Cephalosporins are a group of bactericidal Bactericidal Penicillins beta-lactam Beta-Lactam Penicillins antibiotics (similar to penicillins Penicillins Beta-lactam antibiotics contain a beta-lactam ring as a part of their chemical structure. Drugs in this class include penicillin G and V, penicillinase-sensitive and penicillinase-resistant penicillins, cephalosporins, carbapenems, and aztreonam. Penicillins) that exert their effects by preventing bacteria Bacteria Bacteria are prokaryotic single-celled microorganisms that are metabolically active and divide by binary fission. Some of these organisms play a significant role in the pathogenesis of diseases. Bacteriology from producing their cell walls, ultimately leading to cell death Cell death Injurious stimuli trigger the process of cellular adaptation, whereby cells respond to withstand the harmful changes in their environment. Overwhelmed adaptive mechanisms lead to cell injury. Mild stimuli produce reversible injury. If the stimulus is severe or persistent, injury becomes irreversible. Apoptosis is programmed cell death, a mechanism with both physiologic and pathologic effects. Cell Injury and Death. Cephalosporins are categorized by generation and all drug names begin with “cef-” or “ceph-.” Cephalosporins have expanded antimicrobial activity compared with most penicillins Penicillins Beta-lactam antibiotics contain a beta-lactam ring as a part of their chemical structure. Drugs in this class include penicillin G and V, penicillinase-sensitive and penicillinase-resistant penicillins, cephalosporins, carbapenems, and aztreonam. Penicillins, and are more effective against Enterobacteriaceae; some drugs are active against Pseudomonas Pseudomonas Pseudomonas is a non-lactose-fermenting, gram-negative bacillus that produces pyocyanin, which gives it a characteristic blue-green color. Pseudomonas is found ubiquitously in the environment, as well as in moist reservoirs, such as hospital sinks and respiratory equipment. Pseudomonas and/or anaerobic species as well. Cephalosporins are often used to treat skin Skin The skin, also referred to as the integumentary system, is the largest organ of the body. The skin is primarily composed of the epidermis (outer layer) and dermis (deep layer). The epidermis is primarily composed of keratinocytes that undergo rapid turnover, while the dermis contains dense layers of connective tissue. Skin: Structure and Functions, soft tissue Soft Tissue Soft Tissue Abscess, bone Bone Bone is a compact type of hardened connective tissue composed of bone cells, membranes, an extracellular mineralized matrix, and central bone marrow. The 2 primary types of bone are compact and spongy. Bones: Structure and Types, lung, urinary tract Urinary tract The urinary tract is located in the abdomen and pelvis and consists of the kidneys, ureters, urinary bladder, and urethra. The structures permit the excretion of urine from the body. Urine flows from the kidneys through the ureters to the urinary bladder and out through the urethra. Urinary Tract: Anatomy, intraabdominal, and pelvic infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease caused by susceptible organisms.
Last updated: Oct 6, 2022
Cephalosporins are members of the beta-lactam Beta-Lactam Penicillins drug family and consist of:
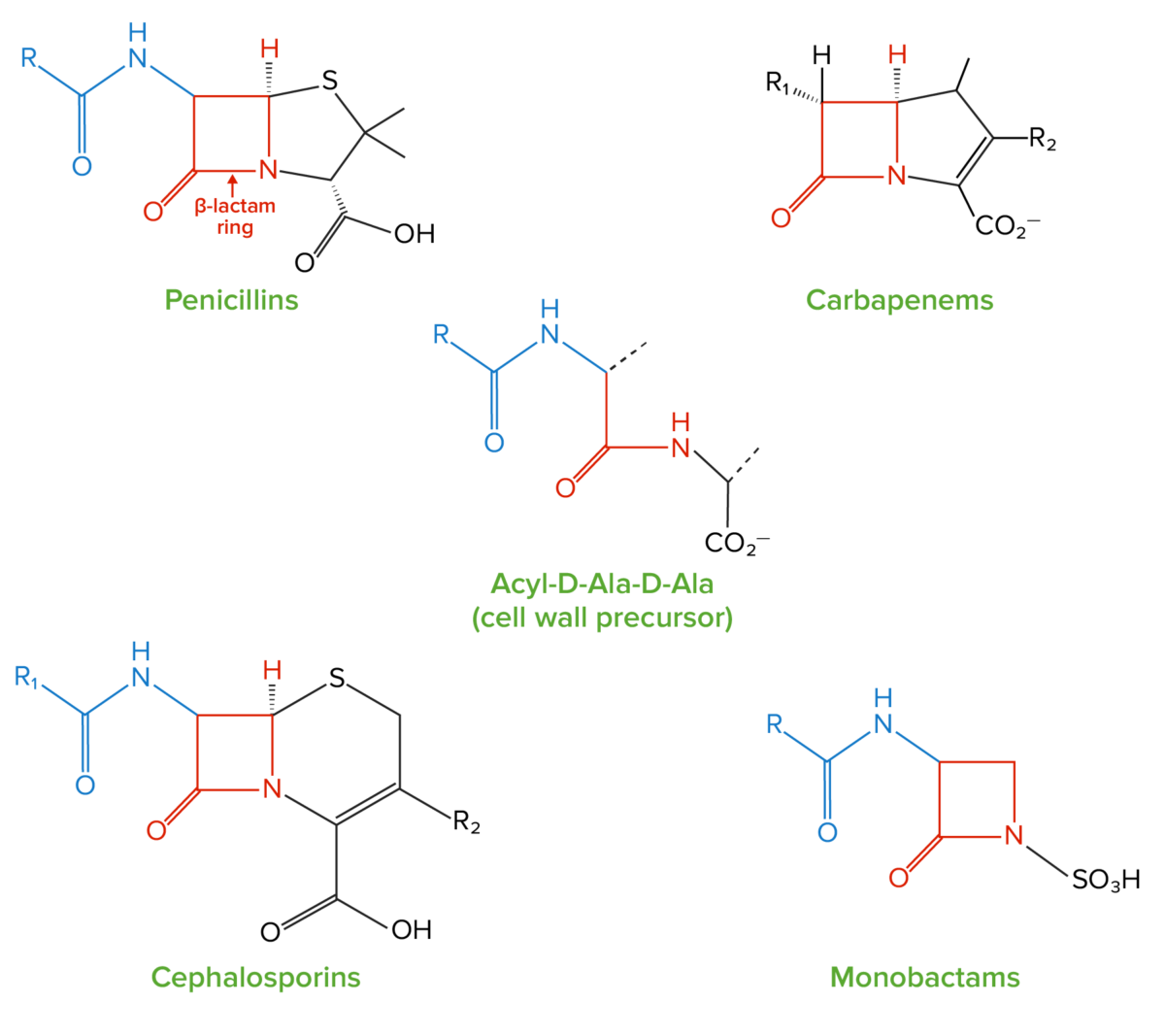
Structure of beta-lactams:
All beta-lactam antibiotics contain the same core 4-membered “beta-lactam” ring (highlighted in red). This ring is responsible for the antibacterial properties of the drug because it is the region that binds to and inhibits the penicillin-binding proteins (PBPs). The PBPs catalyze the formation of the cell wall by generating crosslinks between the peptide chains in the peptidoglycan molecules; PBPs form these crosslinks between acyl-D-Ala-D-Ala peptides, which have a structure similar to the beta-lactam ring.
All beta-lactams, including cephalosporins, exert their effects by inhibiting bacterial cell wall Cell wall The outermost layer of a cell in most plants; bacteria; fungi; and algae. The cell wall is usually a rigid structure that lies external to the cell membrane, and provides a protective barrier against physical or chemical agents. Cell Types: Eukaryotic versus Prokaryotic synthesis Synthesis Polymerase Chain Reaction (PCR).
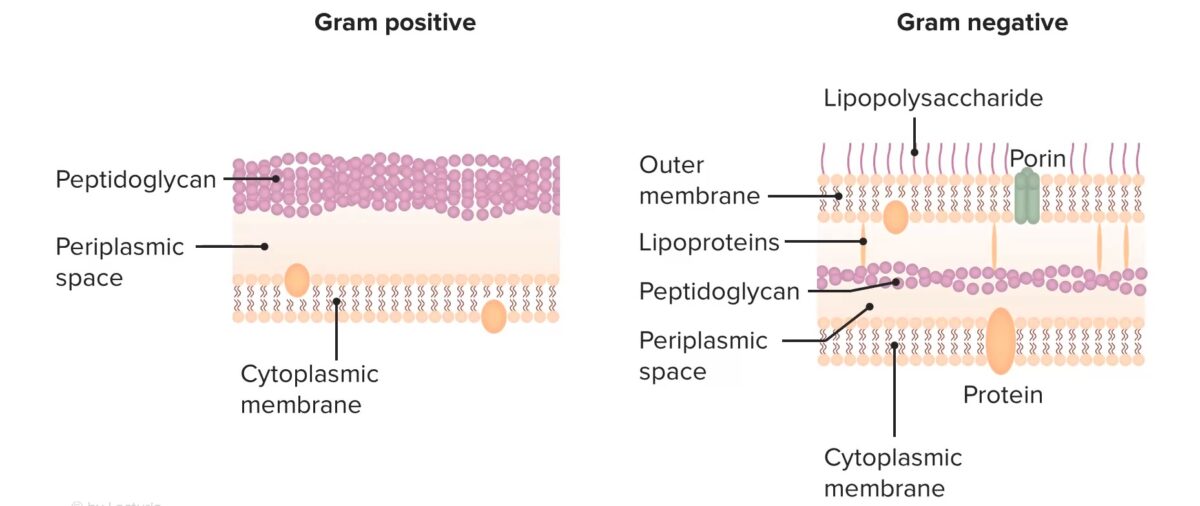
Structure of bacterial cell walls
Image by Lecturio. License: CC BY-NC-SA 4.0All beta-lactams work by irreversibly binding to and inhibiting the PBPs PBPs Bacterial proteins that share the property of binding irreversibly to penicillins and other antibacterial agents derived from lactams. The penicillin-binding proteins are primarily enzymes involved in cell wall biosynthesis including muramoylpentapeptide carboxypeptidase; peptide synthases; transpeptidases; and hexosyltransferases. Penicillins → beta-lactam Beta-Lactam Penicillins antibiotics inhibit cell wall Cell wall The outermost layer of a cell in most plants; bacteria; fungi; and algae. The cell wall is usually a rigid structure that lies external to the cell membrane, and provides a protective barrier against physical or chemical agents. Cell Types: Eukaryotic versus Prokaryotic synthesis Synthesis Polymerase Chain Reaction (PCR).
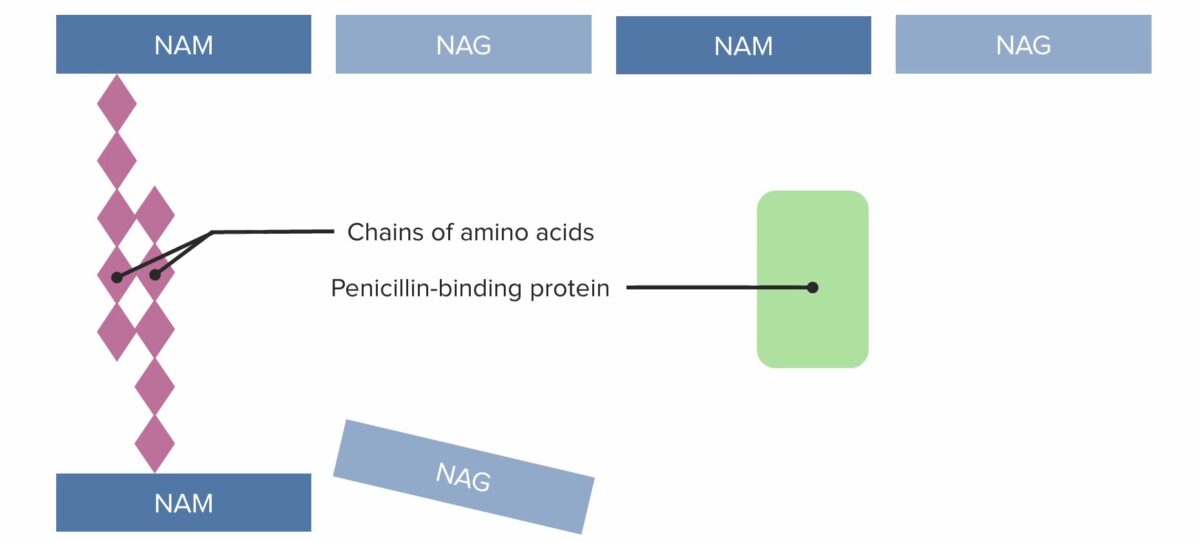
(1) Penicillin-binding protein (PBP) forming crosslinked bridges between adjacent peptidoglycan chains
Image by Lecturio. License: CC BY-NC-SA 4.0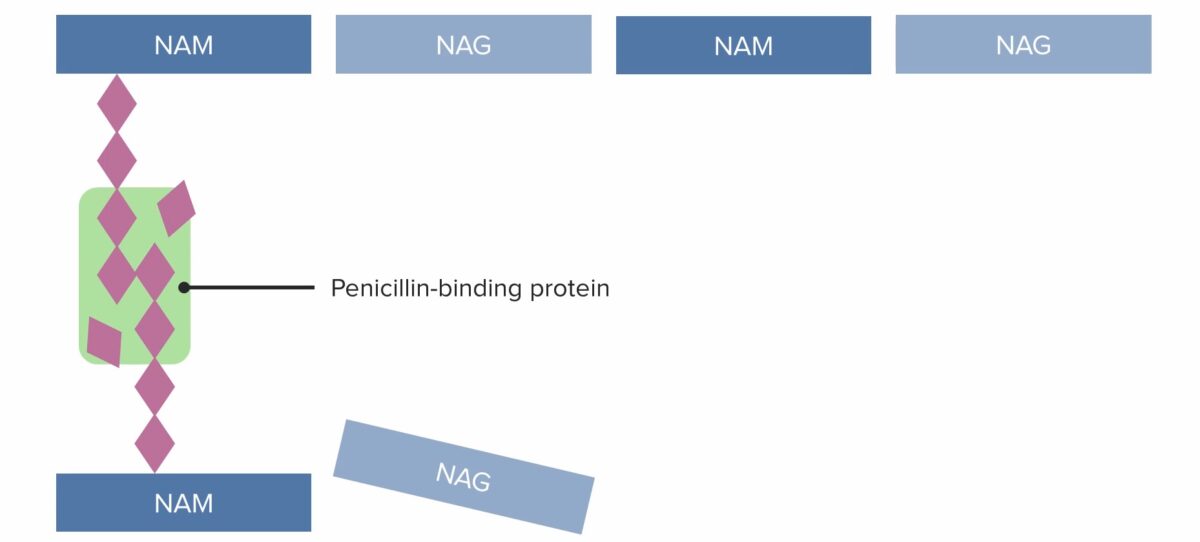
(2) Penicillin-binding protein (PBP) forming crosslinked bridges between adjacent peptidoglycan chains
Image by Lecturio. License: CC BY-NC-SA 4.0
(3) Penicillin-binding protein (PBP) forming crosslinked bridges between adjacent peptidoglycan chains
Image by Lecturio. License: CC BY-NC-SA 4.0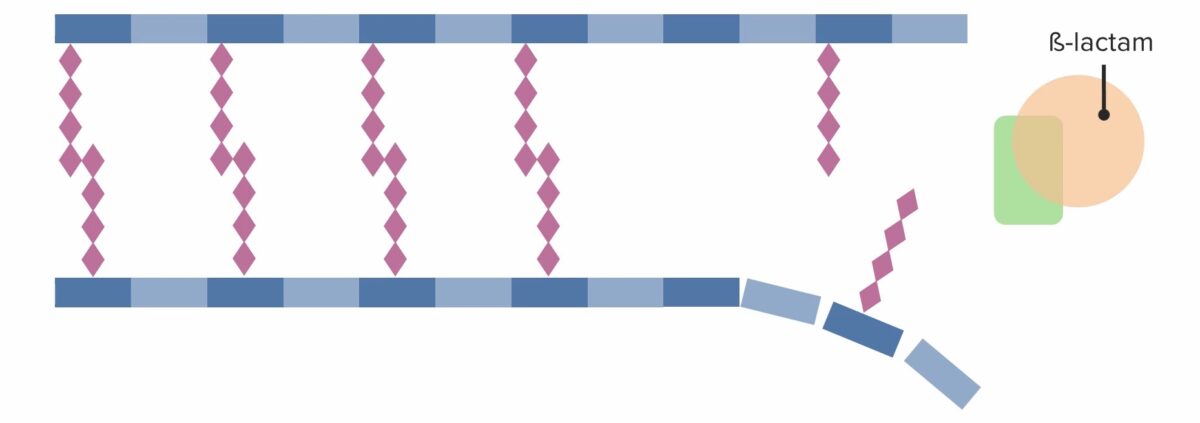
Presence of a beta-lactam antibiotic, which irreversibly binds to and inhibits the PBP, preventing it from forming new crosslinks:
Effective inhibition of further cell wall synthesis ultimately leading to cell death
Beta-lactams, including cephalosporins, have a bactericidal Bactericidal Penicillins (rather than bacteriostatic Bacteriostatic Sulfonamides and Trimethoprim) effect.
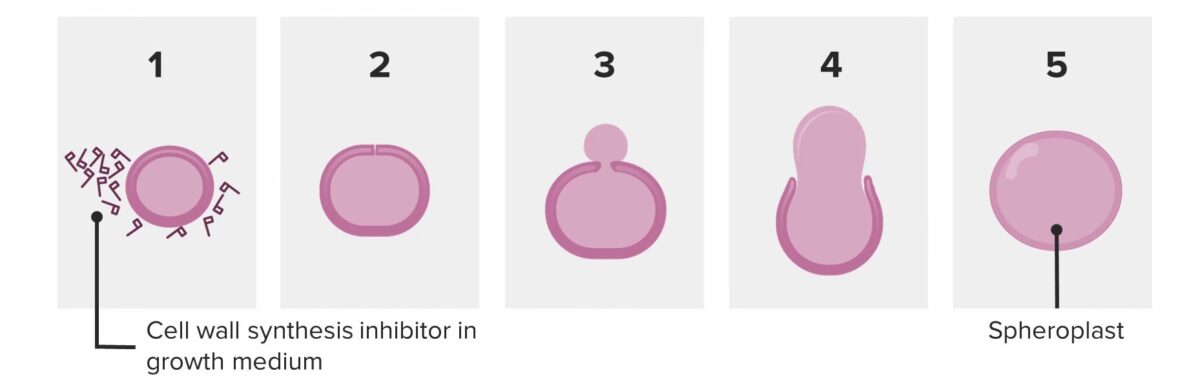
Bacterium attempting to divide in the presence of penicillin:
The bacterium sheds its wall and becomes a spheroplast. The spheroplast is unable to survive and autocatalyzes (dies).
Bacteria Bacteria Bacteria are prokaryotic single-celled microorganisms that are metabolically active and divide by binary fission. Some of these organisms play a significant role in the pathogenesis of diseases. Bacteriology use 3 primary mechanisms to resist cephalosporins:
Thus, ceftriaxone should not be reconstituted/directly mixed with calcium-containing products such as lactated Ringers or total parenteral nutrition Parenteral nutrition The administering of nutrients for assimilation and utilization by a patient who cannot maintain adequate nutrition by enteral feeding alone. Nutrients are administered by a route other than the alimentary canal (e.g., intravenously, subcutaneously). Central Venous Catheter (TPN).
Cephalosporins are generally classified by generation, with the 1st generation drugs being the oldest in this class. Some of the most commonly used drugs are shown in the table below.
| Generation | Parental agents | Oral agents |
|---|---|---|
| 1st generation | Cefazolin (Ancef) | Cephalexin (Keflex) |
| 2nd generation |
|
Cefaclor (Ceclor) |
| 3rd generation |
|
|
| 4th generation | Cefepime | None |
| 5th generation and advanced combinations |
|
None |
| Drugs | Gram-positive Gram-Positive Penicillins cocci Cocci Bacteriology | Gram-negative bacilli Bacilli Shigella | Gram-negative cocci Cocci Bacteriology | Anaerobes Anaerobes Lincosamides | ||
|---|---|---|---|---|---|---|
| Streptococcus Streptococcus Streptococcus is one of the two medically important genera of gram-positive cocci, the other being Staphylococcus. Streptococci are identified as different species on blood agar on the basis of their hemolytic pattern and sensitivity to optochin and bacitracin. There are many pathogenic species of streptococci, including S. pyogenes, S. agalactiae, S. pneumoniae, and the viridans streptococci. Streptococcus, MSSA |
|
SPACE organisms | Pseudomonas Pseudomonas Pseudomonas is a non-lactose-fermenting, gram-negative bacillus that produces pyocyanin, which gives it a characteristic blue-green color. Pseudomonas is found ubiquitously in the environment, as well as in moist reservoirs, such as hospital sinks and respiratory equipment. Pseudomonas | Neisseria Neisseria Neisseria is a genus of bacteria commonly present on mucosal surfaces. Several species exist, but only 2 are pathogenic to humans: N. gonorrhoeae and N. meningitidis. Neisseria species are non-motile, gram-negative diplococci most commonly isolated on modified Thayer-Martin (MTM) agar. Neisseriaspp | Peptostreptococcus Peptostreptococcus A genus of gram-positive, anaerobic, coccoid bacteria that is part of the normal flora of humans. Its organisms are opportunistic pathogens causing bacteremias and soft tissue infections. Perianal and Perirectal Abscess | |
1st generation
|
X | X* | X | |||
2nd generation
|
X | X | X** | X+ | ||
3rd generation
|
X | X | X++ | X | X | |
4th generation
|
X | X | X | X | X+ | |
5th generation and combinations:
|
X | X | X | X | X+ | |
Cephalosporins are used to treat infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease caused by susceptible organisms, including:
Allergic reactions Allergic Reactions Type I hypersensitivity reaction against plasma proteins in donor blood Transfusion Reactions are the most common adverse effects.
Antibiotics can be classified in several ways. One way is to classify them based on their mechanism of action:
| Mechanism | Classes of antibiotics |
|---|---|
| Bacterial cell wall Cell wall The outermost layer of a cell in most plants; bacteria; fungi; and algae. The cell wall is usually a rigid structure that lies external to the cell membrane, and provides a protective barrier against physical or chemical agents. Cell Types: Eukaryotic versus Prokaryotic synthesis Synthesis Polymerase Chain Reaction (PCR) inhibitors |
|
| Bacterial protein synthesis Synthesis Polymerase Chain Reaction (PCR) inhibitors |
|
| Agents acting against DNA DNA A deoxyribonucleotide polymer that is the primary genetic material of all cells. Eukaryotic and prokaryotic organisms normally contain DNA in a double-stranded state, yet several important biological processes transiently involve single-stranded regions. DNA, which consists of a polysugar-phosphate backbone possessing projections of purines (adenine and guanine) and pyrimidines (thymine and cytosine), forms a double helix that is held together by hydrogen bonds between these purines and pyrimidines (adenine to thymine and guanine to cytosine). DNA Types and Structure and/or folate Folate Folate and vitamin B12 are 2 of the most clinically important water-soluble vitamins. Deficiencies can present with megaloblastic anemia, GI symptoms, neuropsychiatric symptoms, and adverse pregnancy complications, including neural tube defects. Folate and Vitamin B12 |
|
| Antimycobacterial agents Antimycobacterial Agents Antimycobacterial agents represent a diverse group of compounds that have activity against mycobacterial infections, including tuberculosis, leprosy and Mycobacterium avium complex (MAC) disease. The 1st-line agents for tuberculosis are rifampin, isoniazid, pyrazinamide, and ethambutol. Antimycobacterial Drugs |
|
Different antibiotics have varying degrees of activity against different bacteria Bacteria Bacteria are prokaryotic single-celled microorganisms that are metabolically active and divide by binary fission. Some of these organisms play a significant role in the pathogenesis of diseases. Bacteriology. The table below outlines the antibiotics that are active against 3 important classes of bacteria Bacteria Bacteria are prokaryotic single-celled microorganisms that are metabolically active and divide by binary fission. Some of these organisms play a significant role in the pathogenesis of diseases. Bacteriology, including gram-positive Gram-Positive Penicillins cocci Cocci Bacteriology, gram-negative bacilli Bacilli Shigella, and anaerobes Anaerobes Lincosamides.
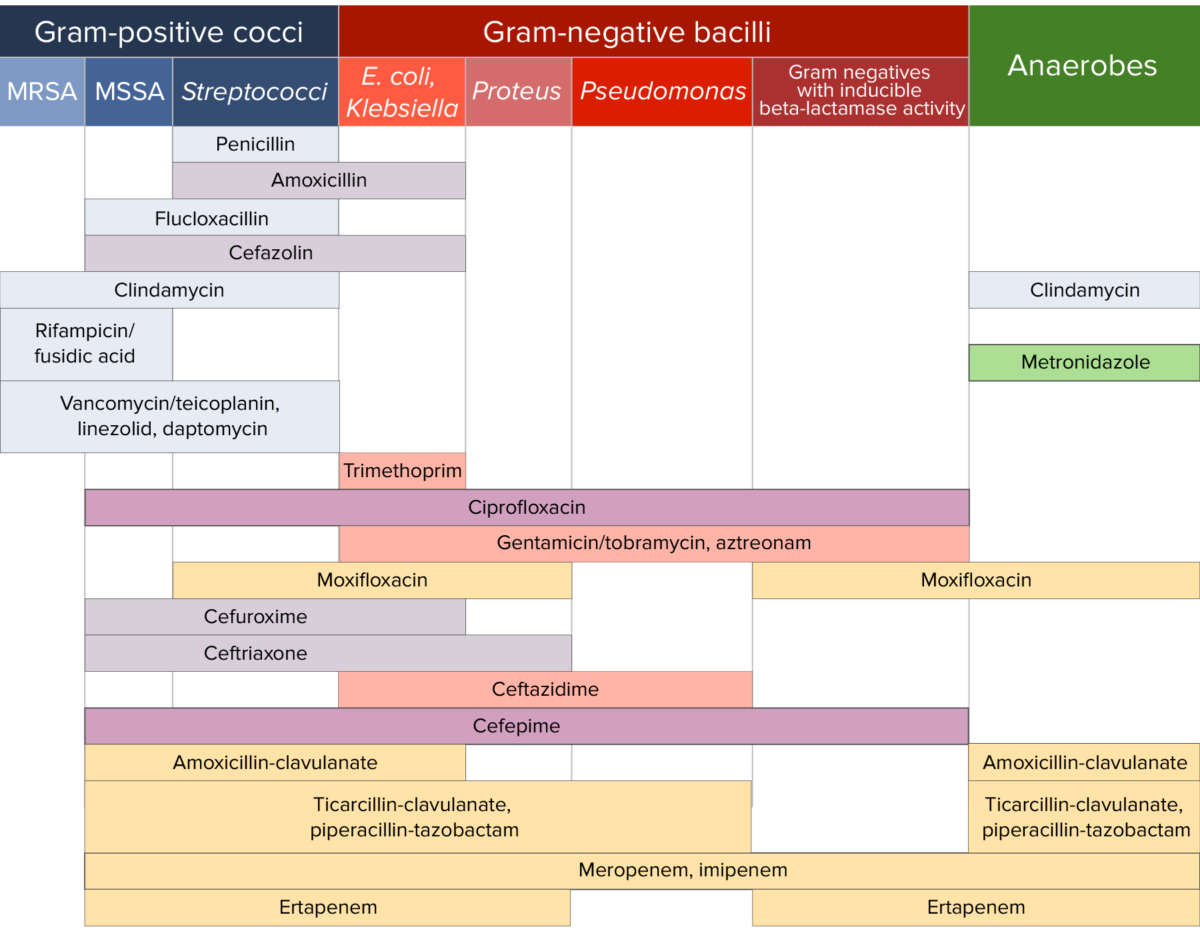
Antibiotic sensitivity:
Chart comparing the microbial coverage of different antibiotics for gram-positive cocci, gram-negative bacilli, and anaerobes.