Although fresh whole blood was the only product available in the early years of transfusion, the advent of whole-blood fractionation techniques has allowed for more efficient use of the various blood components. Fractionated transfusion products, prepared in blood transfusion centers, include RBCs RBCs Erythrocytes, or red blood cells (RBCs), are the most abundant cells in the blood. While erythrocytes in the fetus are initially produced in the yolk sac then the liver, the bone marrow eventually becomes the main site of production. Erythrocytes: Histology, platelets Platelets Platelets are small cell fragments involved in hemostasis. Thrombopoiesis takes place primarily in the bone marrow through a series of cell differentiation and is influenced by several cytokines. Platelets are formed after fragmentation of the megakaryocyte cytoplasm. Platelets: Histology, FFP, and cryoprecipitate. These products are transfused for different indications and each addresses different pathologies.
Last updated: Jun 9, 2025
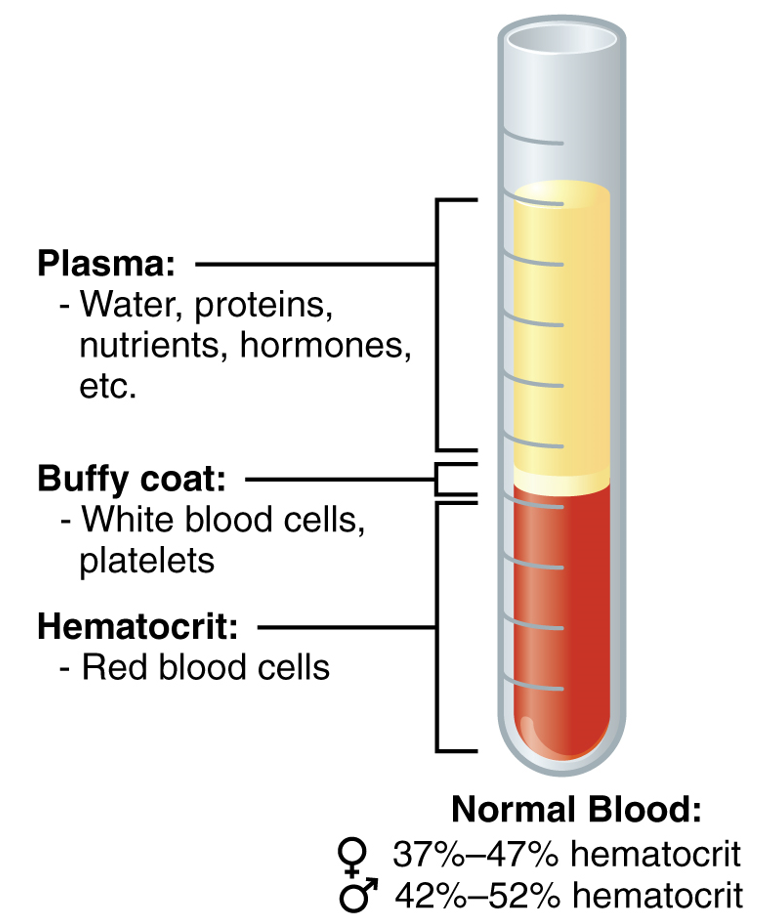
A centrifuged tube showing the components in whole blood (plasma, RBCs, platelets, and WBCs)
Image: “The cellular elements of blood include a vast number of erythrocytes and comparatively fewer leukocytes and platelets” by OpenStax College. License: CC BY 4.0| Blood type | Can give to individuals with blood type: | Can receive from donors with blood type: |
|---|---|---|
| A+ | A+, AB+ | A+, A–, O+, O |
| A– | A+, A-, AB+, AB– | A-, O– |
| B+ | B+, AB+ | B+, B–, O+, O– |
| B- | B+, B–, AB+, AB– | B–, O– |
| O+ | A+, B+, O+, AB+ | O+, O– |
| O– | Everyone | O– |
| AB+ | AB+ | Everyone |
| AB– | AB+, AB– | AB–, A–, B–, O– |
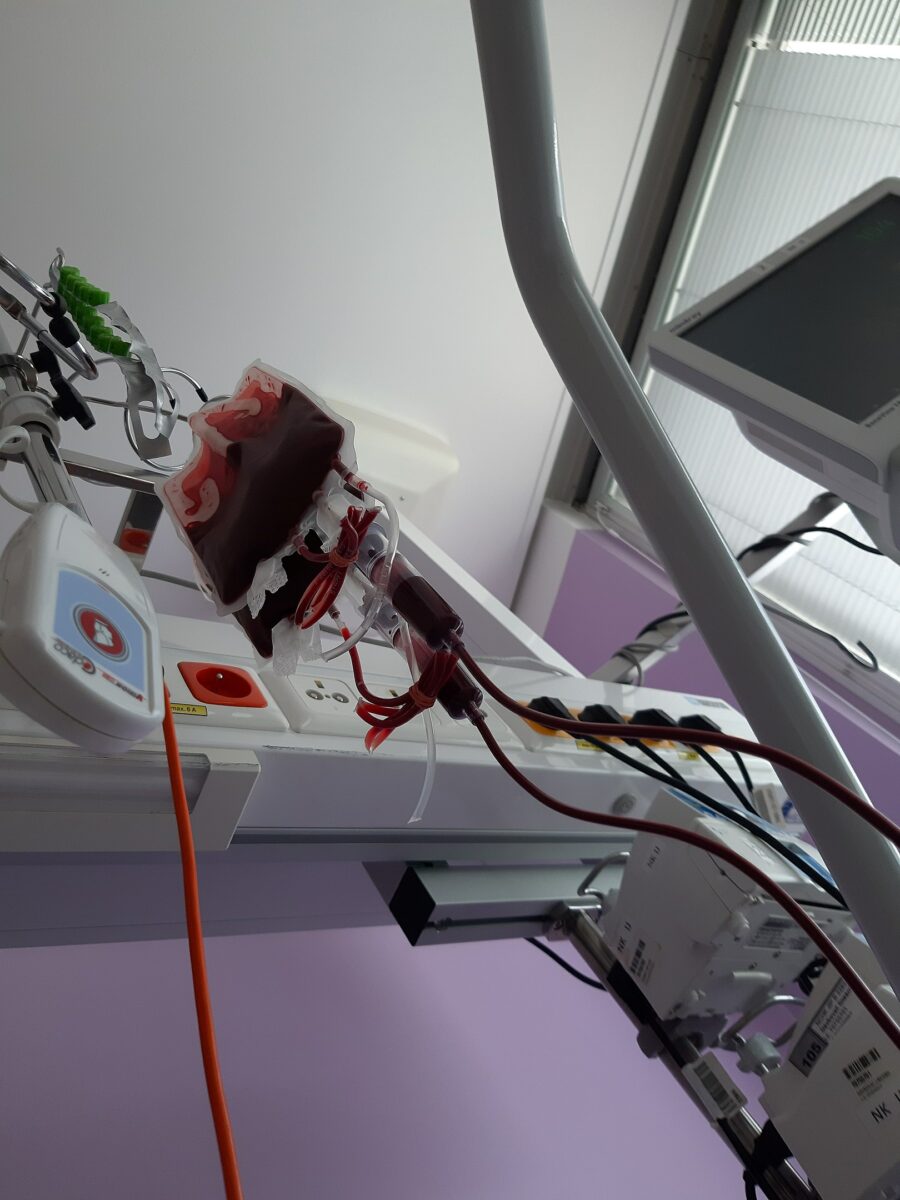
Picture of the blood collection process:
Collection bag hangs as blood is collected from a donor
| Blood components | Subcomponents of the blood | Type | Production site | Main tasks |
|---|---|---|---|---|
| Plasma 43%–63% | Water 92% | Liquid | Absorbed in GI tract or made during metabolism | Transport medium |
| Plasma proteins Proteins Linear polypeptides that are synthesized on ribosomes and may be further modified, crosslinked, cleaved, or assembled into complex proteins with several subunits. The specific sequence of amino acids determines the shape the polypeptide will take, during protein folding, and the function of the protein. Energy Homeostasis 7% | Albumin Albumin Serum albumin from humans. It is an essential carrier of both endogenous substances, such as fatty acids and bilirubin, and of xenobiotics in the blood. Liver Function Tests 54%–60% | Liver Liver The liver is the largest gland in the human body. The liver is found in the superior right quadrant of the abdomen and weighs approximately 1.5 kilograms. Its main functions are detoxification, metabolism, nutrient storage (e.g., iron and vitamins), synthesis of coagulation factors, formation of bile, filtration, and storage of blood. Liver: Anatomy |
|
|
| Globulins 35%–38% | Alpha globulins: liver Liver The liver is the largest gland in the human body. The liver is found in the superior right quadrant of the abdomen and weighs approximately 1.5 kilograms. Its main functions are detoxification, metabolism, nutrient storage (e.g., iron and vitamins), synthesis of coagulation factors, formation of bile, filtration, and storage of blood. Liver: Anatomy | Transport and maintain osmotic concentration | ||
| Beta globulins: liver Liver The liver is the largest gland in the human body. The liver is found in the superior right quadrant of the abdomen and weighs approximately 1.5 kilograms. Its main functions are detoxification, metabolism, nutrient storage (e.g., iron and vitamins), synthesis of coagulation factors, formation of bile, filtration, and storage of blood. Liver: Anatomy | Transport and maintain osmotic concentration | |||
| Gamma globulins ( immunoglobulins Immunoglobulins Immunoglobulins (Igs), also known as antibodies, are glycoprotein molecules produced by plasma cells that act in immune responses by recognizing and binding particular antigens. The various Ig classes are IgG (the most abundant), IgM, IgE, IgD, and IgA, which differ in their biologic features, structure, target specificity, and distribution. Immunoglobulins: Types and Functions): plasma cells Plasma cells Specialized forms of antibody-producing B-lymphocytes. They synthesize and secrete immunoglobulin. They are found only in lymphoid organs and at sites of immune responses and normally do not circulate in the blood or lymph. Humoral Adaptive Immunity | Immune response | |||
| Fibrinogen Fibrinogen Plasma glycoprotein clotted by thrombin, composed of a dimer of three non-identical pairs of polypeptide chains (alpha, beta, gamma) held together by disulfide bonds. Fibrinogen clotting is a sol-gel change involving complex molecular arrangements: whereas fibrinogen is cleaved by thrombin to form polypeptides a and b, the proteolytic action of other enzymes yields different fibrinogen degradation products. Hemostasis 4%–7% | Liver Liver The liver is the largest gland in the human body. The liver is found in the superior right quadrant of the abdomen and weighs approximately 1.5 kilograms. Its main functions are detoxification, metabolism, nutrient storage (e.g., iron and vitamins), synthesis of coagulation factors, formation of bile, filtration, and storage of blood. Liver: Anatomy | Blood clotting during hemostasis Hemostasis Hemostasis refers to the innate, stepwise body processes that occur following vessel injury, resulting in clot formation and cessation of bleeding. Hemostasis occurs in 2 phases, namely, primary and secondary. Primary hemostasis involves forming a plug that stops the bleeding temporarily. Secondary hemostasis involves the activation of the coagulation cascade. Hemostasis | ||
| Regulatory proteins Regulatory proteins Proteins and Peptides < 1% |
|
Various locations | Regulate various body functions | |
| Other dissolved substances 1% |
|
|
Many different functions | |
| Formed elements 37%–54% | Erythrocytes Erythrocytes Erythrocytes, or red blood cells (RBCs), are the most abundant cells in the blood. While erythrocytes in the fetus are initially produced in the yolk sac then the liver, the bone marrow eventually becomes the main site of production. Erythrocytes: Histology 99% | Erythrocytes Erythrocytes Erythrocytes, or red blood cells (RBCs), are the most abundant cells in the blood. While erythrocytes in the fetus are initially produced in the yolk sac then the liver, the bone marrow eventually becomes the main site of production. Erythrocytes: Histology | Red marrow | Transports gasses, O2, and some CO2 |
|
|
Red marrow | Nonspecific immune response | |
|
Lymphocytes Lymphocytes Lymphocytes are heterogeneous WBCs involved in immune response. Lymphocytes develop from the bone marrow, starting from hematopoietic stem cells (HSCs) and progressing to common lymphoid progenitors (CLPs). B and T lymphocytes and natural killer (NK) cells arise from the lineage. Lymphocytes: Histology: bone marrow Bone marrow The soft tissue filling the cavities of bones. Bone marrow exists in two types, yellow and red. Yellow marrow is found in the large cavities of large bones and consists mostly of fat cells and a few primitive blood cells. Red marrow is a hematopoietic tissue and is the site of production of erythrocytes and granular leukocytes. Bone marrow is made up of a framework of connective tissue containing branching fibers with the frame being filled with marrow cells. Bone Marrow: Composition and Hematopoiesis and lymphoid tissue | Lymphocytes Lymphocytes Lymphocytes are heterogeneous WBCs involved in immune response. Lymphocytes develop from the bone marrow, starting from hematopoietic stem cells (HSCs) and progressing to common lymphoid progenitors (CLPs). B and T lymphocytes and natural killer (NK) cells arise from the lineage. Lymphocytes: Histology: specific immune response | ||
| Monocytes Monocytes Large, phagocytic mononuclear leukocytes produced in the vertebrate bone marrow and released into the blood; contain a large, oval or somewhat indented nucleus surrounded by voluminous cytoplasm and numerous organelles. Innate Immunity: Phagocytes and Antigen Presentation: red marrow | Monocytes Monocytes Large, phagocytic mononuclear leukocytes produced in the vertebrate bone marrow and released into the blood; contain a large, oval or somewhat indented nucleus surrounded by voluminous cytoplasm and numerous organelles. Innate Immunity: Phagocytes and Antigen Presentation: nonspecific immune response | |||
| Platelets Platelets Platelets are small cell fragments involved in hemostasis. Thrombopoiesis takes place primarily in the bone marrow through a series of cell differentiation and is influenced by several cytokines. Platelets are formed after fragmentation of the megakaryocyte cytoplasm. Platelets: Histology < 1% | — | Megakaryocytes: red marrow | Hemostasis Hemostasis Hemostasis refers to the innate, stepwise body processes that occur following vessel injury, resulting in clot formation and cessation of bleeding. Hemostasis occurs in 2 phases, namely, primary and secondary. Primary hemostasis involves forming a plug that stops the bleeding temporarily. Secondary hemostasis involves the activation of the coagulation cascade. Hemostasis |
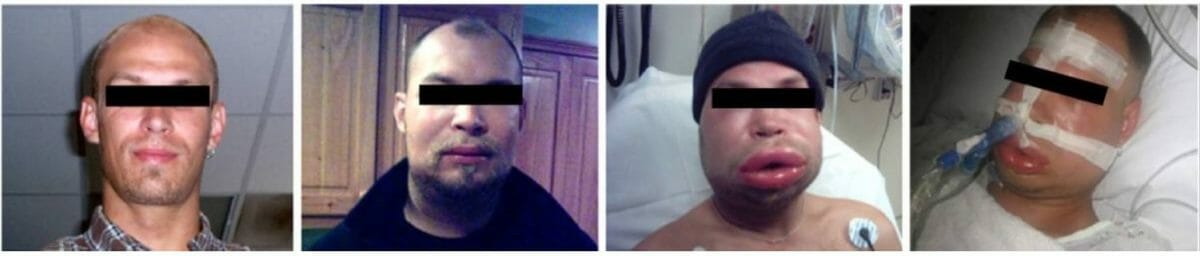
An individual experiencing acute attacks of hereditary angioedema
Image: “F1: HAE patient experiencing HAE attacks” by Bygum A. et al. License: CC BY 2.0