affinity and specificity for TNF |
|
| Certolizumab Certolizumab Disease-Modifying Antirheumatic Drugs (DMARDs) | Pegylated humanized monoclonal antibody specific to TNF |
|