Truncus arteriosus ( TA TA Thyrotoxicosis and Hyperthyroidism) is a congenital heart defect characterized by the persistence of a common cardiac arterial trunk tract that fails to divide into the pulmonary artery Pulmonary artery The short wide vessel arising from the conus arteriosus of the right ventricle and conveying unaerated blood to the lungs. Lungs: Anatomy and aorta Aorta The main trunk of the systemic arteries. Mediastinum and Great Vessels: Anatomy during embryonal development. Truncus arteriosus is a rare congenital malformation with a high mortality Mortality All deaths reported in a given population. Measures of Health Status rate within the 1st 5 weeks of life if not managed promptly. Neonates may be asymptomatic at birth but invariably develop respiratory distress and heart failure Heart Failure A heterogeneous condition in which the heart is unable to pump out sufficient blood to meet the metabolic need of the body. Heart failure can be caused by structural defects, functional abnormalities (ventricular dysfunction), or a sudden overload beyond its capacity. Chronic heart failure is more common than acute heart failure which results from sudden insult to cardiac function, such as myocardial infarction. Total Anomalous Pulmonary Venous Return (TAPVR). Diagnosis is commonly made prenatally through screening Screening Preoperative Care ultrasounds. In the event of a missed antenatal diagnosis, TA TA Thyrotoxicosis and Hyperthyroidism is confirmed through an echocardiogram Echocardiogram Transposition of the Great Arteries. Treatment involves medical stabilization immediately after birth, followed by definitive surgery. With appropriately timed management, the prognosis Prognosis A prediction of the probable outcome of a disease based on a individual's condition and the usual course of the disease as seen in similar situations. Non-Hodgkin Lymphomas is excellent.
Last updated: Mar 27, 2025
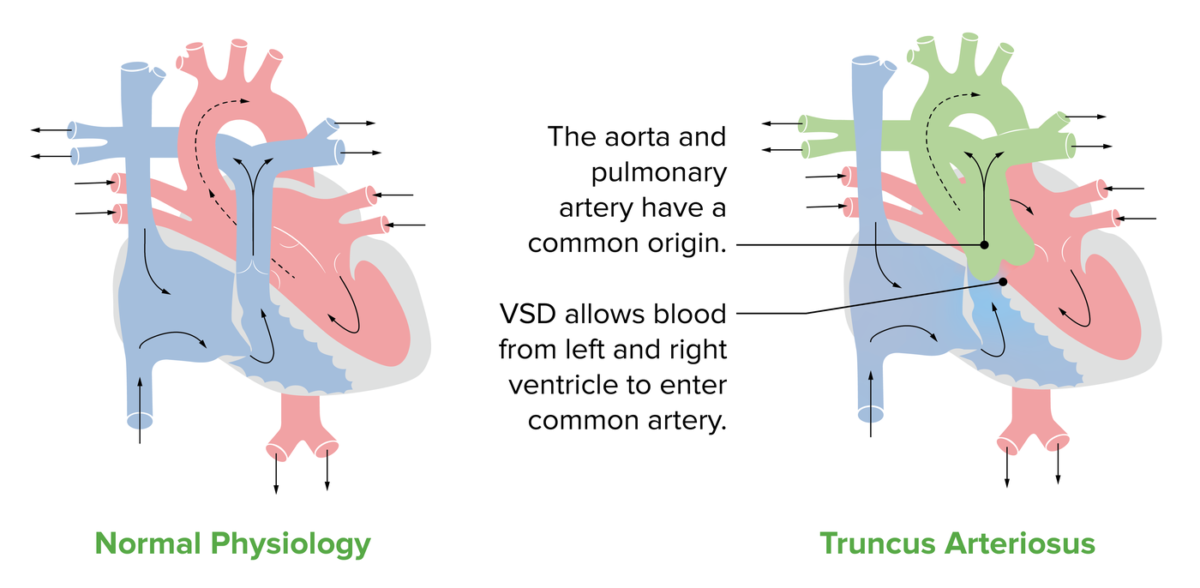
Anatomy of the TA:
A normal heart compared to a heart depicting the features of TA. The green on the diagram represents the common trunk, which gives rise to the pulmonary artery and the aorta. The diagram also shows the most common feature associated with TA, a VSD.
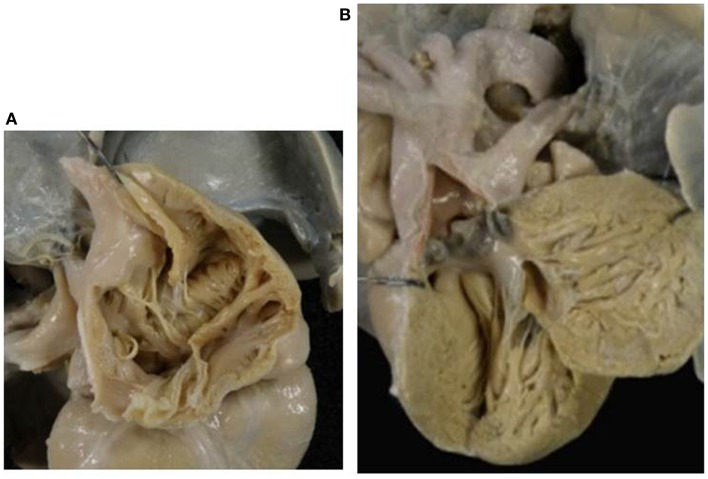
Absent right AV valve (tricuspid atresia) with TA:
A: There is no orifice in the floor of the right atrium.
B: A common arterial trunk arises from the left ventricle, and gives origin to both systemic and pulmonary circulations. Note the dysplastic, tricuspid truncal valve.
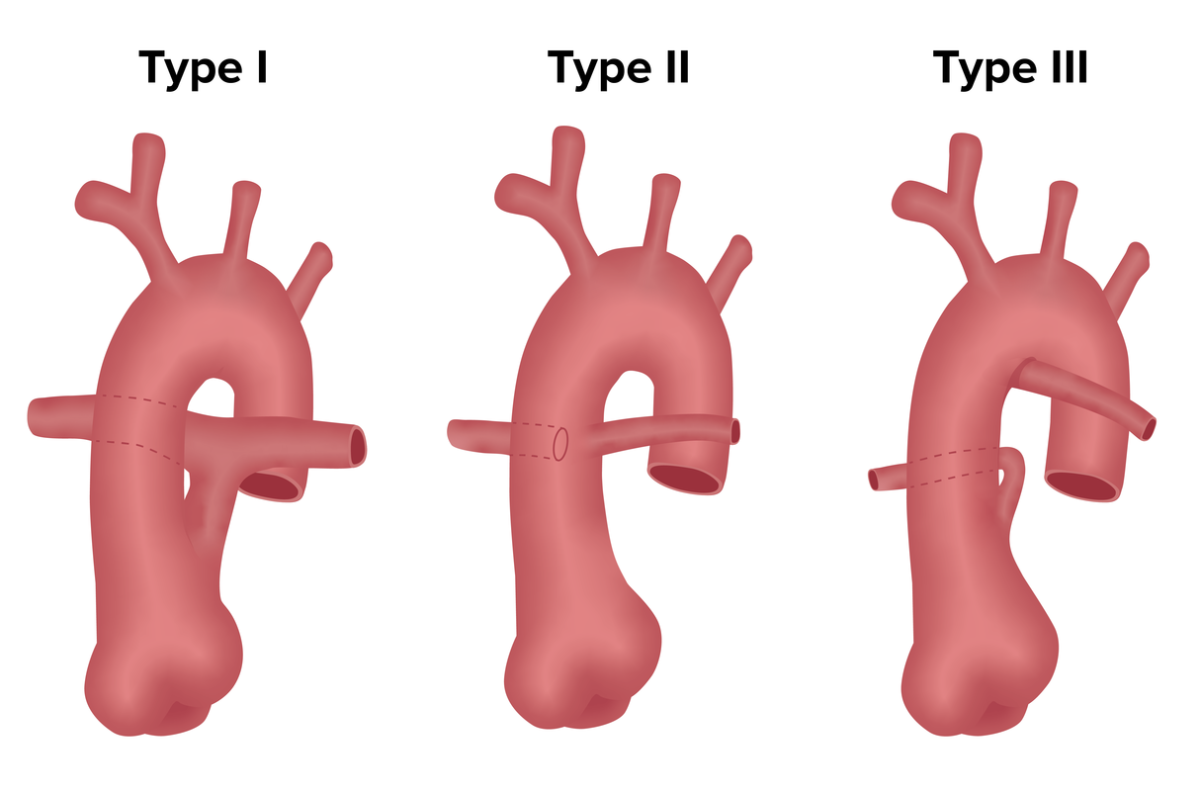
Truncus arteriosus (TA) classification:
Different forms of TA are classified based on how and where the pulmonary arteries arise from the outflow trunk.
In type 1, the posterior left side of TA gives rise to pulmonary trunk, which then branches into left and right pulmonary arteries.
In type 2, there is no true pulmonary trunk; right and left pulmonary arteries arise separately directly from posterior aspect of TA.
Type 3 is similar to type 2 because there is no true pulmonary trunk, but the pulmonary arteries arise from the lateral aspect of TA.
Patients Patients Individuals participating in the health care system for the purpose of receiving therapeutic, diagnostic, or preventive procedures. Clinician–Patient Relationship may be asymptomatic at birth but develop symptoms within 1st few days of life.
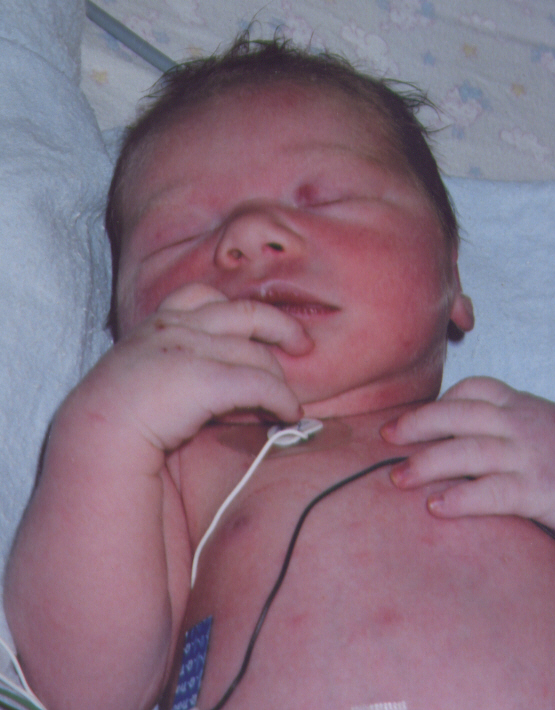
Cyanosis in a neonate due to congenital heart disease:
The infant in the image displays cyanosis due to poor perfusion caused by congenital heart disease (in this case, transposition of the great vessels, but all cyanosis due to congenital heart disease looks similar). Cyanosis can be subtle and is most clearly seen around the lips and fingertips.
The initial management aims to medically stabilize the patient prior to surgical intervention.