Bone Bone Bone is a compact type of hardened connective tissue composed of bone cells, membranes, an extracellular mineralized matrix, and central bone marrow. The 2 primary types of bone are compact and spongy. Bones: Structure and Types marrow, the primary site of hematopoiesis, is found in the cavities of cancellous bones and the medullary canals of long bones Long bones Length greater than width. Bones: Structure and Types. There are 2 types: red marrow (hematopoietic with abundant blood cells) and yellow marrow (predominantly filled with adipocytes Adipocytes Cells in the body that store fats, usually in the form of triglycerides. White adipocytes are the predominant type and found mostly in the abdominal cavity and subcutaneous tissue. Brown adipocytes are thermogenic cells that can be found in newborns of some species and hibernating mammals. Adipose Tissue: Histology). Human marrow composition changes with age. In the young, the entire bone Bone Bone is a compact type of hardened connective tissue composed of bone cells, membranes, an extracellular mineralized matrix, and central bone marrow. The 2 primary types of bone are compact and spongy. Bones: Structure and Types marrow is red, as there is increased blood cell production. As age increases, there is gradual change to the yellow marrow variety. The yellow marrow can revert to red marrow when hematopoiesis is needed (e.g., anemia Anemia Anemia is a condition in which individuals have low Hb levels, which can arise from various causes. Anemia is accompanied by a reduced number of RBCs and may manifest with fatigue, shortness of breath, pallor, and weakness. Subtypes are classified by the size of RBCs, chronicity, and etiology. Anemia: Overview and Types). The red marrow, by hematopoiesis, produces about 6 billion cells per kilogram per day. The process relies on hematopoietic cells (stem cells and progenitors) producing mature effector cells Effector cells Cells have been activated by a matching antigen Adaptive Immune Response ( lymphocytes Lymphocytes Lymphocytes are heterogeneous WBCs involved in immune response. Lymphocytes develop from the bone marrow, starting from hematopoietic stem cells (HSCs) and progressing to common lymphoid progenitors (CLPs). B and T lymphocytes and natural killer (NK) cells arise from the lineage. Lymphocytes: Histology, platelets Platelets Platelets are small cell fragments involved in hemostasis. Thrombopoiesis takes place primarily in the bone marrow through a series of cell differentiation and is influenced by several cytokines. Platelets are formed after fragmentation of the megakaryocyte cytoplasm. Platelets: Histology, granulocytes Granulocytes Leukocytes with abundant granules in the cytoplasm. They are divided into three groups according to the staining properties of the granules: neutrophilic, eosinophilic, and basophilic. Mature granulocytes are the neutrophils; eosinophils; and basophils. White Myeloid Cells: Histology, erythrocytes Erythrocytes Erythrocytes, or red blood cells (RBCs), are the most abundant cells in the blood. While erythrocytes in the fetus are initially produced in the yolk sac then the liver, the bone marrow eventually becomes the main site of production. Erythrocytes: Histology) with the aid of nonhematopoietic elements. Production is regulated by cytokines Cytokines Non-antibody proteins secreted by inflammatory leukocytes and some non-leukocytic cells, that act as intercellular mediators. They differ from classical hormones in that they are produced by a number of tissue or cell types rather than by specialized glands. They generally act locally in a paracrine or autocrine rather than endocrine manner. Adaptive Immune Response released in the bone Bone Bone is a compact type of hardened connective tissue composed of bone cells, membranes, an extracellular mineralized matrix, and central bone marrow. The 2 primary types of bone are compact and spongy. Bones: Structure and Types marrow environment and feedback from target tissues. The marrow structure allows hematopoiesis to take place in the extravascular area, and after staged differentiation, blood cells are released into circulation Circulation The movement of the blood as it is pumped through the cardiovascular system. ABCDE Assessment.
Last updated: Mar 27, 2025
Bone Bone Bone is a compact type of hardened connective tissue composed of bone cells, membranes, an extracellular mineralized matrix, and central bone marrow. The 2 primary types of bone are compact and spongy. Bones: Structure and Types marrow is the spongy tissue found in the medullary canals of long bones Long bones Length greater than width. Bones: Structure and Types and the cavities of cancellous bones.
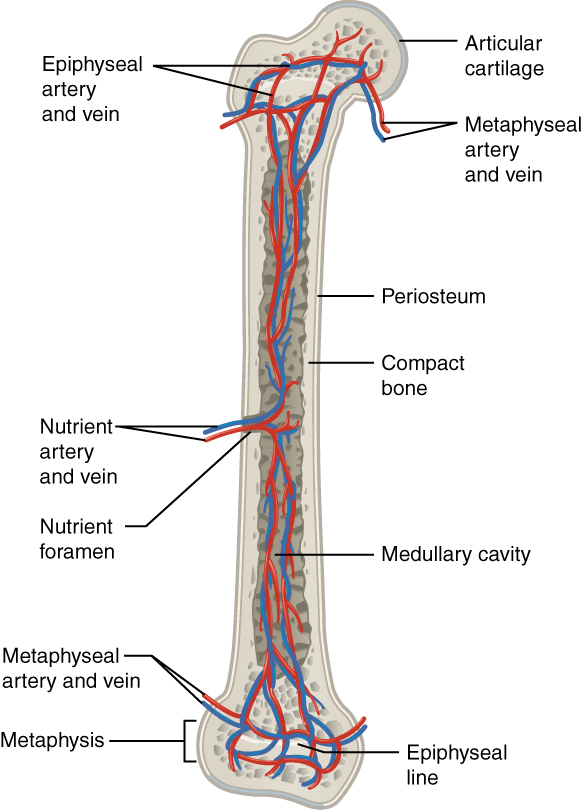
Image shows the blood supply (artery and vein)
Image: “609 Body Supply to the Bone” by OpenStax College. License: CC BY 3.0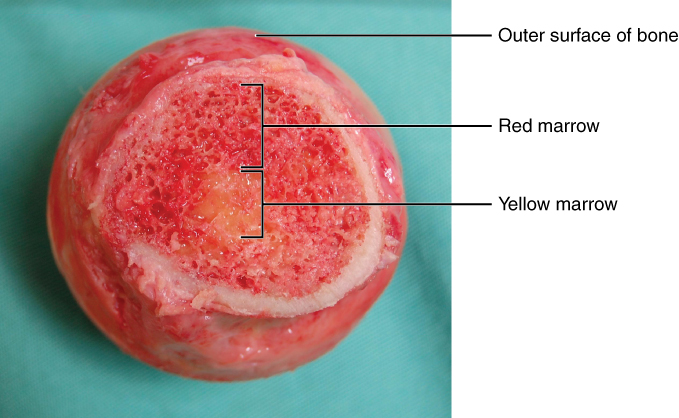
Bone marrow:
Cross-section showing the red and yellow marrow portions of the bone
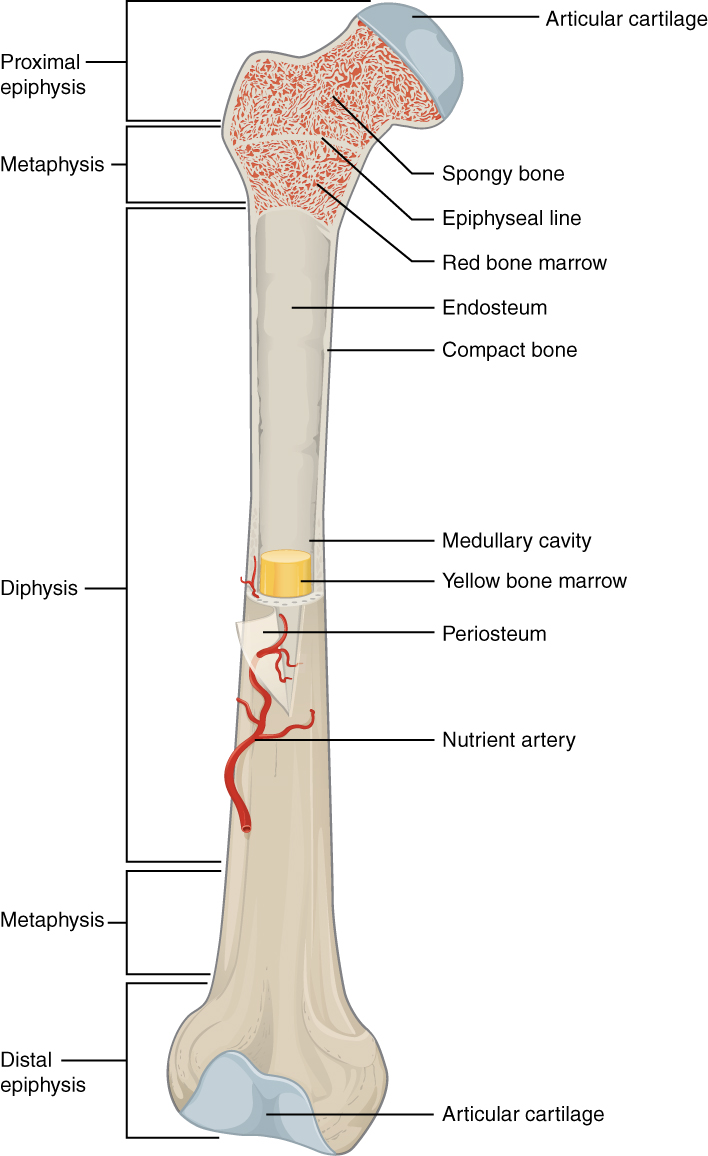
Bone marrow inside the femur
Image: “603 Anatomy of Long Bone” by OpenStax College. License: CC BY 3.0Structures that provide a microenvironment that supports the differentiation of hematopoietic cells and proliferation of blood cells:
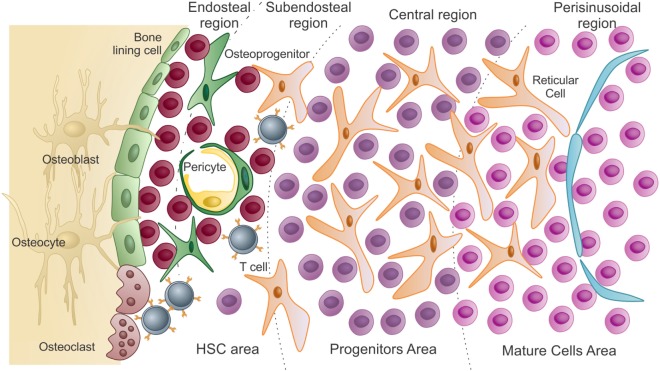
Schematic presentation of the bone marrow microenvironment:
In the hematopoietic stem cell (HSC) area are the hematopoietic stem cells and uncommitted progenitors. These are in close association with endosteal osteoblasts and bone-lining cells. As HSCs exit quiescence to proliferative states, they migrate and colonize the subendosteal region, then the central region of the marrow (progenitors).
Differentiated cells reach the mature area, closer to the sinusoids. To be released into circulation, these mature cells pass through the endothelium (lining the sinusoids) by transcellular migration.
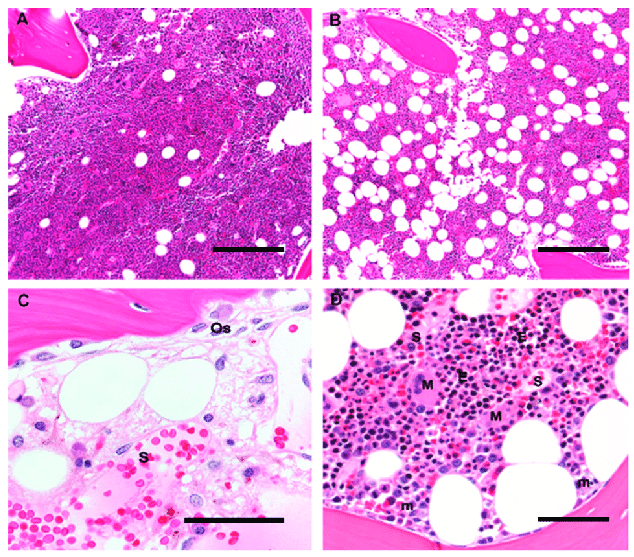
Human bone marrow histological architecture: The images show the different cells and structures that are in the marrow.
A and B: Original magnification 10×; scale bar 100 μm
A: A bone marrow biopsy from a 5-year-old child is > 90% cellular, with a predominance of trilineage hematopoiesis and little admixed adipose tissue.
B: A bone marrow biopsy from a 60-year-old adult is composed of 50% hematopoietic elements and 50% admixed mature adipose tissue.
C: A post-chemotherapy marrow shows trabecular bone with apposed osteoblasts (Os), and a thin osteoid seam of unmineralized collagen. Sinusoids (S) are filled with RBCs and have stromal cells with ovoid nuclei. Scattered mononuclear cells include plasma cells, mast cells, and macrophages; original magnification 60×; scale bar 25 μm
D: Erythroid colonies (E) appear as colonies of round cells with dark nuclei and are located away from trabecular bone, close to thin-walled sinusoidal vessels (S); megakaryocytes (M) are likewise located in close contact with sinusoids, whereas immature myeloid precursors (m) are localized near trabecular bone. Original magnification 20×; scale bar 50 μm
Hematopoiesis starts with an HSC, which is prompted to divide and differentiate with appropriate chemical stimuli (hemopoietic growth factors).
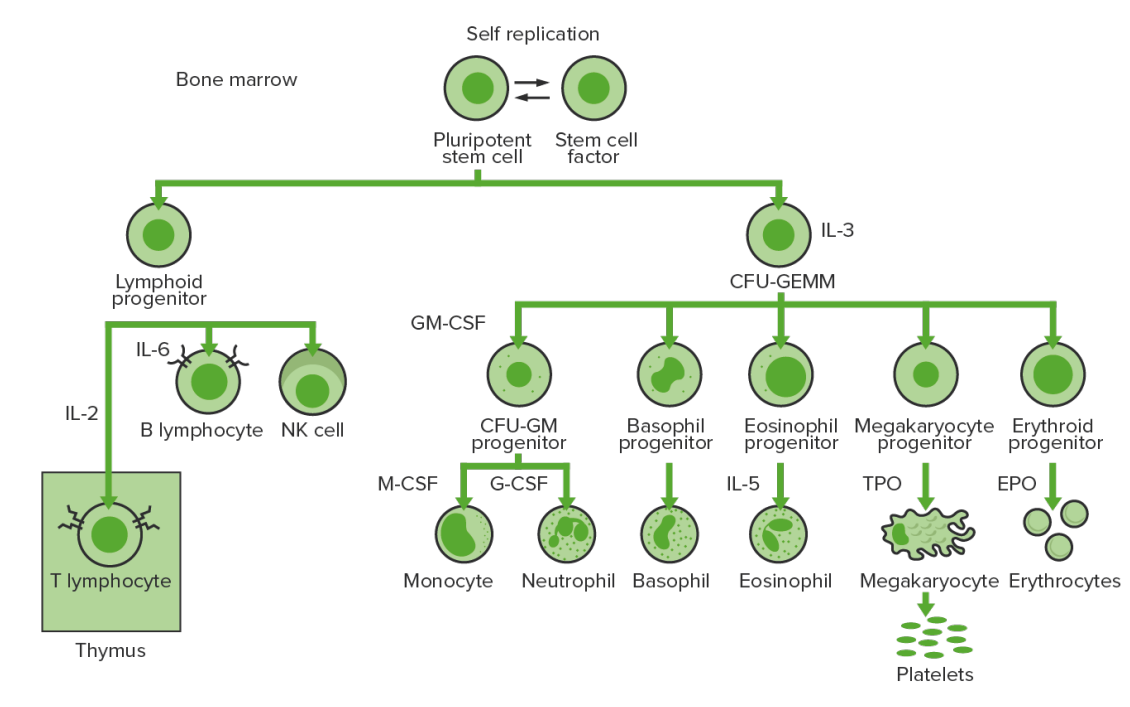
Bone marrow hematopoiesis: proliferation and differentiation of the formed elements of blood
CFU-GEMM: colony-forming unit: granulocyte, erythrocyte, monocyte, megakaryocyte
CFU-GM: colony-forming unit: granulocyte-macrophage
GM-CSF: granulocyte-macrophage colony-stimulating factor
M-CSF: macrophage colony-stimulating factor
G-CSF: granulocyte colony-stimulating factor
NK: natural killer
TPO: thrombopoietin
| Cytokines Cytokines Non-antibody proteins secreted by inflammatory leukocytes and some non-leukocytic cells, that act as intercellular mediators. They differ from classical hormones in that they are produced by a number of tissue or cell types rather than by specialized glands. They generally act locally in a paracrine or autocrine rather than endocrine manner. Adaptive Immune Response/growth factors | Activities | Source |
|---|---|---|
| Erythropoietin Erythropoietin Glycoprotein hormone, secreted chiefly by the kidney in the adult and the liver in the fetus, that acts on erythroid stem cells of the bone marrow to stimulate proliferation and differentiation. Erythrocytes: Histology ( EPO EPO Glycoprotein hormone, secreted chiefly by the kidney in the adult and the liver in the fetus, that acts on erythroid stem cells of the bone marrow to stimulate proliferation and differentiation. Erythrocytes: Histology) | Stimulates erythropoiesis Erythropoiesis The production of red blood cells (erythrocytes). In humans, erythrocytes are produced by the yolk sac in the first trimester; by the liver in the second trimester; by the bone marrow in the third trimester and after birth. In normal individuals, the erythrocyte count in the peripheral blood remains relatively constant implying a balance between the rate of erythrocyte production and rate of destruction. Erythrocytes: Histology, including differentiation |
|
| Thrombopoietin Thrombopoietin A humoral factor that stimulates the production of thrombocytes (blood platelets). Thrombopoietin stimulates the proliferation of bone marrow megakaryocytes and their release of blood platelets. The process is called thrombopoiesis. Platelets: Histology (TPO) | Stimulates thrombopoiesis Thrombopoiesis The process of generating thrombocytes (blood platelets) from the pluripotent hematopoietic stem cells in the bone marrow via the megakaryocytes. The humoral factor with thrombopoiesis-stimulating activity is designated thrombopoietin. Platelets: Histology |
|
| Stem cell factor (SCF) | Stimulates all hematopoietic progenitor cells | Bone Bone Bone is a compact type of hardened connective tissue composed of bone cells, membranes, an extracellular mineralized matrix, and central bone marrow. The 2 primary types of bone are compact and spongy. Bones: Structure and Types marrow stromal cells |
| Granulocyte-macrophage colony-stimulating factor ( GM-CSF GM-CSF An acidic glycoprotein of mw 23 kda with internal disulfide bonds. The protein is produced in response to a number of inflammatory mediators by mesenchymal cells present in the hemopoietic environment and at peripheral sites of inflammation. GM-CSF is able to stimulate the production of neutrophilic granulocytes, macrophages, and mixed granulocyte-macrophage colonies from bone marrow cells and can stimulate the formation of eosinophil colonies from fetal liver progenitor cells. GM-CSF can also stimulate some functional activities in mature granulocytes and macrophages. White Myeloid Cells: Histology) | Stimulates myeloid progenitor cells Myeloid progenitor cells Stem cells derived from hematopoietic stem cells. Derived from these myeloid progenitor cells are the megakaryocytes; erythroid cells; myeloid cells; and some dendritic cells. Acute Myeloid Leukemia |
|
| Granulocyte colony-stimulating factor Granulocyte colony-stimulating factor A glycoprotein of mw 25 kda containing internal disulfide bonds. It induces the survival, proliferation, and differentiation of neutrophilic granulocyte precursor cells and functionally activates mature blood neutrophils. Among the family of colony-stimulating factors, G-CSF is the most potent inducer of terminal differentiation to granulocytes and macrophages of leukemic myeloid cell lines. White Myeloid Cells: Histology (G-CSF) | Stimulates neutrophil precursor cells |
|
| Monocyte colony-stimulating factor ( M-CSF M-CSF A mononuclear phagocyte colony-stimulating factor (M-CSF) synthesized by mesenchymal cells. The compound stimulates the survival, proliferation, and differentiation of hematopoietic cells of the monocyte-macrophage series. M-CSF is a disulfide-bonded glycoprotein dimer with a mw of 70 kda. It binds to a specific high affinity receptor. White Myeloid Cells: Histology) | Stimulates monocyte precursor cells |
|
| Interleukin (IL) | Activities | Source |
|---|---|---|
| IL-1 | Regulation of cytokine secretion Secretion Coagulation Studies of many leukocytes Leukocytes White blood cells. These include granular leukocytes (basophils; eosinophils; and neutrophils) as well as non-granular leukocytes (lymphocytes and monocytes). White Myeloid Cells: Histology |
|
| IL-2 |
|
T helper cells |
| IL-3 | Mitogen for all granulocyte and megakaryocyte Megakaryocyte Very large bone marrow cells which release mature blood platelets. Platelets: Histology/erythrocyte progenitor cells | T helper cells |
| IL-4 |
|
T helper cells |
| IL-5 | Development and activation of eosinophils Eosinophils Granular leukocytes with a nucleus that usually has two lobes connected by a slender thread of chromatin, and cytoplasm containing coarse, round granules that are uniform in size and stainable by eosin. Innate Immunity: Phagocytes and Antigen Presentation | T helper cells |
| IL-6 |
|
|
| IL-7 IL-7 A proinflammatory cytokine produced primarily by T-lymphocytes or their precursors. Several subtypes of interleukin-17 have been identified, each of which is a product of a unique gene. Severe Combined Immunodeficiency (SCID) | Stimulation of all lymphoid stem cells | Stromal cells of bone Bone Bone is a compact type of hardened connective tissue composed of bone cells, membranes, an extracellular mineralized matrix, and central bone marrow. The 2 primary types of bone are compact and spongy. Bones: Structure and Types marrow |
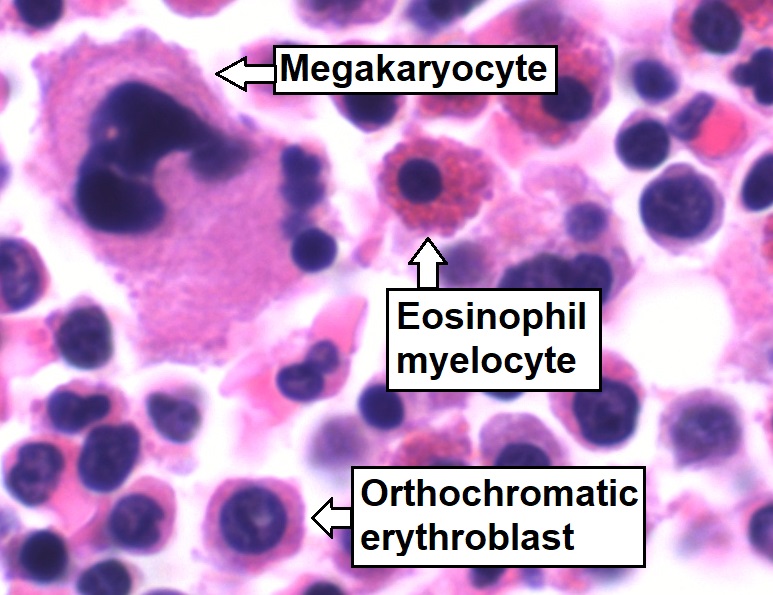
Bone marrow aspirate showing normal trilineage hematopoiesis: myelomonocytic cells (marked eosinophil myelocyte), erythroid cells (marked orthochromatic erythroblast), and megakaryocytic cells
Image: “Trilineage hematopoiesis” by Mikael Häggström. License: CC0 1.0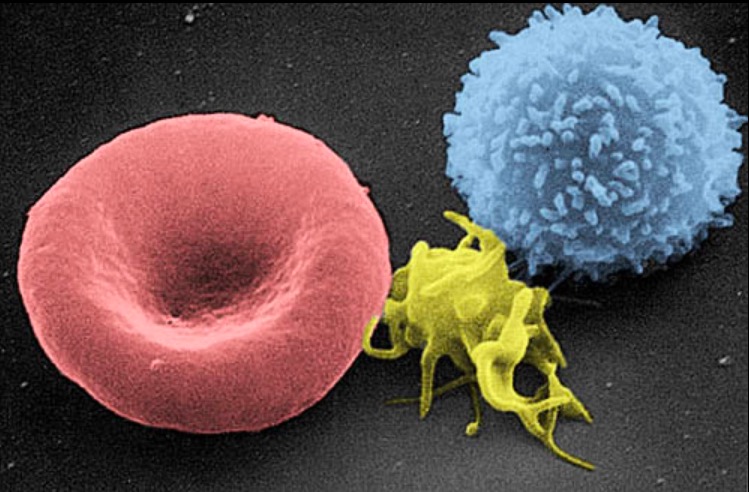
A scanning electron micrograph image of a blood cell: from left to right, a human red blood cell, a thrombocyte (platelet), and a leukocyte
Image: “Electron micrograph of blood cells” by Electron Microscopy Facility at The National Cancer Institute at Frederick. License: Public Domain