Acetaminophen is an over-the-counter nonopioid analgesic and antipyretic medication and the most commonly used analgesic worldwide. Despite the widespread use of acetaminophen, its mechanism of action is not entirely understood. However, acetaminophen is believed to exert its effects through indirect and reversible inhibition of cyclooxygenase Cyclooxygenase Nonsteroidal Antiinflammatory Drugs (NSAIDs) (COX)-1 and COX-2. The effects are generally limited to the CNS. Acetaminophen is primarily metabolized by the liver Liver The liver is the largest gland in the human body. The liver is found in the superior right quadrant of the abdomen and weighs approximately 1.5 kilograms. Its main functions are detoxification, metabolism, nutrient storage (e.g., iron and vitamins), synthesis of coagulation factors, formation of bile, filtration, and storage of blood. Liver: Anatomy; therefore, an overdose can lead to life-threatening hepatotoxicity. In adults, limiting the total acetaminophen dose (from all sources and routes) to < 4000 mg/day is highly recommended. Contraindications Contraindications A condition or factor associated with a recipient that makes the use of a drug, procedure, or physical agent improper or inadvisable. Contraindications may be absolute (life threatening) or relative (higher risk of complications in which benefits may outweigh risks). Noninvasive Ventilation to acetaminophen use include hypersensitivity, severe hepatic impairment, or severe active hepatic disease.
Last updated: Dec 15, 2025
Acetaminophen, also known as paracetamol (N-acetyl-p-aminophenol), is a nonopioid analgesic and antipyretic agent used to treat pain Pain An unpleasant sensation induced by noxious stimuli which are detected by nerve endings of nociceptive neurons. Pain: Types and Pathways and fever Fever Fever is defined as a measured body temperature of at least 38°C (100.4°F). Fever is caused by circulating endogenous and/or exogenous pyrogens that increase levels of prostaglandin E2 in the hypothalamus. Fever is commonly associated with chills, rigors, sweating, and flushing of the skin. Fever.
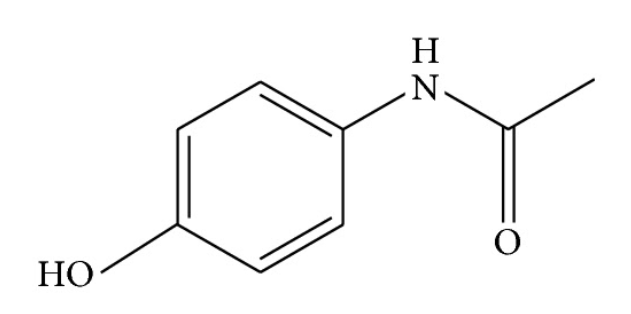
Acetaminophen (N-acetyl-p-aminophenol)
Image: “Acetaminophen (N-acetyl-p-aminophenol)” by Huang. License: CC BY 3.0Acetaminophen is an equivalent of aspirin Aspirin The prototypical analgesic used in the treatment of mild to moderate pain. It has anti-inflammatory and antipyretic properties and acts as an inhibitor of cyclooxygenase which results in the inhibition of the biosynthesis of prostaglandins. Aspirin also inhibits platelet aggregation and is used in the prevention of arterial and venous thrombosis. Nonsteroidal Antiinflammatory Drugs (NSAIDs) and has analgesic and antipyretic effects; however, acetaminophen does not have platelet-inhibiting effects nor does it affect uric acid Uric acid An oxidation product, via xanthine oxidase, of oxypurines such as xanthine and hypoxanthine. It is the final oxidation product of purine catabolism in humans and primates, whereas in most other mammals urate oxidase further oxidizes it to allantoin. Nephrolithiasis levels.
| Drugs | Interactions/effects |
|---|---|
| Alcohol (ethyl) | ↑ Hepatotoxicity |
| Barbiturates Barbiturates A class of chemicals derived from barbituric acid or thiobarbituric acid. Many of these are gaba modulators used as hypnotics and sedatives, as anesthetics, or as anticonvulsants. Intravenous Anesthetics |
|
| Carbamazepine Carbamazepine A dibenzazepine that acts as a sodium channel blocker. It is used as an anticonvulsant for the treatment of grand mal and psychomotor or focal seizures. It may also be used in the management of bipolar disorder, and has analgesic properties. First-Generation Anticonvulsant Drugs |
|
| Fosphenytoin-phenytoin |
|
| Isoniazid Isoniazid Antibacterial agent used primarily as a tuberculostatic. It remains the treatment of choice for tuberculosis. Antimycobacterial Drugs | ↑ Hepatotoxicity |
| Lamotrigine Lamotrigine A phenyltriazine compound, sodium and calcium channel blocker that is used for the treatment of seizures and bipolar disorder. Second-Generation Anticonvulsant Drugs | ↓ Serum concentration of lamotrigine Lamotrigine A phenyltriazine compound, sodium and calcium channel blocker that is used for the treatment of seizures and bipolar disorder. Second-Generation Anticonvulsant Drugs |
| Local anesthetics Anesthetics Agents that are capable of inducing a total or partial loss of sensation, especially tactile sensation and pain. They may act to induce general anesthesia, in which an unconscious state is achieved, or may act locally to induce numbness or lack of sensation at a targeted site. Anesthesiology: History and Basic Concepts | ↑ Risk of methemoglobinemia Methemoglobinemia Methemoglobinemia is a condition characterized by elevated levels of methemoglobin in the blood. Methemoglobin is the oxidized form of hemoglobin, where the heme iron has been converted from the usual ferrous (Fe2+) to the ferric (Fe3+) form. The Fe3+ form of iron cannot bind O2, and, thus, leads to tissue hypoxia. Methemoglobinemia |
| Prilocaine Prilocaine A local anesthetic that is similar pharmacologically to lidocaine. Currently, it is used most often for infiltration anesthesia in dentistry. Local Anesthetics | ↑ Likelihood of methemoglobinemia Methemoglobinemia Methemoglobinemia is a condition characterized by elevated levels of methemoglobin in the blood. Methemoglobin is the oxidized form of hemoglobin, where the heme iron has been converted from the usual ferrous (Fe2+) to the ferric (Fe3+) form. The Fe3+ form of iron cannot bind O2, and, thus, leads to tissue hypoxia. Methemoglobinemia |
| Rifampin Rifampin A semisynthetic antibiotic produced from streptomyces mediterranei. It has a broad antibacterial spectrum, including activity against several forms of Mycobacterium. In susceptible organisms it inhibits dna-dependent RNA polymerase activity by forming a stable complex with the enzyme. It thus suppresses the initiation of RNA synthesis. Rifampin is bactericidal, and acts on both intracellular and extracellular organisms. Epiglottitis | ↑ Hepatotoxicity of acetaminophen |
| Drug class | Acetaminophen | NSAIDs NSAIDS Primary vs Secondary Headaches |
|---|---|---|
| Mechanism of action | Indirect inhibition of COX | Inhibition of COX |
| Physiologic effect | Decreased inflammatory response | Decreased inflammatory response |
| Indications |
|
|
| Contraindications Contraindications A condition or factor associated with a recipient that makes the use of a drug, procedure, or physical agent improper or inadvisable. Contraindications may be absolute (life threatening) or relative (higher risk of complications in which benefits may outweigh risks). Noninvasive Ventilation |
|
|