Nocardia is a branching, filamentous, gram-positive Gram-Positive Penicillins bacilli Bacilli Shigella. It is partially acid fast Acid fast Tuberculosis due to the presence of mycolic acids Mycolic acids Long fatty acids found in the cell walls of some actinobacteria, including Mycobacterium tuberculosis. Mycobacterium in the cell wall Cell wall The outermost layer of a cell in most plants; bacteria; fungi; and algae. The cell wall is usually a rigid structure that lies external to the cell membrane, and provides a protective barrier against physical or chemical agents. Cell Types: Eukaryotic versus Prokaryotic. Nocardia is a ubiquitous soil organism that most commonly affects immunocompromised immunocompromised A human or animal whose immunologic mechanism is deficient because of an immunodeficiency disorder or other disease or as the result of the administration of immunosuppressive drugs or radiation. Gastroenteritis patients Patients Individuals participating in the health care system for the purpose of receiving therapeutic, diagnostic, or preventive procedures. Clinician–Patient Relationship. Nocardia is transmitted via inhalation of aerosolized bacteria Bacteria Bacteria are prokaryotic single-celled microorganisms that are metabolically active and divide by binary fission. Some of these organisms play a significant role in the pathogenesis of diseases. Bacteriology or less commonly, via direct contact with wounds. Nocardia causes opportunistic infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease, primarily pulmonary infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease ( pneumonia Pneumonia Pneumonia or pulmonary inflammation is an acute or chronic inflammation of lung tissue. Causes include infection with bacteria, viruses, or fungi. In more rare cases, pneumonia can also be caused through toxic triggers through inhalation of toxic substances, immunological processes, or in the course of radiotherapy. Pneumonia, abscess Abscess Accumulation of purulent material in tissues, organs, or circumscribed spaces, usually associated with signs of infection. Chronic Granulomatous Disease, or cavitary lesions), which may spread to form brain Brain The part of central nervous system that is contained within the skull (cranium). Arising from the neural tube, the embryonic brain is comprised of three major parts including prosencephalon (the forebrain); mesencephalon (the midbrain); and rhombencephalon (the hindbrain). The developed brain consists of cerebrum; cerebellum; and other structures in the brain stem. Nervous System: Anatomy, Structure, and Classification abscesses. In immunocompetent patients Patients Individuals participating in the health care system for the purpose of receiving therapeutic, diagnostic, or preventive procedures. Clinician–Patient Relationship, Nocardia can cause a cutaneous infection. Treatment for nocardiosis is with trimethoprim Trimethoprim The sulfonamides are a class of antimicrobial drugs inhibiting folic acid synthesize in pathogens. The prototypical drug in the class is sulfamethoxazole. Although not technically sulfonamides, trimethoprim, dapsone, and pyrimethamine are also important antimicrobial agents inhibiting folic acid synthesis. The agents are often combined with sulfonamides, resulting in a synergistic effect. Sulfonamides and Trimethoprim– sulfamethoxazole Sulfamethoxazole A bacteriostatic antibacterial agent that interferes with folic acid synthesis in susceptible bacteria. Its broad spectrum of activity has been limited by the development of resistance. Sulfonamides and Trimethoprim and/or surgical intervention as indicated.
Last updated: Dec 15, 2025
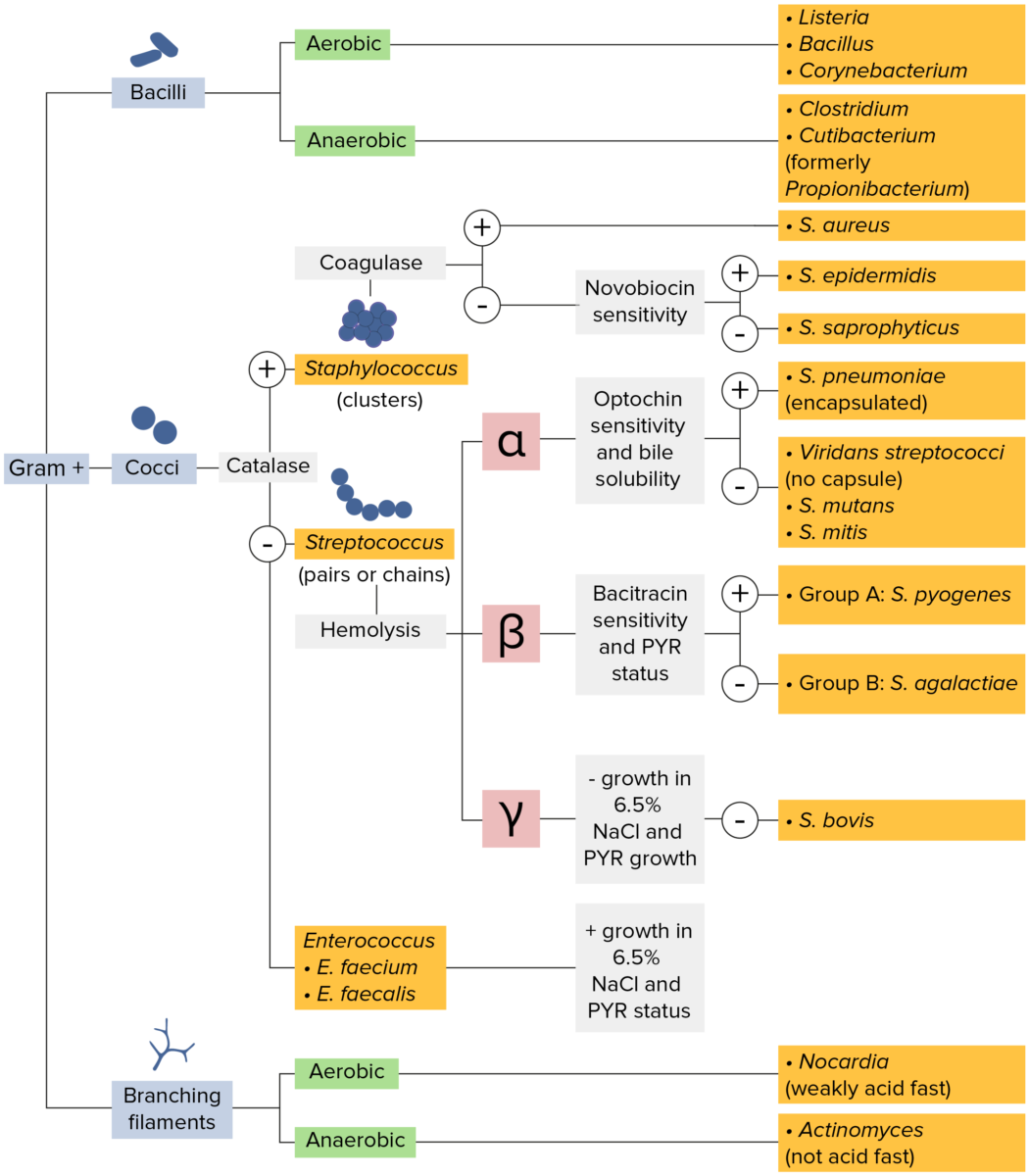
Gram-positive bacteria:
Most bacteria can be classified according to a lab procedure called Gram staining.
Bacteria with cell walls that have a thick layer of peptidoglycan retain the crystal violet stain utilized in Gram staining but are not affected by the safranin counterstain. These bacteria appear as purple-blue on the stain, indicating that they are gram positive. The bacteria can be further classified according to morphology (branching filaments, bacilli, and cocci in clusters or chains) and their ability to grow in the presence of oxygen (aerobic versus anaerobic). The cocci can also be further identified. Staphylococci can be narrowed down on the basis of the presence of the enzyme coagulase and on their sensitivity to the antibiotic novobiocin. Streptococci are grown on blood agar and classified on the basis of which form of hemolysis they employ (α, β, or γ). Streptococci are further narrowed on the basis of their response to the pyrrolidonyl-β-naphthylamide (PYR) test, their sensitivity to specific antimicrobials (optochin and bacitracin), and their ability to grow on sodium chloride (NaCl) media.
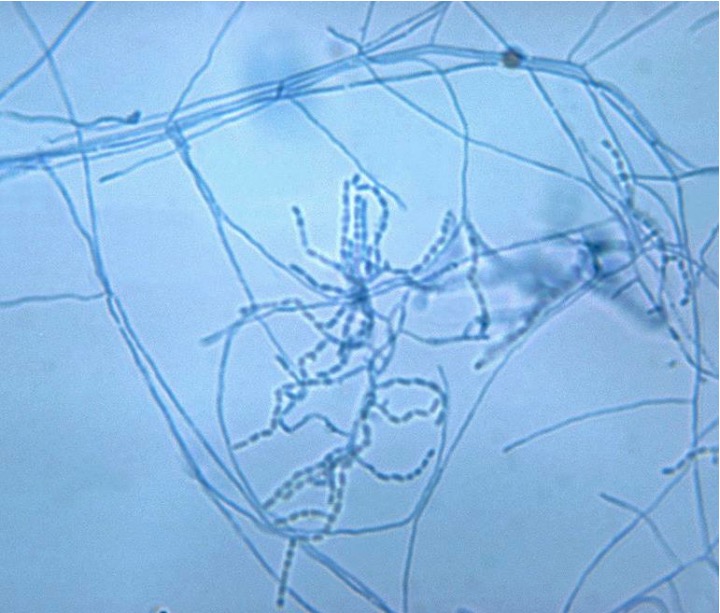
This slide with gram-positive aerobic Nocardia asteroides reveals chains of bacteria among aerial mycelia.
Image by Lecturio.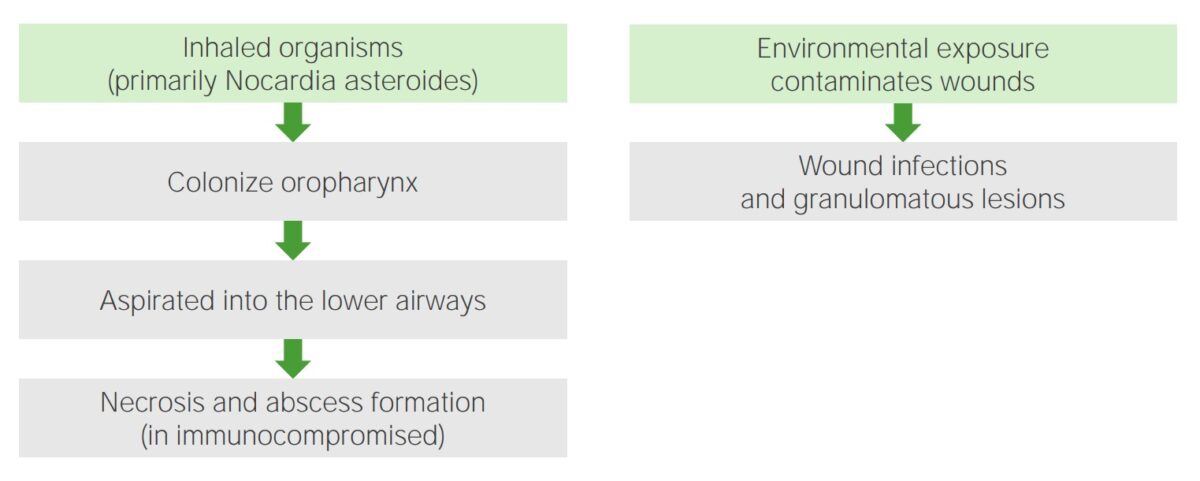
Pathogenesis of Nocardia:
Left: Inhaled organisms colonize the oropharynx and are passively aspirated into the lower airways, resulting in necrosis and abscess formation (in immunocompromised patients).
Right: Environmental exposure and contamination of wounds lead to wound infections or granulomatous lesions.
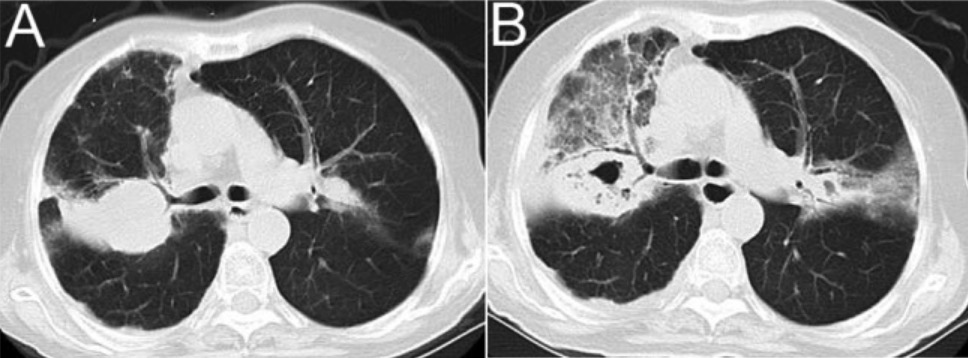
Computed tomography (CT) scan imaging of the lung in a patient with chronic obstructive pulmonary disease and Nocardia infection:
A: right upper lobar consolidation
B: computed tomography images after 12 days, showing cavitary consolidation and ground glass opacity
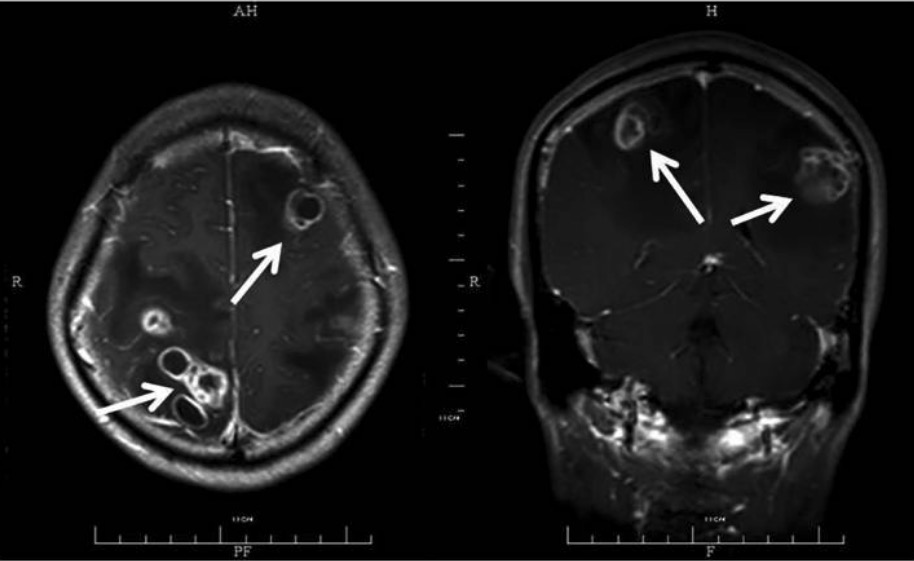
Magnetic resonance imaging (MRI) with contrast showing multiple abscesses (arrows): The patient has disseminated CNS nocardiosis, with bilateral involvement of the brain. Multiple ring lesions with surrounding edema are shown.
Image: “45-year-old male” by the State Key Laboratory for Diagnosis and Treatment of Infectious Diseases, The First Affiliated Hospital, School of Medicine, Zhejiang University, Hangzhou, Zhejiang 310003, P.R. China. License: CC BY 3.0.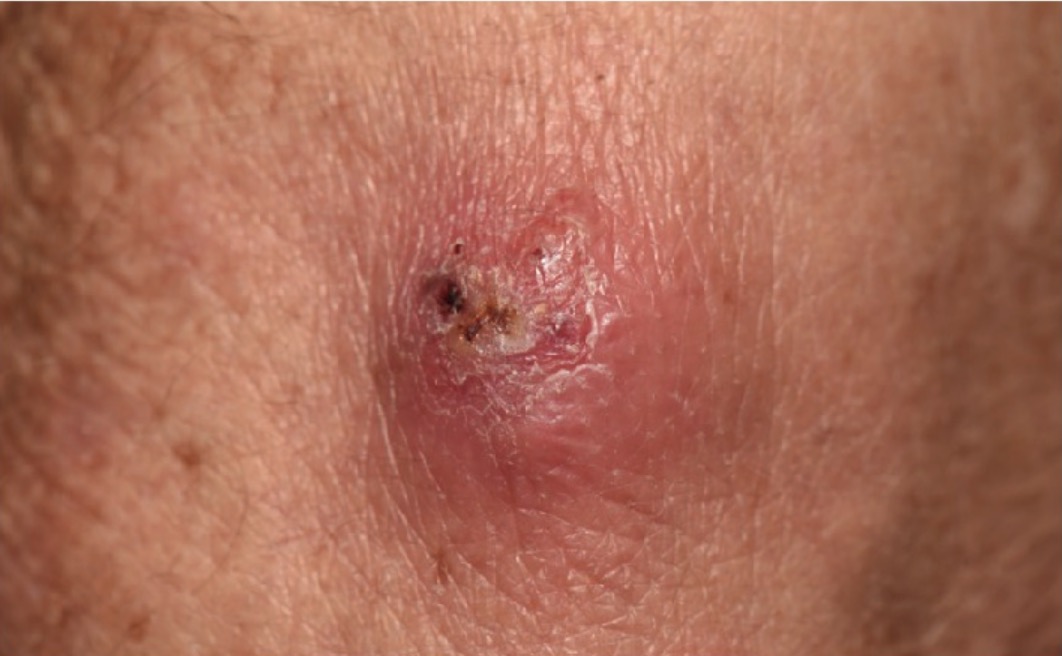
Image of cutaneous nocardiosis: fluctuant, purulent, and crusted nodule noted in a patient receiving long-term immunosuppressive therapy. Tissue cultures revealed N. brasiliensis.
Image: “Lesional skin” by Alison M. Fernandes et al. License: CC BY 4.0.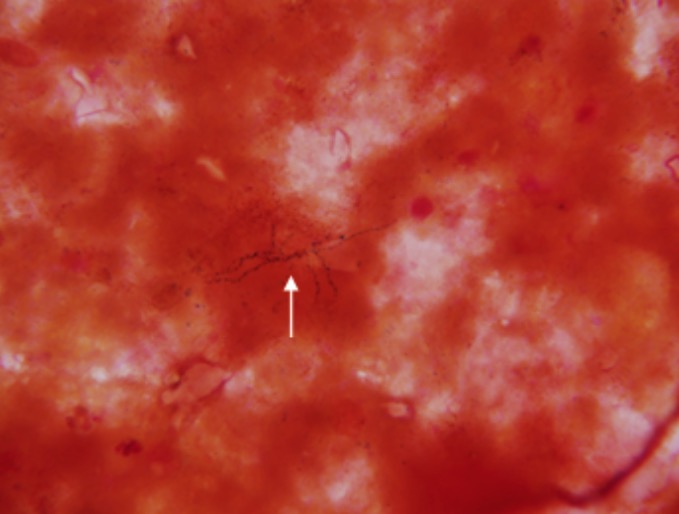
Nocardia (white arrow) as found on a brain biopsy
Image: “Nocardia as found on a brain biopsy” by Doc James. License: Public domain, edited by Lecturio.| Differentiating factors | Actinomyces Actinomyces Actinomyces is an anaerobic, gram-positive, branching, filamentous rod. Actinomyces israelii is the most common species involved in human disease. The organism is commonly found as part of the normal flora in the oral cavity, gastrointestinal tract, and reproductive tract. Actinomyces/Actinomycosis | Nocardia |
|---|---|---|
| Oxygen requirement | Anaerobic | Aerobic |
| Acid-fast stain Acid-Fast Stain Meningitis in Children | Not acid fast Acid fast Tuberculosis | Acid fast Acid fast Tuberculosis (partially) |
| Reservoir Reservoir Animate or inanimate sources which normally harbor disease-causing organisms and thus serve as potential sources of disease outbreaks. Reservoirs are distinguished from vectors (disease vectors) and carriers, which are agents of disease transmission rather than continuing sources of potential disease outbreaks. Humans may serve both as disease reservoirs and carriers. Escherichia coli/habitat | Normal oral, gastrointestinal, and reproductive flora | Found in soil, water, decaying organic matter |
| Infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease caused |
|
|
| Management | Treatment with penicillin Penicillin Rheumatic Fever | Treatment with trimethoprim-sulfamethoxazole |
| Differentiating factors | Mycobacterium Mycobacterium Mycobacterium is a genus of the family Mycobacteriaceae in the phylum Actinobacteria. Mycobacteria comprise more than 150 species of facultative intracellular bacilli that are mostly obligate aerobes. Mycobacteria are responsible for multiple human infections including serious diseases, such as tuberculosis (M. tuberculosis), leprosy (M. leprae), and M. avium complex infections. Mycobacterium | Nocardia |
|---|---|---|
| Oxygen requirement | Aerobic | Aerobic |
| Spore Spore The reproductive elements of lower organisms, such as bacteria; fungi; and cryptogamic plants. Microsporidia/Microsporidiosis formation | Non-spore forming | Non-spore forming |
| Acid-fast stain Acid-Fast Stain Meningitis in Children | Acid fast Acid fast Tuberculosis | Partially acid fast Acid fast Tuberculosis |
| Structure | Straight or slightly curved rods | Branching filamentous rods |
| PPD* test | PPD* positive | PPD* negative |