Ethanol is a chemical compound that is produced in small amounts within the small intestine Small intestine The small intestine is the longest part of the GI tract, extending from the pyloric orifice of the stomach to the ileocecal junction. The small intestine is the major organ responsible for chemical digestion and absorption of nutrients. It is divided into 3 segments: the duodenum, the jejunum, and the ileum. Small Intestine: Anatomy and is also ingested from alcoholic Alcoholic Persons who have a history of physical or psychological dependence on ethanol. Mallory-Weiss Syndrome (Mallory-Weiss Tear) drinks. Ethanol's digestion Digestion Digestion refers to the process of the mechanical and chemical breakdown of food into smaller particles, which can then be absorbed and utilized by the body. Digestion and Absorption involves a complex catabolic pathway that mainly takes place in the liver Liver The liver is the largest gland in the human body. The liver is found in the superior right quadrant of the abdomen and weighs approximately 1.5 kilograms. Its main functions are detoxification, metabolism, nutrient storage (e.g., iron and vitamins), synthesis of coagulation factors, formation of bile, filtration, and storage of blood. Liver: Anatomy. Ethanol is turned into acetaldehyde, then to acetate, and finally into acetyl-CoA Acetyl-CoA Acetyl CoA participates in the biosynthesis of fatty acids and sterols, in the oxidation of fatty acids and in the metabolism of many amino acids. It also acts as a biological acetylating agent. Citric Acid Cycle, which becomes a substrate Substrate A substance upon which the enzyme acts. Basics of Enzymes for the citric acid cycle Citric acid cycle The citric acid cycle, also known as the tricarboxylic acid (TCA) cycle or the Krebs cycle, is a cyclic set of reactions that occurs in the mitochondrial matrix. The TCA cycle is the continuation of any metabolic pathway that produces pyruvate, which is converted into its main substrate, acetyl-CoA. Citric Acid Cycle and produces energy. Excessive ethanol intake can have pathologic metabolic consequences including alcoholism Alcoholism A primary, chronic disease with genetic, psychosocial, and environmental factors influencing its development and manifestations. The disease is often progressive and fatal. It is characterized by impaired control over drinking, preoccupation with the drug alcohol, use of alcohol despite adverse consequences, and distortions in thinking, most notably denial. Each of these symptoms may be continuous or periodic. Wernicke Encephalopathy and Korsakoff Syndrome, liver Liver The liver is the largest gland in the human body. The liver is found in the superior right quadrant of the abdomen and weighs approximately 1.5 kilograms. Its main functions are detoxification, metabolism, nutrient storage (e.g., iron and vitamins), synthesis of coagulation factors, formation of bile, filtration, and storage of blood. Liver: Anatomy disease, and cancer.
Last updated: Apr 17, 2025
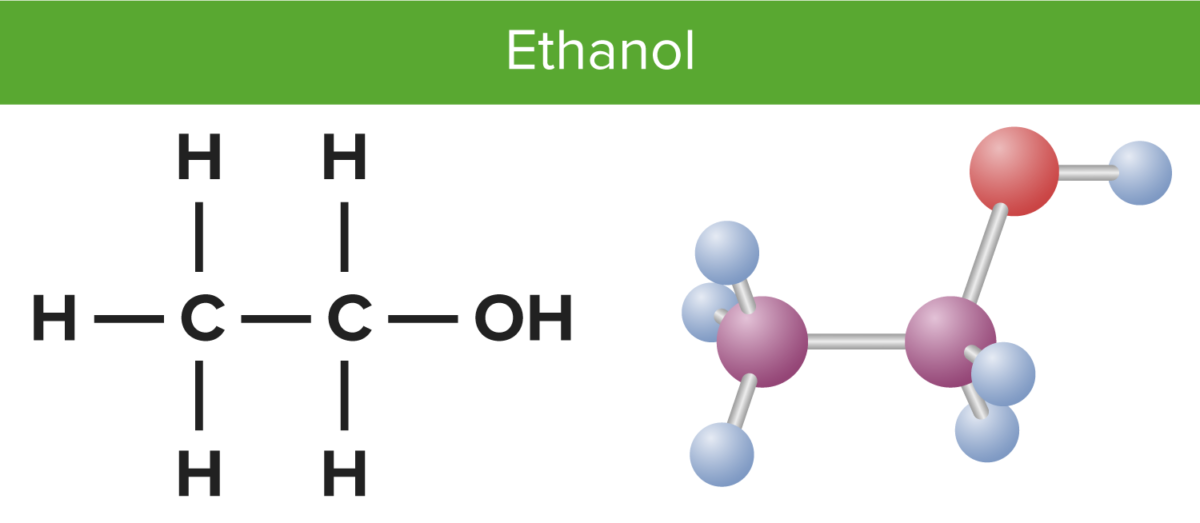
Structure of the ethanol molecule
Image by Lecturio.The primary site of alcohol catabolism is the liver Liver The liver is the largest gland in the human body. The liver is found in the superior right quadrant of the abdomen and weighs approximately 1.5 kilograms. Its main functions are detoxification, metabolism, nutrient storage (e.g., iron and vitamins), synthesis of coagulation factors, formation of bile, filtration, and storage of blood. Liver: Anatomy.
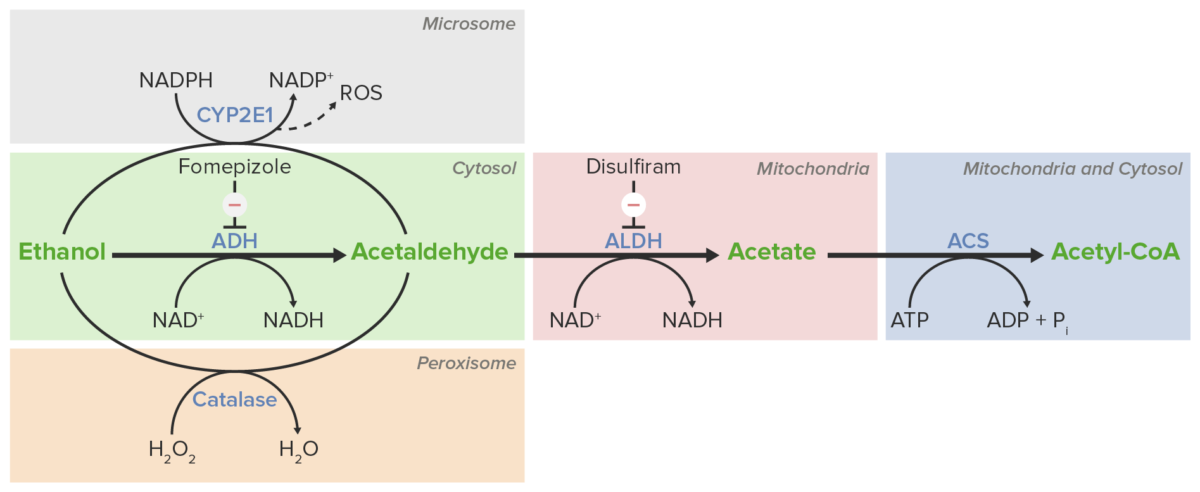
Schematic diagram of the steps of ethanol metabolism
Image by Lecturio.Many common medications can inhibit enzymes Enzymes Enzymes are complex protein biocatalysts that accelerate chemical reactions without being consumed by them. Due to the body’s constant metabolic needs, the absence of enzymes would make life unsustainable, as reactions would occur too slowly without these molecules. Basics of Enzymes involved in the metabolism of ethanol, leading to the accumulation of toxic products (e.g., acetaldehyde):
| Medication | Inhibited enzyme | Effects |
|---|---|---|
| Fomepizole Fomepizole A pyrazole and competitive inhibitor of alcohol dehydrogenase that is used for the treatment of poisoning by ethylene glycol or methanol. Antidotes of Common Poisonings | Alcohol dehydrogenase (ADH) |
|
| Disulfiram | Acetaldehyde dehydrogenase (ALDH) |
|
|
|
Excessive ethanol consumption, such as in alcohol use disorder Alcohol use disorder Alcohol is one of the most commonly used addictive substances in the world. Alcohol use disorder (AUD) is defined as pathologic consumption of alcohol leading to impaired daily functioning. Acute alcohol intoxication presents with impairment in speech and motor functions and can be managed in most cases with supportive care. Alcohol Use Disorder, leads to saturation of the ethanol metabolism pathway and consequent accumulation of toxic metabolites, as well as alteration of other metabolic pathways.
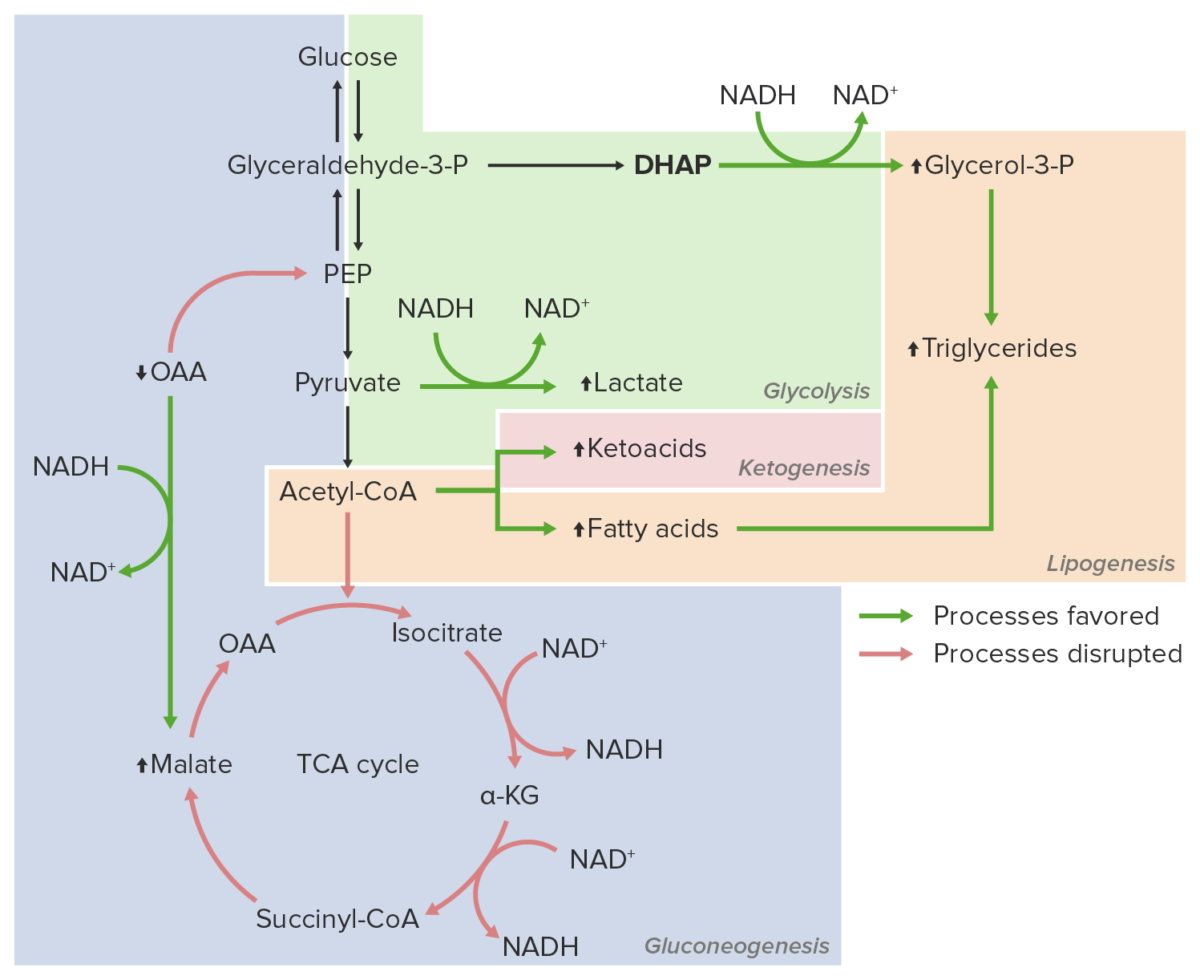
Excessive ethanol consumption
Image by Lecturio.The following conditions are associated with excessive ethanol consumption: