The glycopeptide antibiotics (GPAs) vancomycin and teicoplanin are inhibitors of bacterial cell wall Cell wall The outermost layer of a cell in most plants; bacteria; fungi; and algae. The cell wall is usually a rigid structure that lies external to the cell membrane, and provides a protective barrier against physical or chemical agents. Cell Types: Eukaryotic versus Prokaryotic synthesis Synthesis Polymerase Chain Reaction (PCR) and considered the last resort treatment of severe infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease due to gram-positive Gram-Positive Penicillins bacteria Bacteria Bacteria are prokaryotic single-celled microorganisms that are metabolically active and divide by binary fission. Some of these organisms play a significant role in the pathogenesis of diseases. Bacteriology such as Staphylococcus aureus Staphylococcus aureus Potentially pathogenic bacteria found in nasal membranes, skin, hair follicles, and perineum of warm-blooded animals. They may cause a wide range of infections and intoxications. Brain Abscess, Enterococcus Enterococcus Enterococcus is a genus of oval-shaped gram-positive cocci that are arranged in pairs or short chains. Distinguishing factors include optochin resistance and the presence of pyrrolidonyl arylamidase (PYR) and Lancefield D antigen. Enterococcus is part of the normal flora of the human GI tract. Enterococcus spp., and Clostridiodes difficile. Vancomycin is the only GPA GPA A multisystemic disease of a complex genetic background. It is characterized by inflammation of the blood vessels (vasculitis) leading to damage in any number of organs. The common features include granulomatous inflammation of the respiratory tract and kidneys. Most patients have measurable autoantibodies (antineutrophil cytoplasmic antibodies) against myeloblastin. Granulomatosis with Polyangiitis available in the United States. The medication has poor absorption Absorption Absorption involves the uptake of nutrient molecules and their transfer from the lumen of the GI tract across the enterocytes and into the interstitial space, where they can be taken up in the venous or lymphatic circulation. Digestion and Absorption in the GI tract; therefore, oral vancomycin is used for infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease of the intestinal lumen including C. difficile. Intravenous vancomycin is indicated for severe gram-positive Gram-Positive Penicillins infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease including endocarditis Endocarditis Endocarditis is an inflammatory disease involving the inner lining (endometrium) of the heart, most commonly affecting the cardiac valves. Both infectious and noninfectious etiologies lead to vegetations on the valve leaflets. Patients may present with nonspecific symptoms such as fever and fatigue. Endocarditis, pneumonia Pneumonia Pneumonia or pulmonary inflammation is an acute or chronic inflammation of lung tissue. Causes include infection with bacteria, viruses, or fungi. In more rare cases, pneumonia can also be caused through toxic triggers through inhalation of toxic substances, immunological processes, or in the course of radiotherapy. Pneumonia, and bacteremia. Significant adverse effects include anaphylaxis Anaphylaxis An acute hypersensitivity reaction due to exposure to a previously encountered antigen. The reaction may include rapidly progressing urticaria, respiratory distress, vascular collapse, systemic shock, and death. Type I Hypersensitivity Reaction, hypersensitivity reactions, red man syndrome (related to rapid infusion), nephrotoxicity, and ototoxicity.
Last updated: May 16, 2024
Glycopeptide antibiotics (GPAs) are actinomycete-derived, glycosylated, nonribosomal peptides, which target gram-positive Gram-Positive Penicillins bacteria Bacteria Bacteria are prokaryotic single-celled microorganisms that are metabolically active and divide by binary fission. Some of these organisms play a significant role in the pathogenesis of diseases. Bacteriology by inhibition of cell wall Cell wall The outermost layer of a cell in most plants; bacteria; fungi; and algae. The cell wall is usually a rigid structure that lies external to the cell membrane, and provides a protective barrier against physical or chemical agents. Cell Types: Eukaryotic versus Prokaryotic synthesis Synthesis Polymerase Chain Reaction (PCR):
Both vancomycin and teicoplanin are heptapeptides, but the carbohydrate groups of each drug differ.
Chemical structure of vancomycin
Image: “Vancomycin” by BartVL71. License: Public Domain
Chemical structure of teicoplanin
Image: “Teicoplanin core and major components” by Fvasconcellos. License: Public Domain, cropped by Lecturio.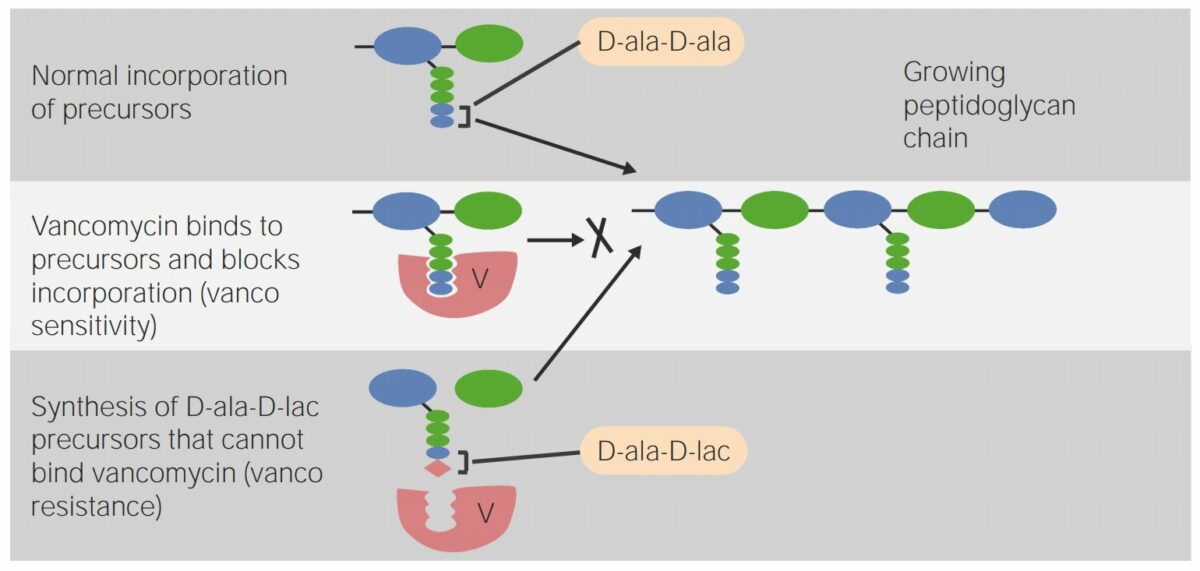
Mechanism of action:
Vancomycin interferes with the normal incorporation of peptidoglycan precursors.
The bottom row shows vancomycin resistance:
Synthesis of D-alanyl-D-lactate precursors in the bacterial cell wall results in poor binding with vancomycin, which reduces the effectivity of the antibiotic.
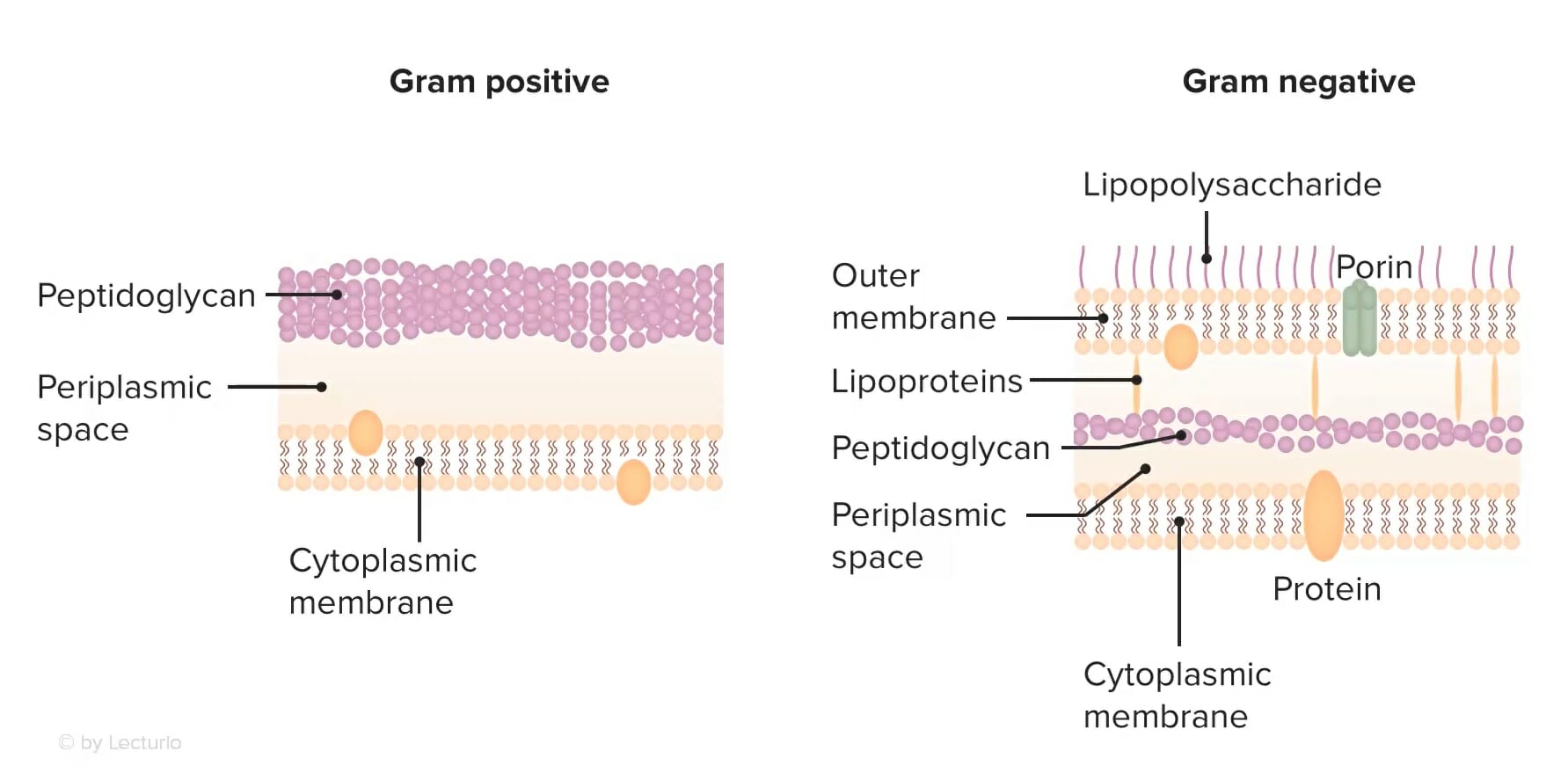
The cell wall structure of gram-positive versus gram-negative bacteria:
Note the peptidoglycan layer of the cell wall is located on the surface, allowing easier access for glycopeptide antibiotics to exert a bactericidal effect. Conversely, gram-negative bacteria have a lipopolysaccharide membrane that is impermeable to large molecules (such as glycopeptides).
Glycopeptide antibiotics have broad activity against gram-positive Gram-Positive Penicillins bacterial infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease and are often used as the last resort treatment for serious conditions.
| Drug | Indications | Clinical Pearls |
|---|---|---|
| Vancomycin |
|
|
| Teicoplanin | Similar spectrum of activity as vancomycin |
|
The following antibiotics are agents with activity against gram-positive Gram-Positive Penicillins bacteria Bacteria Bacteria are prokaryotic single-celled microorganisms that are metabolically active and divide by binary fission. Some of these organisms play a significant role in the pathogenesis of diseases. Bacteriology. All act on the bacterial cell wall Cell wall The outermost layer of a cell in most plants; bacteria; fungi; and algae. The cell wall is usually a rigid structure that lies external to the cell membrane, and provides a protective barrier against physical or chemical agents. Cell Types: Eukaryotic versus Prokaryotic through varying mechanisms.
| Class of antibiotics | Mechanism of action | Drugs |
|---|---|---|
| Lipopeptides Lipopeptides Lipopeptides and lipoglycopeptides are classes of antibiotics that have activity against gram-positive bacteria and that act on the bacterial cell wall. Lipopeptides (daptomycin) disrupt the cell membrane by creating an ion-conducting channel and causing potassium efflux and membrane depolarization. Lipopeptides and Lipoglycopeptides | Disruption of the bacterial cell membrane Cell Membrane A cell membrane (also known as the plasma membrane or plasmalemma) is a biological membrane that separates the cell contents from the outside environment. A cell membrane is composed of a phospholipid bilayer and proteins that function to protect cellular DNA and mediate the exchange of ions and molecules. The Cell: Cell Membrane by generating an ion-conducting channel, depolarizing the membrane, and leading to cell death Cell death Injurious stimuli trigger the process of cellular adaptation, whereby cells respond to withstand the harmful changes in their environment. Overwhelmed adaptive mechanisms lead to cell injury. Mild stimuli produce reversible injury. If the stimulus is severe or persistent, injury becomes irreversible. Apoptosis is programmed cell death, a mechanism with both physiologic and pathologic effects. Cell Injury and Death | Daptomycin Daptomycin A cyclic lipopeptide antibiotic that inhibits gram-positive bacteria. Lipopeptides and Lipoglycopeptides |
| Glycopeptides | Inhibition of cell wall Cell wall The outermost layer of a cell in most plants; bacteria; fungi; and algae. The cell wall is usually a rigid structure that lies external to the cell membrane, and provides a protective barrier against physical or chemical agents. Cell Types: Eukaryotic versus Prokaryotic synthesis Synthesis Polymerase Chain Reaction (PCR) by binding to the D-alanyl-D-alanine terminus of cell wall Cell wall The outermost layer of a cell in most plants; bacteria; fungi; and algae. The cell wall is usually a rigid structure that lies external to the cell membrane, and provides a protective barrier against physical or chemical agents. Cell Types: Eukaryotic versus Prokaryotic peptidoglycan Peptidoglycan Penicillins (PG) precursors |
|
| Lipoglycopeptides Lipoglycopeptides Lipopeptides and lipoglycopeptides are classes of antibiotics that have activity against gram-positive bacteria and that act on the bacterial cell wall. Lipoglycopeptides have a dual action of inhibiting bacterial cell-wall synthesis by binding peptidoglycan precursors and causing cell membrane depolarization. Lipopeptides and Lipoglycopeptides | Dual action of inhibition of cell wall Cell wall The outermost layer of a cell in most plants; bacteria; fungi; and algae. The cell wall is usually a rigid structure that lies external to the cell membrane, and provides a protective barrier against physical or chemical agents. Cell Types: Eukaryotic versus Prokaryotic synthesis Synthesis Polymerase Chain Reaction (PCR) and depolarization Depolarization Membrane Potential of the cell membrane Cell Membrane A cell membrane (also known as the plasma membrane or plasmalemma) is a biological membrane that separates the cell contents from the outside environment. A cell membrane is composed of a phospholipid bilayer and proteins that function to protect cellular DNA and mediate the exchange of ions and molecules. The Cell: Cell Membrane |
|
Different antibiotics have varying degrees of activity against different bacteria Bacteria Bacteria are prokaryotic single-celled microorganisms that are metabolically active and divide by binary fission. Some of these organisms play a significant role in the pathogenesis of diseases. Bacteriology. The table below outlines the antibiotics with activity against 3 important classes of bacteria Bacteria Bacteria are prokaryotic single-celled microorganisms that are metabolically active and divide by binary fission. Some of these organisms play a significant role in the pathogenesis of diseases. Bacteriology: gram-positive Gram-Positive Penicillins cocci Cocci Bacteriology, gram-negative bacilli Bacilli Shigella, and anaerobes Anaerobes Lincosamides.
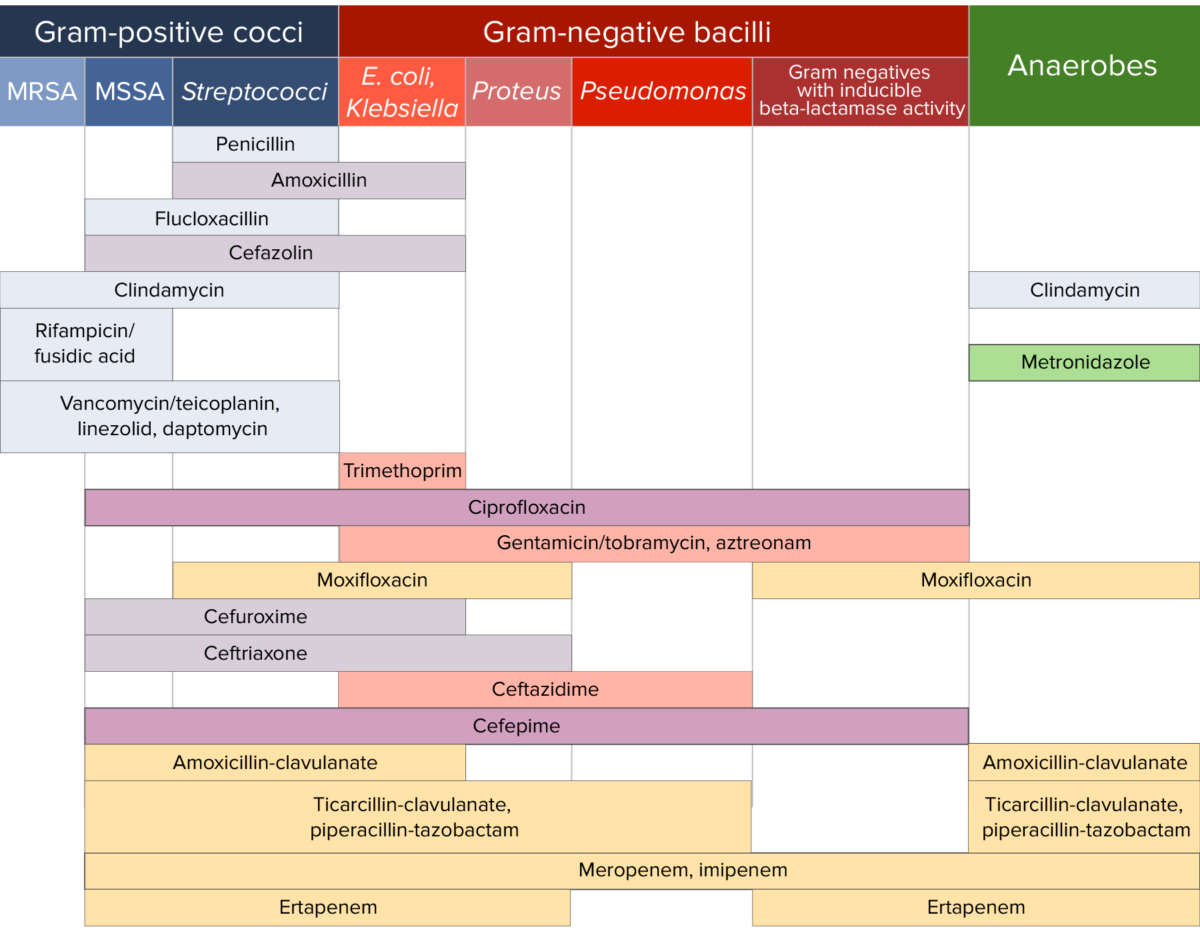
Antibiotic sensitivity:
Chart comparing the microbial coverage of different antibiotics for gram-positive cocci, gram-negative bacilli, and anaerobes.