Azoles are a widely used class of antifungal medications inhibiting the production of ergosterol, a critical component in the fungal cell membrane Cell Membrane A cell membrane (also known as the plasma membrane or plasmalemma) is a biological membrane that separates the cell contents from the outside environment. A cell membrane is composed of a phospholipid bilayer and proteins that function to protect cellular DNA and mediate the exchange of ions and molecules. The Cell: Cell Membrane. The 2 primary subclasses of azoles are the imidazoles, older agents typically only used for topical applications, and the triazoles, newer agents with a wide spectrum of uses. Various members of the class are indicated in the treatment and prophylaxis Prophylaxis Cephalosporins of candidiasis Candidiasis Candida is a genus of dimorphic, opportunistic fungi. Candida albicans is part of the normal human flora and is the most common cause of candidiasis. The clinical presentation varies and can include localized mucocutaneous infections (e.g., oropharyngeal, esophageal, intertriginous, and vulvovaginal candidiasis) and invasive disease (e.g., candidemia, intraabdominal abscess, pericarditis, and meningitis). Candida/Candidiasis, aspergillosis Aspergillosis Aspergillosis is an opportunistic fungal infection caused by Aspergillus species, which are common spore-forming molds found in our environment. As Aspergillus species are opportunistic, they cause disease primarily in patients who are immunocompromised. The organs that are most commonly involved are the lungs and sinuses. Aspergillus/Aspergillosis, cryptococcus Cryptococcus Cryptococcosis is an opportunistic, fungal infection caused by the Cryptococcus species. The principal pathogens in humans are C. neoformans (primary) and C. gattii. Cryptococcus neoformans is typically found in pigeon droppings and acquired by inhaling dust from contaminated soil. The majority of affected patients are immunocompromised. Cryptococcus/Cryptococcosis meningitis Meningitis Meningitis is inflammation of the meninges, the protective membranes of the brain, and spinal cord. The causes of meningitis are varied, with the most common being bacterial or viral infection. The classic presentation of meningitis is a triad of fever, altered mental status, and nuchal rigidity. Meningitis, dimorphic fungal infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease (e.g., blastomycosis Blastomycosis Blastomycosis is an infection caused by inhalation of the spores of the fungus, Blastomyces. Blastomyces species thrive in moist soil and decaying material and are common in the Ohio and Mississippi River valleys and the Great Lakes regions of the United States and Canada. Although most patients are asymptomatic, some can develop pneumonia. Blastomyces/Blastomycosis), and mucormycosis Mucormycosis Mucormycosis is an angioinvasive fungal infection caused by multiple fungi within the order, Mucorales. The fungi are ubiquitous in the environment, but mucormycosis is very rare and almost always occurs in patients who are immunocompromised. Inhalation of fungal spores can cause rhinocerebral or pulmonary mucormycosis, direct inoculation can cause cutaneous mucormycosis, and ingestion can cause gastrointestinal mucormycosis. Mucorales/Mucormycosis. Significant adverse effects are possible and include hepatotoxicity Hepatotoxicity Acetaminophen, GI distress, cardiac issues, and neurotoxicity. Azoles interact with the CYP450 system causing significant drug-to-drug interactions with many other medications, potentially limiting the usefulness in medically complex individuals.
Last updated: Mar 27, 2025
Azoles are a group of widely used antifungal medications, which can be classified into 2 subgroups:

Fluconazole
Image: “Fluconazole skeletal formula” by Vaccinationist. License: Public Domain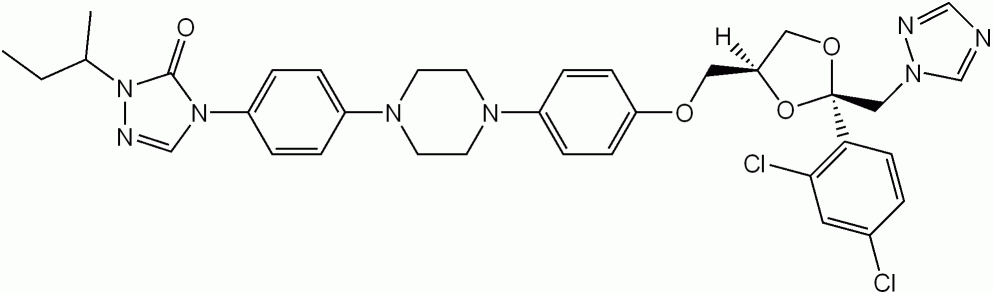
Itraconazole
Image: “Itraconazole” by Shaddack. License: Public Domain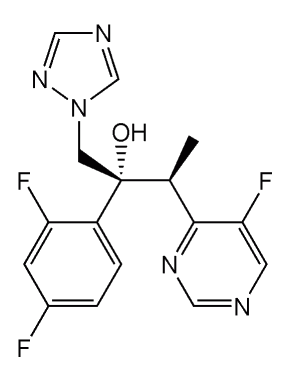
Voriconazole
Image: “Voriconazole” by Shaddack. License: Public Domain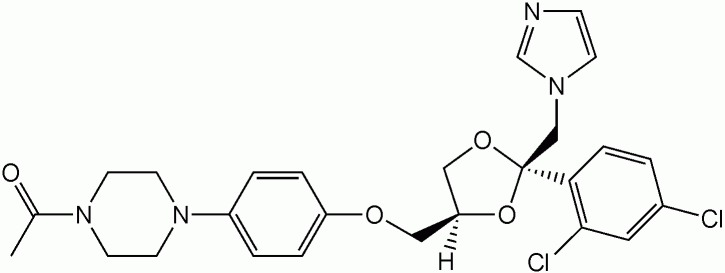
Ketoconazole
Image: “Ketoconazole” by Shaddack. License: Public DomainAzoles cause deterioration in the fungal cell membrane Cell Membrane A cell membrane (also known as the plasma membrane or plasmalemma) is a biological membrane that separates the cell contents from the outside environment. A cell membrane is composed of a phospholipid bilayer and proteins that function to protect cellular DNA and mediate the exchange of ions and molecules. The Cell: Cell Membrane by inhibiting ergosterol production.
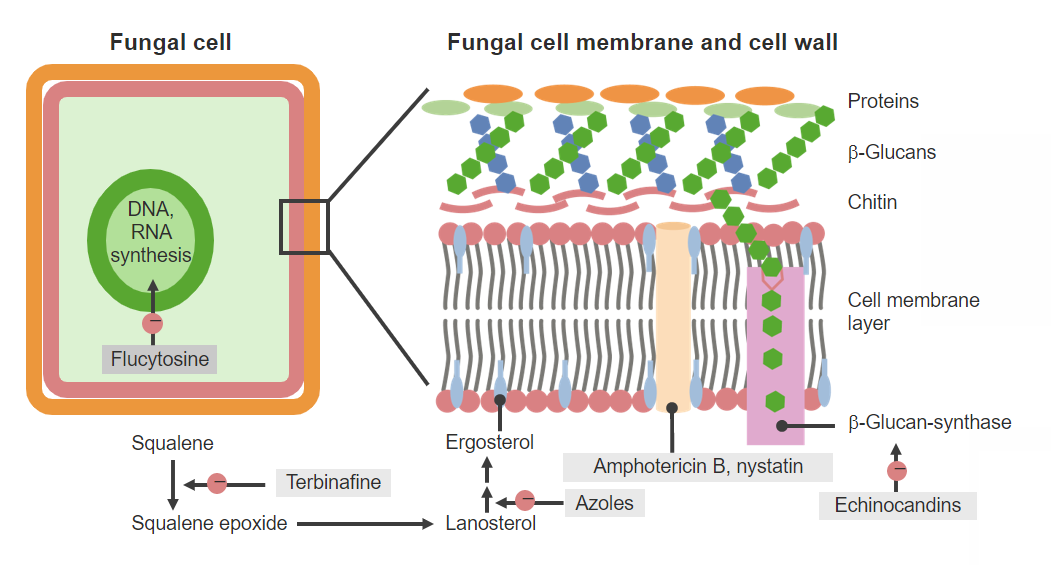
Antifungal agents and mechanisms of action
Image by Lecturio. License: CC BY-NC-SA 4.0Different azoles have variable Variable Variables represent information about something that can change. The design of the measurement scales, or of the methods for obtaining information, will determine the data gathered and the characteristics of that data. As a result, a variable can be qualitative or quantitative, and may be further classified into subgroups. Types of Variables (and sometimes complex) pharmacokinetics Pharmacokinetics Pharmacokinetics is the science that analyzes how the human body interacts with a drug. Pharmacokinetics examines how the drug is absorbed, distributed, metabolized, and excreted by the body. Pharmacokinetics and Pharmacodynamics.
| Drugs | Protein binding | Metabolism and clearance | Half-life Half-Life The time it takes for a substance (drug, radioactive nuclide, or other) to lose half of its pharmacologic, physiologic, or radiologic activity. Pharmacokinetics and Pharmacodynamics | Enzyme inhibition Enzyme inhibition Enzyme inhibitors bind to enzymes and decrease their activity. Enzyme activators bind to enzymes and increase their activity. Molecules that decrease the catalytic activity of enzymes can come in various forms, including reversible or irreversible inhibition. Enzyme Inhibition |
|---|---|---|---|---|
| Imidazoles | ||||
| Ketoconazole | 99% | Partial hepatic metabolism by CYP3A4 CYP3A4 Class 3 Antiarrhythmic Drugs (Potassium Channel Blockers) | Biphasic:
|
Significantly more inhibition of mammalian CYP450 than the triazoles |
| Triazoles | ||||
| Fluconazole | Approximately 10% |
|
25 hours |
|
| Itraconazole | 99% |
|
24–48 hours |
|
| Voriconazole | Approximately 60% | Extensive hepatic metabolism by CYP2C19 (major), 2C9, and 3A4 (minor) | 6 hours |
|
| Posaconazole | 98% |
|
25–35 hours | CYP3A4 CYP3A4 Class 3 Antiarrhythmic Drugs (Potassium Channel Blockers) (moderate) |
| Isavuconazole | 99% | Hepatic by CYP3A4 CYP3A4 Class 3 Antiarrhythmic Drugs (Potassium Channel Blockers) and glucuronidation | 130 hours |
|
Azoles have a wide variety of uses and unique indications for each medication.
In addition to the specific indications listed below, most of the triazoles can also be used as prophylaxis Prophylaxis Cephalosporins against fungal infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease in immunosuppressed individuals.
The common mechanisms contributing to antifungal resistance Resistance Physiologically, the opposition to flow of air caused by the forces of friction. As a part of pulmonary function testing, it is the ratio of driving pressure to the rate of air flow. Ventilation: Mechanics of Breathing include:
| Drug class (examples) | Mechanism of action | Clinical relevance |
|---|---|---|
| Azoles (Fluconazole, Voriconazole) | Inhibits the production of ergosterol (a critical component of the fungal cell membrane Cell Membrane A cell membrane (also known as the plasma membrane or plasmalemma) is a biological membrane that separates the cell contents from the outside environment. A cell membrane is composed of a phospholipid bilayer and proteins that function to protect cellular DNA and mediate the exchange of ions and molecules. The Cell: Cell Membrane) by blocking the lanosterol Lanosterol A triterpene that derives from the chair-boat-chair-boat folding of 2, 3-oxidosqualene. It is metabolized to cholesterol and cucurbitacins. Cholesterol Metabolism 14-α-demethylase enzyme |
|
| Polyenes Polyenes Polyenes are a class of fungicidal agents that consist of 2 primary drugs in current use, namely, nystatin and amphotericin B. Both these drugs exert their effects by binding to ergosterol (a critical component of fungal cell membranes) and creating pores in the membrane, leading to the leakage of intracellular components and ultimately cell lysis. Polyenes ( Amphotericin B Amphotericin B Macrolide antifungal antibiotic produced by streptomyces nodosus obtained from soil of the orinoco river region of venezuela. Polyenes, Nystatin Nystatin Macrolide antifungal antibiotic complex produced by streptomyces noursei, s. Aureus, and other streptomyces species. The biologically active components of the complex are nystatin a1, a2, and a3. Polyenes) | Binds to ergosterol in the fungal cell membrane Cell Membrane A cell membrane (also known as the plasma membrane or plasmalemma) is a biological membrane that separates the cell contents from the outside environment. A cell membrane is composed of a phospholipid bilayer and proteins that function to protect cellular DNA and mediate the exchange of ions and molecules. The Cell: Cell Membrane creating artificial pores in the membrane → results in leakage of cellular components and leads to cell lysis (death) |
Amphotericin B
Amphotericin B
Macrolide antifungal antibiotic produced by streptomyces nodosus obtained from soil of the orinoco river region of venezuela.
Polyenes:
Nystatin Nystatin Macrolide antifungal antibiotic complex produced by streptomyces noursei, s. Aureus, and other streptomyces species. The biologically active components of the complex are nystatin a1, a2, and a3. Polyenes:
|
| Echinocandins Echinocandins Echinocandins are a group of fungicidal agents that target the fungal cell wall. Echinocandins inhibit β-glucan synthase, which in turn inhibits the production of β-glucan, a key structural component of fungal cell walls. The 3 primary drugs in this class include caspofungin, micafungin, and anidulafungin. Echinocandins ( Caspofungin Caspofungin A cyclic lipopeptide echinocandin and beta-(1, 3)-d-glucan synthase inhibitor that is used to treat internal or systemic mycoses. Echinocandins, Micafungin Micafungin A cyclic lipo-hexapeptide echinocandin antifungal agent that is used for the treatment and prevention of candidiasis. Echinocandins, Anidulafungin Anidulafungin Echinocandin antifungal agent that is used in the treatment of candidemia and candidiasis. Echinocandins) | Inhibits β-glucan synthase (the enzyme synthesizing β-glucan and an important structural component of the fungal cell wall Fungal Cell Wall Echinocandins) → weakened cell wall Cell wall The outermost layer of a cell in most plants; bacteria; fungi; and algae. The cell wall is usually a rigid structure that lies external to the cell membrane, and provides a protective barrier against physical or chemical agents. Cell Types: Eukaryotic versus Prokaryotic → cell lysis |
|
| Griseofulvin Griseofulvin In addition to the 3 other major classes of antifungal agents (azoles, polyenes, and echinocandins), several other clinically important antifungal agents are used, including flucytosine, griseofulvin, and terbinafine. Griseofulvin acts within the stratum corneum of the skin and are used to treat dermatophyte infections of the skin, hair, and nails. Flucytosine, Griseofulvin, and Terbinafine |
|
|
| Terbinafine Terbinafine In addition to the 3 other major classes of antifungal agents (azoles, polyenes, and echinocandins), several other clinically important antifungal agents are used, including flucytosine, griseofulvin, and terbinafine. Terbinafine acts within the stratum corneum of the skin and are used to treat dermatophyte infections of the skin, hair, and nails. Flucytosine, Griseofulvin, and Terbinafine | Inhibits the squalene epoxidase Squalene Epoxidase Flucytosine, Griseofulvin, and Terbinafine enzyme → blocks the production of squalene epoxide Squalene Epoxide Flucytosine, Griseofulvin, and Terbinafine, which is a precursor to ergosterol and a critical component of the cell membrane Cell Membrane A cell membrane (also known as the plasma membrane or plasmalemma) is a biological membrane that separates the cell contents from the outside environment. A cell membrane is composed of a phospholipid bilayer and proteins that function to protect cellular DNA and mediate the exchange of ions and molecules. The Cell: Cell Membrane |
|
| Flucytosine Flucytosine Flucytosine is a pyrimidine analog that disrupts fungal DNA and RNA synthesis. Flucytosine is always used in combination with other antifungal agents and is primarily used to treat cryptococcal meningitis. Flucytosine, Griseofulvin, and Terbinafine | A
pyrimidine analog
Pyrimidine Analog
Flucytosine, Griseofulvin, and Terbinafine with metabolites:
|
|