Polyenes are a class of fungicidal agents that consist of 2 primary drugs in current use, namely, nystatin and amphotericin B. Both these drugs exert their effects by binding to ergosterol Ergosterol A steroid occurring in fungi. Irradiation with ultraviolet rays results in formation of ergocalciferol (vitamin d2). Azoles (a critical component of fungal cell membranes) and creating pores in the membrane, leading to the leakage of intracellular components and ultimately cell lysis. Both nystatin and amphotericin B have a broad spectrum Broad Spectrum Macrolides and Ketolides of antifungal Antifungal Azoles activity, little to no oral absorption Absorption Absorption involves the uptake of nutrient molecules and their transfer from the lumen of the GI tract across the enterocytes and into the interstitial space, where they can be taken up in the venous or lymphatic circulation. Digestion and Absorption, and significant toxicity Toxicity Dosage Calculation (especially nephrotoxicity Nephrotoxicity Glycopeptides) when used parenterally. Nystatin is too toxic to be administered intravenously; thus, it is only used to treat fungal infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease of the skin Skin The skin, also referred to as the integumentary system, is the largest organ of the body. The skin is primarily composed of the epidermis (outer layer) and dermis (deep layer). The epidermis is primarily composed of keratinocytes that undergo rapid turnover, while the dermis contains dense layers of connective tissue. Skin: Structure and Functions, mucous membranes, and GI lumen. Amphotericin B is administered via IV, intrathecal, or intraperitoneal Intraperitoneal Peritoneum: Anatomy routes, or using aerosols Aerosols Colloids with a gaseous dispersing phase and either liquid (fog) or solid (smoke) dispersed phase; used in fumigation or in inhalation therapy; may contain propellant agents. Coxiella/Q Fever to treat serious, life-threatening fungal infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease. Secondary resistance Resistance Physiologically, the opposition to flow of air caused by the forces of friction. As a part of pulmonary function testing, it is the ratio of driving pressure to the rate of air flow. Ventilation: Mechanics of Breathing to nystatin and amphotericin B is rare but emerging.
Last updated: Dec 15, 2025
Polyenes are one of the original classes of antifungal Antifungal Azoles medications. There are 2 primary polyenes that are currently used:
Nystatin and amphotericin B have very similar chemical structures, including:
Chemical structure of nystatin
Image: “Nystatin” by Hoffmeier K. License: Public Domain
Chemical structure of amphotericin B
Image: “Amphotericin B” by The chemistds. License: Public DomainPolyenes exert their effects by creating pores in the fungal cell membrane Cell Membrane A cell membrane (also known as the plasma membrane or plasmalemma) is a biological membrane that separates the cell contents from the outside environment. A cell membrane is composed of a phospholipid bilayer and proteins that function to protect cellular DNA and mediate the exchange of ions and molecules. The Cell: Cell Membrane through binding to ergosterol Ergosterol A steroid occurring in fungi. Irradiation with ultraviolet rays results in formation of ergocalciferol (vitamin d2). Azoles; however, the exact mechanism of action is unclear.
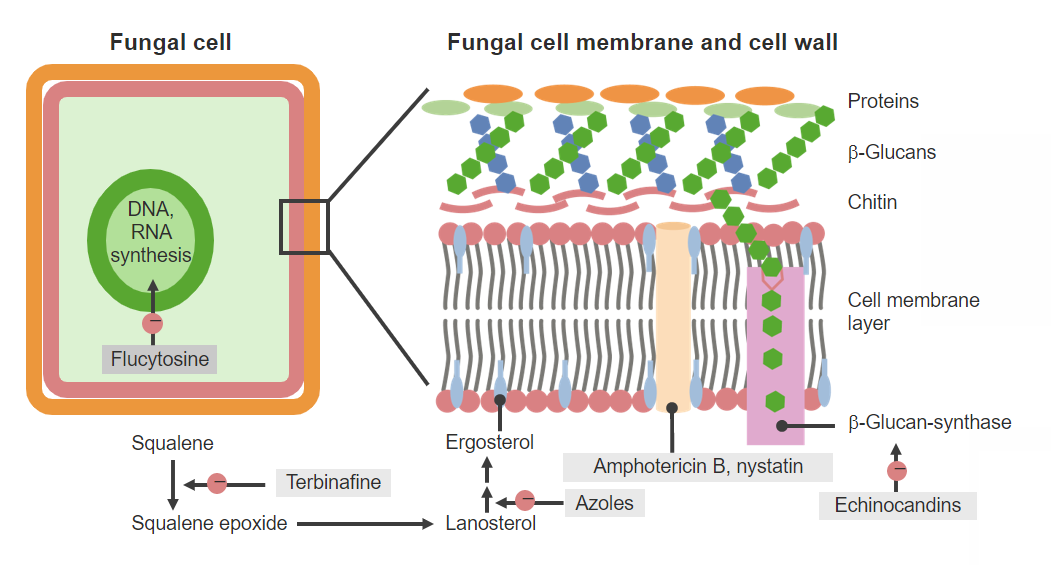
Antifungal agents and mechanisms of action
Image by Lecturio. License: CC BY-NC-SA 4.0Nystatin is only used topically and as an oral suspension (administered with instructions to “swish and swallow”).
Nystatin is used for susceptible Candida Candida Candida is a genus of dimorphic, opportunistic fungi. Candida albicans is part of the normal human flora and is the most common cause of candidiasis. The clinical presentation varies and can include localized mucocutaneous infections (e.g., oropharyngeal, esophageal, intertriginous, and vulvovaginal candidiasis) and invasive disease (e.g., candidemia, intraabdominal abscess, pericarditis, and meningitis). Candida/Candidiasis species that cause fungal infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease in:
Owing to its toxicity Toxicity Dosage Calculation and the availability of less toxic agents, amphotericin B should be used only for individuals with severe, life-threatening, invasive fungal infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease, or those unable to tolerate alternative agents.
Spectrum of activity:
Amphotericin B is active against:
Resistance Resistance Physiologically, the opposition to flow of air caused by the forces of friction. As a part of pulmonary function testing, it is the ratio of driving pressure to the rate of air flow. Ventilation: Mechanics of Breathing:
Organisms with intrinsic resistance Resistance Physiologically, the opposition to flow of air caused by the forces of friction. As a part of pulmonary function testing, it is the ratio of driving pressure to the rate of air flow. Ventilation: Mechanics of Breathing to amphotericin B:
Topical and oral nystatin have very few adverse effects. The only contraindication is a known allergy Allergy An abnormal adaptive immune response that may or may not involve antigen-specific IgE Type I Hypersensitivity Reaction.
Therapy with amphotericin B is often limited by its toxicity Toxicity Dosage Calculation, especially drug-induced renal impairment. Adverse events can be divided into immediate infusion-related reactions Infusion-related reactions Cancer Immunotherapy and effects due to cumulative toxicity Toxicity Dosage Calculation.
Infusion-related toxicity Toxicity Dosage Calculation:
Cumulative toxicity Toxicity Dosage Calculation:
Contraindications Contraindications A condition or factor associated with a recipient that makes the use of a drug, procedure, or physical agent improper or inadvisable. Contraindications may be absolute (life threatening) or relative (higher risk of complications in which benefits may outweigh risks). Noninvasive Ventilation and Precautions:
Secondary resistance Resistance Physiologically, the opposition to flow of air caused by the forces of friction. As a part of pulmonary function testing, it is the ratio of driving pressure to the rate of air flow. Ventilation: Mechanics of Breathing to amphotericin B is rare but emerging. The primary mechanisms of resistance Resistance Physiologically, the opposition to flow of air caused by the forces of friction. As a part of pulmonary function testing, it is the ratio of driving pressure to the rate of air flow. Ventilation: Mechanics of Breathing include:
| Drug class (examples) | Mechanism of action | Clinical relevance |
|---|---|---|
| Azoles Azoles Azoles are a widely used class of antifungal medications inhibiting the production of ergosterol, a critical component in the fungal cell membrane. The 2 primary subclasses of azoles are the imidazoles, older agents typically only used for topical applications, and the triazoles, newer agents with a wide spectrum of uses. Azoles ( Fluconazole Fluconazole Triazole antifungal agent that is used to treat oropharyngeal candidiasis and cryptococcal meningitis in aids. Azoles, Voriconazole Voriconazole A triazole antifungal agent that specifically inhibits sterol 14-alpha-demethylase and cytochrome p-450 cyp3a. Azoles) | Inhibits the production of ergosterol Ergosterol A steroid occurring in fungi. Irradiation with ultraviolet rays results in formation of ergocalciferol (vitamin d2). Azoles (a critical component of the fungal cell membrane Cell Membrane A cell membrane (also known as the plasma membrane or plasmalemma) is a biological membrane that separates the cell contents from the outside environment. A cell membrane is composed of a phospholipid bilayer and proteins that function to protect cellular DNA and mediate the exchange of ions and molecules. The Cell: Cell Membrane) by blocking the lanosterol Lanosterol A triterpene that derives from the chair-boat-chair-boat folding of 2, 3-oxidosqualene. It is metabolized to cholesterol and cucurbitacins. Cholesterol Metabolism 14-α-demethylase enzyme |
|
| Polyenes (Amphotericin B, Nystatin) | Binds to ergosterol Ergosterol A steroid occurring in fungi. Irradiation with ultraviolet rays results in formation of ergocalciferol (vitamin d2). Azoles in the fungal cell membrane Cell Membrane A cell membrane (also known as the plasma membrane or plasmalemma) is a biological membrane that separates the cell contents from the outside environment. A cell membrane is composed of a phospholipid bilayer and proteins that function to protect cellular DNA and mediate the exchange of ions and molecules. The Cell: Cell Membrane creating artificial pores in the membrane → results in leakage of cellular components and leads to cell lysis (death) | Amphotericin B:
Nystatin:
|
| Echinocandins Echinocandins Echinocandins are a group of fungicidal agents that target the fungal cell wall. Echinocandins inhibit β-glucan synthase, which in turn inhibits the production of β-glucan, a key structural component of fungal cell walls. The 3 primary drugs in this class include caspofungin, micafungin, and anidulafungin. Echinocandins ( Caspofungin Caspofungin A cyclic lipopeptide echinocandin and beta-(1, 3)-d-glucan synthase inhibitor that is used to treat internal or systemic mycoses. Echinocandins, Micafungin Micafungin A cyclic lipo-hexapeptide echinocandin antifungal agent that is used for the treatment and prevention of candidiasis. Echinocandins, Anidulafungin Anidulafungin Echinocandin antifungal agent that is used in the treatment of candidemia and candidiasis. Echinocandins) | Inhibits β-glucan synthase (the enzyme synthesizing β-glucan and an important structural component of the fungal cell wall Fungal Cell Wall Echinocandins) → weakened cell wall Cell wall The outermost layer of a cell in most plants; bacteria; fungi; and algae. The cell wall is usually a rigid structure that lies external to the cell membrane, and provides a protective barrier against physical or chemical agents. Cell Types: Eukaryotic versus Prokaryotic → cell lysis |
|
| Griseofulvin Griseofulvin In addition to the 3 other major classes of antifungal agents (azoles, polyenes, and echinocandins), several other clinically important antifungal agents are used, including flucytosine, griseofulvin, and terbinafine. Griseofulvin acts within the stratum corneum of the skin and are used to treat dermatophyte infections of the skin, hair, and nails. Flucytosine, Griseofulvin, and Terbinafine |
|
|
| Terbinafine Terbinafine In addition to the 3 other major classes of antifungal agents (azoles, polyenes, and echinocandins), several other clinically important antifungal agents are used, including flucytosine, griseofulvin, and terbinafine. Terbinafine acts within the stratum corneum of the skin and are used to treat dermatophyte infections of the skin, hair, and nails. Flucytosine, Griseofulvin, and Terbinafine | Inhibits the squalene epoxidase Squalene Epoxidase Flucytosine, Griseofulvin, and Terbinafine enzyme → blocks the production of squalene epoxide Squalene Epoxide Flucytosine, Griseofulvin, and Terbinafine, which is a precursor to ergosterol Ergosterol A steroid occurring in fungi. Irradiation with ultraviolet rays results in formation of ergocalciferol (vitamin d2). Azoles and a critical component of the cell membrane Cell Membrane A cell membrane (also known as the plasma membrane or plasmalemma) is a biological membrane that separates the cell contents from the outside environment. A cell membrane is composed of a phospholipid bilayer and proteins that function to protect cellular DNA and mediate the exchange of ions and molecules. The Cell: Cell Membrane |
|
| Flucytosine Flucytosine Flucytosine is a pyrimidine analog that disrupts fungal DNA and RNA synthesis. Flucytosine is always used in combination with other antifungal agents and is primarily used to treat cryptococcal meningitis. Flucytosine, Griseofulvin, and Terbinafine | A
pyrimidine analog
Pyrimidine Analog
Flucytosine, Griseofulvin, and Terbinafine with metabolites:
|
|