Diabetes mellitus (DM) is a metabolic disease characterized by hyperglycemia and dysfunction of the regulation of glucose Glucose A primary source of energy for living organisms. It is naturally occurring and is found in fruits and other parts of plants in its free state. It is used therapeutically in fluid and nutrient replacement. Lactose Intolerance metabolism by insulin Insulin Insulin is a peptide hormone that is produced by the beta cells of the pancreas. Insulin plays a role in metabolic functions such as glucose uptake, glycolysis, glycogenesis, lipogenesis, and protein synthesis. Exogenous insulin may be needed for individuals with diabetes mellitus, in whom there is a deficiency in endogenous insulin or increased insulin resistance. Insulin. Type 1 Type 1 Spinal Muscular Atrophy DM is diagnosed mostly in children and young adults as the result of autoimmune destruction of β cells in the pancreas Pancreas The pancreas lies mostly posterior to the stomach and extends across the posterior abdominal wall from the duodenum on the right to the spleen on the left. This organ has both exocrine and endocrine tissue. Pancreas: Anatomy and the resulting lack of insulin Insulin Insulin is a peptide hormone that is produced by the beta cells of the pancreas. Insulin plays a role in metabolic functions such as glucose uptake, glycolysis, glycogenesis, lipogenesis, and protein synthesis. Exogenous insulin may be needed for individuals with diabetes mellitus, in whom there is a deficiency in endogenous insulin or increased insulin resistance. Insulin. Type 2 DM has a significant association with obesity Obesity Obesity is a condition associated with excess body weight, specifically with the deposition of excessive adipose tissue. Obesity is considered a global epidemic. Major influences come from the western diet and sedentary lifestyles, but the exact mechanisms likely include a mixture of genetic and environmental factors. Obesity and is characterized by insulin Insulin Insulin is a peptide hormone that is produced by the beta cells of the pancreas. Insulin plays a role in metabolic functions such as glucose uptake, glycolysis, glycogenesis, lipogenesis, and protein synthesis. Exogenous insulin may be needed for individuals with diabetes mellitus, in whom there is a deficiency in endogenous insulin or increased insulin resistance. Insulin resistance Resistance Physiologically, the opposition to flow of air caused by the forces of friction. As a part of pulmonary function testing, it is the ratio of driving pressure to the rate of air flow. Ventilation: Mechanics of Breathing, as well as relative insulin Insulin Insulin is a peptide hormone that is produced by the beta cells of the pancreas. Insulin plays a role in metabolic functions such as glucose uptake, glycolysis, glycogenesis, lipogenesis, and protein synthesis. Exogenous insulin may be needed for individuals with diabetes mellitus, in whom there is a deficiency in endogenous insulin or increased insulin resistance. Insulin deficiency. Genetically determined causes of diabetes (e.g., maturity-onset diabetes of the young and late autoimmune diabetes in adults) are being increasingly recognized, but they make up a small portion of cases. There is no definitive cure for DM. The objective of management is the prevention of complications, which may include coronary artery Coronary Artery Truncus Arteriosus disease, CKD CKD Chronic kidney disease (CKD) is kidney impairment that lasts for ≥ 3 months, implying that it is irreversible. Hypertension and diabetes are the most common causes; however, there are a multitude of other etiologies. In the early to moderate stages, CKD is usually asymptomatic and is primarily diagnosed by laboratory abnormalities. Chronic Kidney Disease, retinopathy Retinopathy Degenerative changes to the retina due to hypertension. Alport Syndrome, and neuropathy Neuropathy Leprosy. Long-term monitoring and maintenance of optimal blood glucose Glucose A primary source of energy for living organisms. It is naturally occurring and is found in fruits and other parts of plants in its free state. It is used therapeutically in fluid and nutrient replacement. Lactose Intolerance levels are key to preventing complications. Treatment is specific to the type of diabetes, with glycemic control as the goal in all types; insulin Insulin Insulin is a peptide hormone that is produced by the beta cells of the pancreas. Insulin plays a role in metabolic functions such as glucose uptake, glycolysis, glycogenesis, lipogenesis, and protein synthesis. Exogenous insulin may be needed for individuals with diabetes mellitus, in whom there is a deficiency in endogenous insulin or increased insulin resistance. Insulin replacement is essential in type 1 Type 1 Spinal Muscular Atrophy, and a healthy diet, lifestyle changes, and medications are important in type 2.
Last updated: May 17, 2024
Diabetes mellitus (DM) is a disorder of carbohydrate metabolism. Diabetes mellitus usually occurs in genetically predisposed individuals and is characterized by inadequate production of insulin Insulin Insulin is a peptide hormone that is produced by the beta cells of the pancreas. Insulin plays a role in metabolic functions such as glucose uptake, glycolysis, glycogenesis, lipogenesis, and protein synthesis. Exogenous insulin may be needed for individuals with diabetes mellitus, in whom there is a deficiency in endogenous insulin or increased insulin resistance. Insulin or resistance Resistance Physiologically, the opposition to flow of air caused by the forces of friction. As a part of pulmonary function testing, it is the ratio of driving pressure to the rate of air flow. Ventilation: Mechanics of Breathing to insulin Insulin Insulin is a peptide hormone that is produced by the beta cells of the pancreas. Insulin plays a role in metabolic functions such as glucose uptake, glycolysis, glycogenesis, lipogenesis, and protein synthesis. Exogenous insulin may be needed for individuals with diabetes mellitus, in whom there is a deficiency in endogenous insulin or increased insulin resistance. Insulin’s action on the pancreas Pancreas The pancreas lies mostly posterior to the stomach and extends across the posterior abdominal wall from the duodenum on the right to the spleen on the left. This organ has both exocrine and endocrine tissue. Pancreas: Anatomy. These features result in hyperglycemia and the long-term pathologic sequelae of DM.
Risk factors:
Type 2 DM is a combination of defective insulin Insulin Insulin is a peptide hormone that is produced by the beta cells of the pancreas. Insulin plays a role in metabolic functions such as glucose uptake, glycolysis, glycogenesis, lipogenesis, and protein synthesis. Exogenous insulin may be needed for individuals with diabetes mellitus, in whom there is a deficiency in endogenous insulin or increased insulin resistance. Insulin secretion Secretion Coagulation Studies and decreased insulin Insulin Insulin is a peptide hormone that is produced by the beta cells of the pancreas. Insulin plays a role in metabolic functions such as glucose uptake, glycolysis, glycogenesis, lipogenesis, and protein synthesis. Exogenous insulin may be needed for individuals with diabetes mellitus, in whom there is a deficiency in endogenous insulin or increased insulin resistance. Insulin sensitivity.
There are several effects of chronic hyperglycemia:
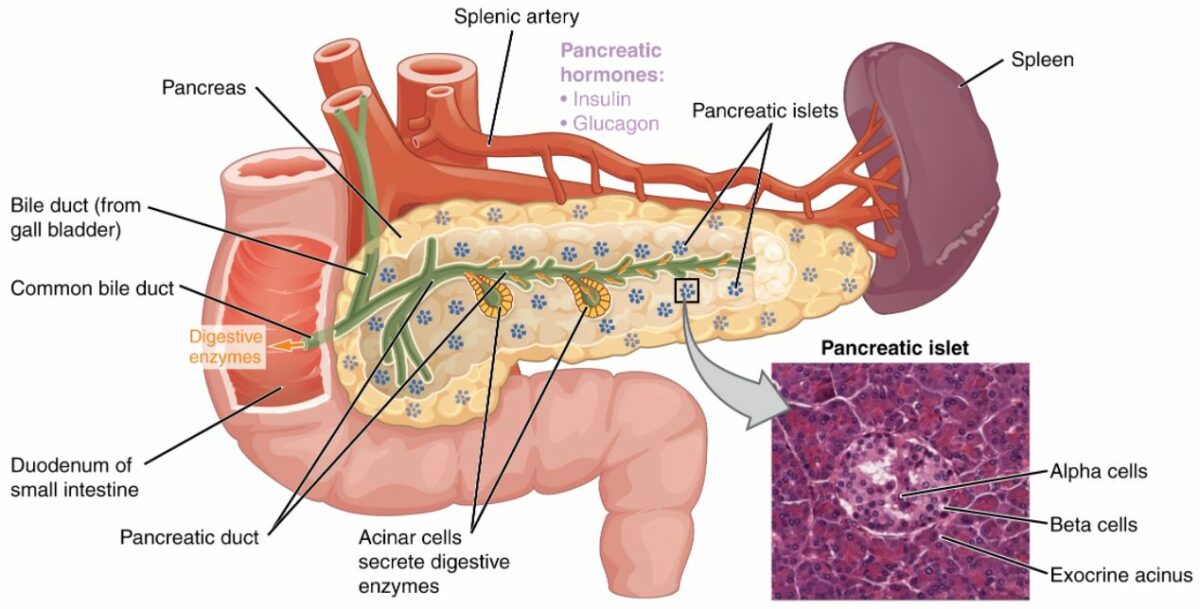
Pancreas:
Pancreatic exocrine function involves the acinar cells secreting digestive enzymes that are transported into the small intestine by the pancreatic duct. The endocrine function of the pancreas involves the secretion of insulin (produced by beta cells) and glucagon (produced by alpha cells) within the pancreatic islets. These 2 hormones regulate the rate of glucose metabolism in the body. The photomicrograph shows pancreatic islets.
DKA DKA Diabetic ketoacidosis (DKA) and hyperosmolar hyperglycemic state (HHS) are serious, acute complications of diabetes mellitus. Diabetic ketoacidosis is characterized by hyperglycemia and ketoacidosis due to an absolute insulin deficiency. Hyperglycemic Crises:
Type 1 Type 1 Spinal Muscular Atrophy DM often presents urgently with DKA DKA Diabetic ketoacidosis (DKA) and hyperosmolar hyperglycemic state (HHS) are serious, acute complications of diabetes mellitus. Diabetic ketoacidosis is characterized by hyperglycemia and ketoacidosis due to an absolute insulin deficiency. Hyperglycemic Crises:
Hyperglycemia:
Type 1 Type 1 Spinal Muscular Atrophy DM can also present with classic symptoms of hyperglycemia:
Type 2 DM has a gradual onset, initially remaining asymptomatic for several years:
Diagnosis of diabetes is based on the presence of inappropriate hyperglycemia in the context of suspicious clinical symptoms.
| Test | Normal range | Increasd risk for diabetes (prediabetes) | Diabetes |
|---|---|---|---|
| Random plasma Plasma The residual portion of blood that is left after removal of blood cells by centrifugation without prior blood coagulation. Transfusion Products glucose Glucose A primary source of energy for living organisms. It is naturally occurring and is found in fruits and other parts of plants in its free state. It is used therapeutically in fluid and nutrient replacement. Lactose Intolerance | Classic hyperglycemic symptoms plus a random plasma Plasma The residual portion of blood that is left after removal of blood cells by centrifugation without prior blood coagulation. Transfusion Products glucose Glucose A primary source of energy for living organisms. It is naturally occurring and is found in fruits and other parts of plants in its free state. It is used therapeutically in fluid and nutrient replacement. Lactose Intolerance > 200 mg/dL | ||
|
Fasting
plasma
Plasma
The residual portion of blood that is left after removal of blood cells by centrifugation without prior blood coagulation.
Transfusion Products
glucose
Glucose
A primary source of energy for living organisms. It is naturally occurring and is found in fruits and other parts of plants in its free state. It is used therapeutically in fluid and nutrient replacement.
Lactose Intolerance (fasting 8 hours) |
< 100 mg/dL | 100–125 mg/dL | ≥ 126 mg/dL |
|
Plasma
Plasma
The residual portion of blood that is left after removal of blood cells by centrifugation without prior blood coagulation.
Transfusion Products
glucose
Glucose
A primary source of energy for living organisms. It is naturally occurring and is found in fruits and other parts of plants in its free state. It is used therapeutically in fluid and nutrient replacement.
Lactose Intolerance after a 2-hour, 75-g OGTT |
< 140 mg/dL | 140–199 mg/dL |
≥ 200 mg/dL
|
| Hemoglobin A1c | < 5.7% | 5.7%–6.4% | ≥ 6.5% |
Oral glucose-tolerance test is recommended during the 24th–28th week of pregnancy Pregnancy The status during which female mammals carry their developing young (embryos or fetuses) in utero before birth, beginning from fertilization to birth. Pregnancy: Diagnosis, Physiology, and Care. Abnormal results are:
No definitive cure exists for diabetes. Management centers around correcting high blood glucose Glucose A primary source of energy for living organisms. It is naturally occurring and is found in fruits and other parts of plants in its free state. It is used therapeutically in fluid and nutrient replacement. Lactose Intolerance with insulin Insulin Insulin is a peptide hormone that is produced by the beta cells of the pancreas. Insulin plays a role in metabolic functions such as glucose uptake, glycolysis, glycogenesis, lipogenesis, and protein synthesis. Exogenous insulin may be needed for individuals with diabetes mellitus, in whom there is a deficiency in endogenous insulin or increased insulin resistance. Insulin ( type 1 Type 1 Spinal Muscular Atrophy) or oral medication (type 2), avoiding low blood glucose Low blood glucose Hypoglycemia is an emergency condition defined as a serum glucose level ≤ 70 mg/dl (≤ 3. 9 mmol/l) in diabetic patients. In nondiabetic patients, there is no specific or defined limit for normal serum glucose levels, and hypoglycemia is defined mainly by its clinical features. Hypoglycemia, and treating the clinical effects of chronic hyperglycemia.
Initial management is with patient education and support.
Classes of oral medications used to treat insulin-resistant diabetes (usually type 2):
Insulin Insulin Insulin is a peptide hormone that is produced by the beta cells of the pancreas. Insulin plays a role in metabolic functions such as glucose uptake, glycolysis, glycogenesis, lipogenesis, and protein synthesis. Exogenous insulin may be needed for individuals with diabetes mellitus, in whom there is a deficiency in endogenous insulin or increased insulin resistance. Insulin therapy is used to treat type 1 Type 1 Spinal Muscular Atrophy DM and sometimes type 2 DM when oral medications alone are no longer sufficient.
The dawn phenomenon:
The Somogyi effect:
Type 1 Type 1 Spinal Muscular Atrophy:
Type 2:
Gestational diabetes:
The risk of complications is proportional to the level of hyperglycemia:
Both type 1 Type 1 Spinal Muscular Atrophy and type 2 DM can cause diabetic complications.