Mycobacterium is a genus of the family Mycobacteriaceae in the phylum Actinobacteria. Mycobacteria comprise more than 150 species of facultative intracellular Facultative intracellular Yersinia spp./Yersiniosis bacilli Bacilli Shigella that are mostly obligate aerobes Obligate aerobes Bacteriology. Mycobacteria are responsible for multiple human infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease including serious diseases, such as tuberculosis Tuberculosis Tuberculosis (TB) is an infectious disease caused by Mycobacterium tuberculosis complex bacteria. The bacteria usually attack the lungs but can also damage other parts of the body. Approximately 30% of people around the world are infected with this pathogen, with the majority harboring a latent infection. Tuberculosis spreads through the air when a person with active pulmonary infection coughs or sneezes. Tuberculosis (M. tuberculosis Tuberculosis Tuberculosis (TB) is an infectious disease caused by Mycobacterium tuberculosis complex bacteria. The bacteria usually attack the lungs but can also damage other parts of the body. Approximately 30% of people around the world are infected with this pathogen, with the majority harboring a latent infection. Tuberculosis spreads through the air when a person with active pulmonary infection coughs or sneezes. Tuberculosis), leprosy Leprosy Leprosy, also known as Hansen's disease, is a chronic bacterial infection caused by Mycobacterium leprae complex bacteria. Symptoms primarily affect the skin and peripheral nerves, resulting in cutaneous manifestations (e.g., hypopigmented macules) and neurologic manifestations (e.g., loss of sensation). Leprosy (M. leprae), and M. avium complex infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease. While lungs Lungs Lungs are the main organs of the respiratory system. Lungs are paired viscera located in the thoracic cavity and are composed of spongy tissue. The primary function of the lungs is to oxygenate blood and eliminate CO2. Lungs: Anatomy are the most common site of infection, mycobacteria can colonize and infect other organ systems including the lymph nodes Lymph Nodes They are oval or bean shaped bodies (1 - 30 mm in diameter) located along the lymphatic system. Lymphatic Drainage System: Anatomy, skin Skin The skin, also referred to as the integumentary system, is the largest organ of the body. The skin is primarily composed of the epidermis (outer layer) and dermis (deep layer). The epidermis is primarily composed of keratinocytes that undergo rapid turnover, while the dermis contains dense layers of connective tissue. Skin: Structure and Functions, sinuses, eyes, ears, bones, CNS, and urinary tract Urinary tract The urinary tract is located in the abdomen and pelvis and consists of the kidneys, ureters, urinary bladder, and urethra. The structures permit the excretion of urine from the body. Urine flows from the kidneys through the ureters to the urinary bladder and out through the urethra. Urinary Tract: Anatomy.
Last updated: Jan 20, 2026
Morphology and properties:
Staining properties:
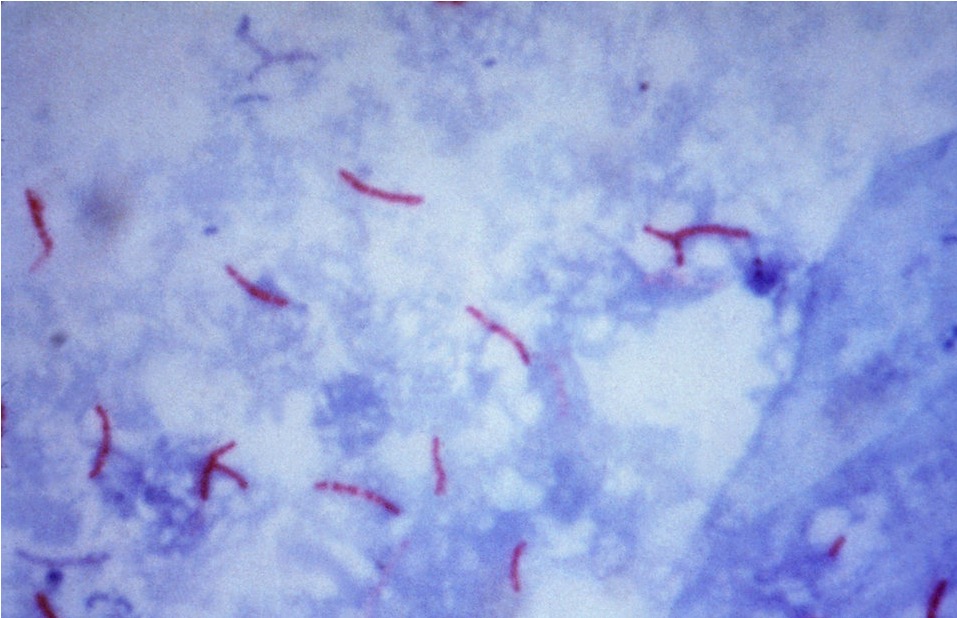
Acid-fast stain of Mycobacterium tuberculosis
Image: “Mycobacterium tuberculosis bacteria” by CDC/Dr. George P. Kubica. License: Public Domain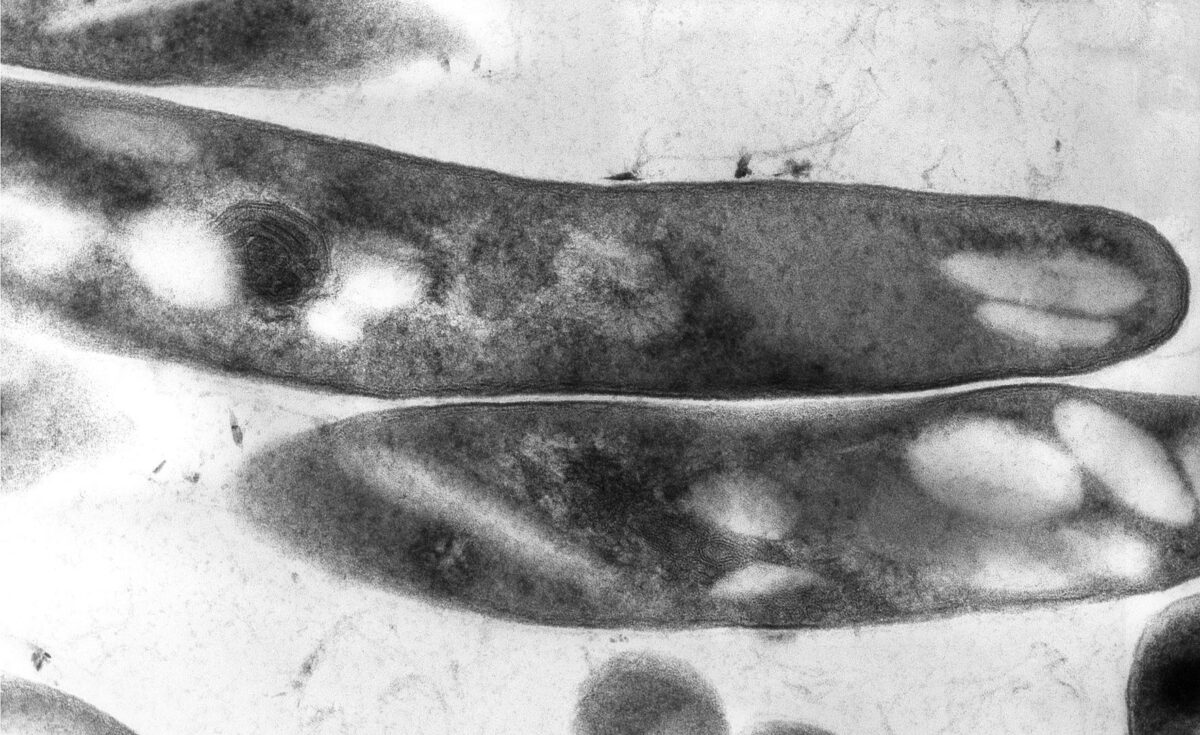
Electron micrograph of Mycobacterium tuberculosis
Image: “Mycobacterium tuberculosis 01” by Elizabeth White. License: Public DomainMycobacteria:
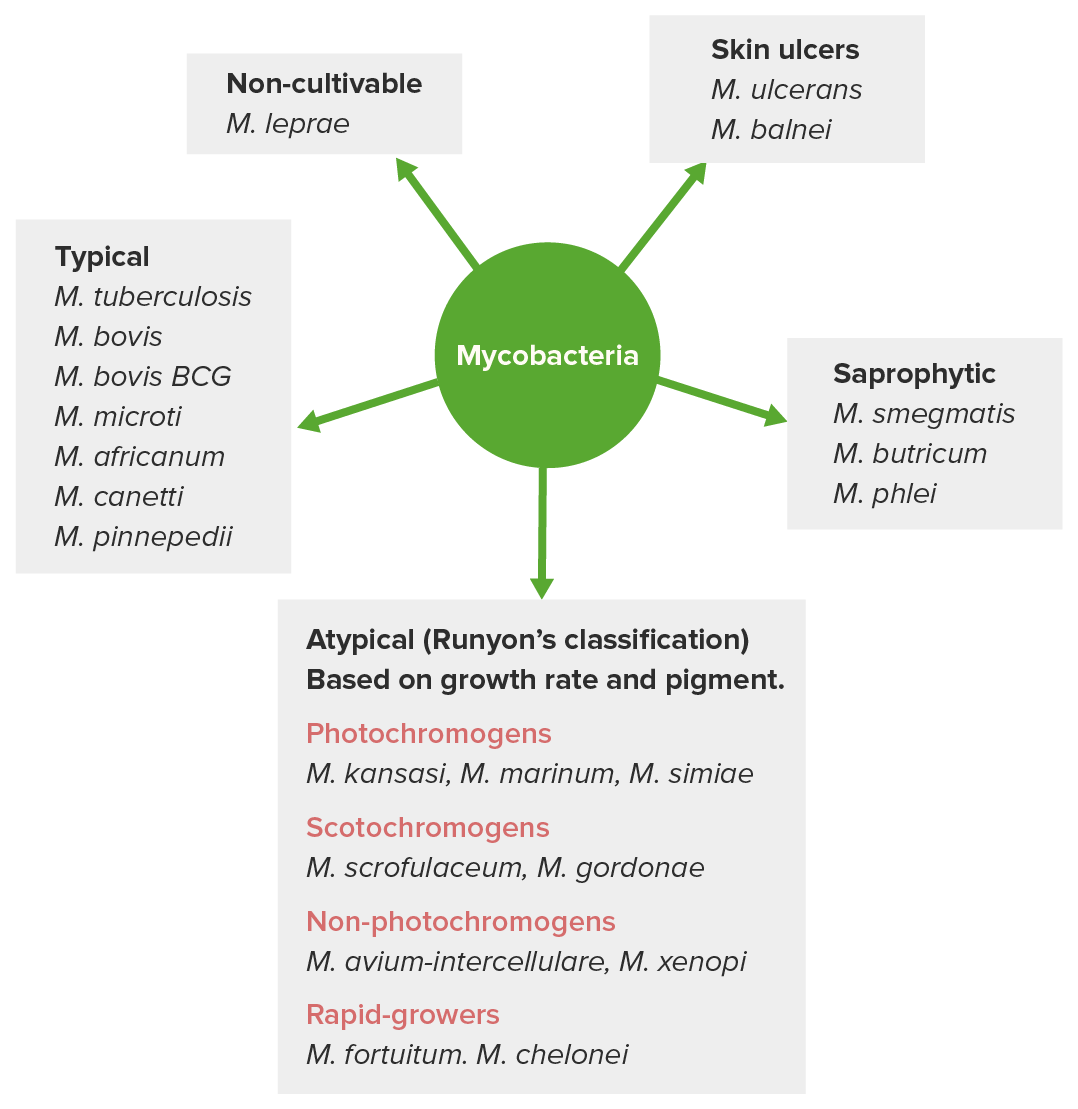
Classification of mycobacteria
Image by Lecturio. License: CC BY-NC-SA 4.0Most important pathogenic species:
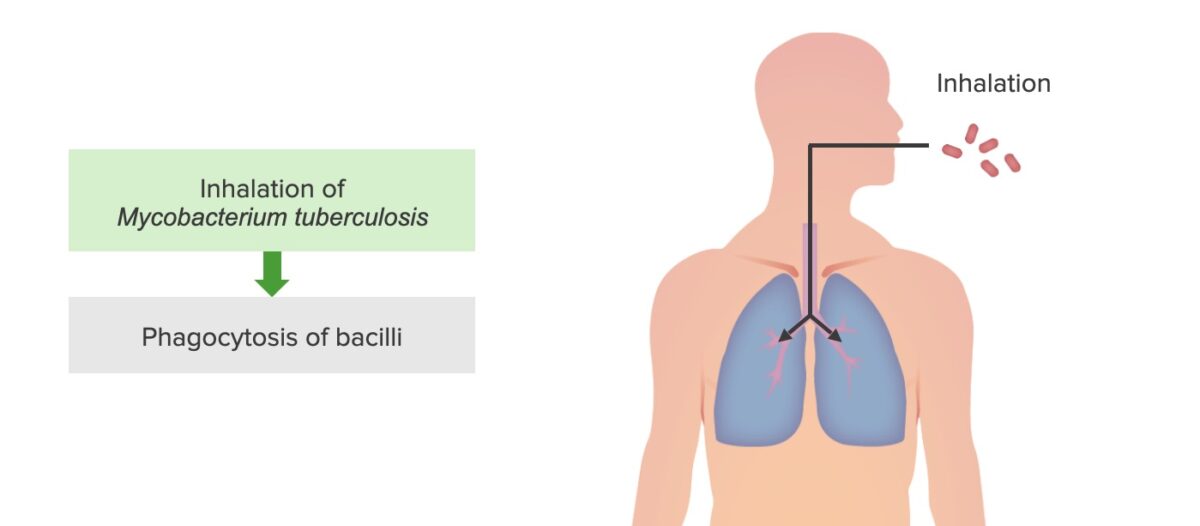
Tuberculosis infection step 1: M. tuberculosis inhalation and phagocytosis by alveolar macrophages
Image by Lecturio. License: CC BY-NC-SA 4.0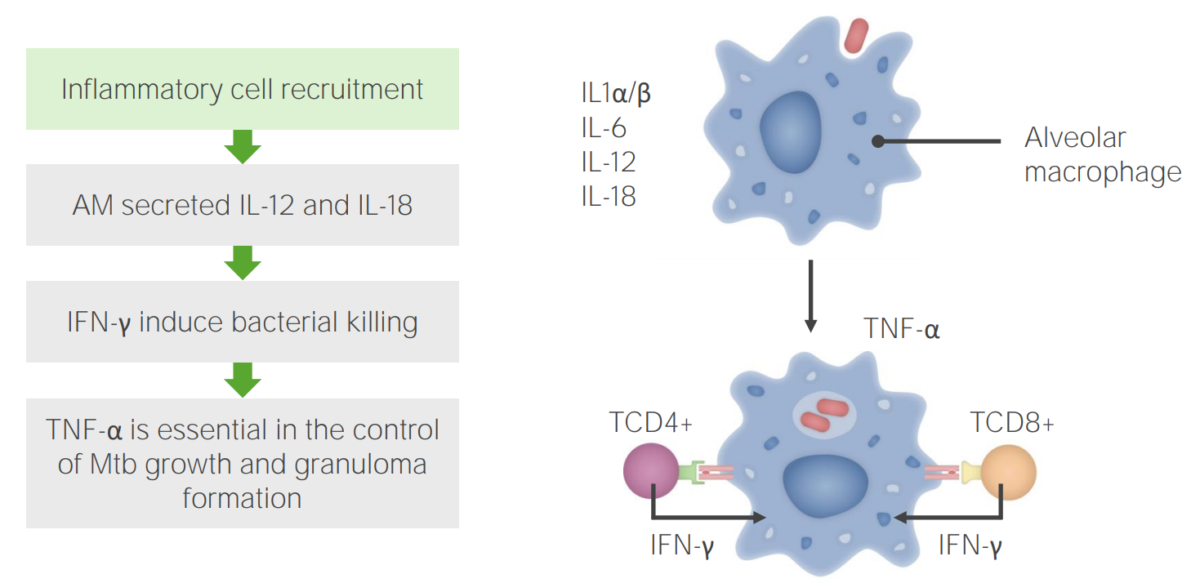
Tuberculosis infection step 2: Inflammatory cell recruitment
AM: alveolar macrophages
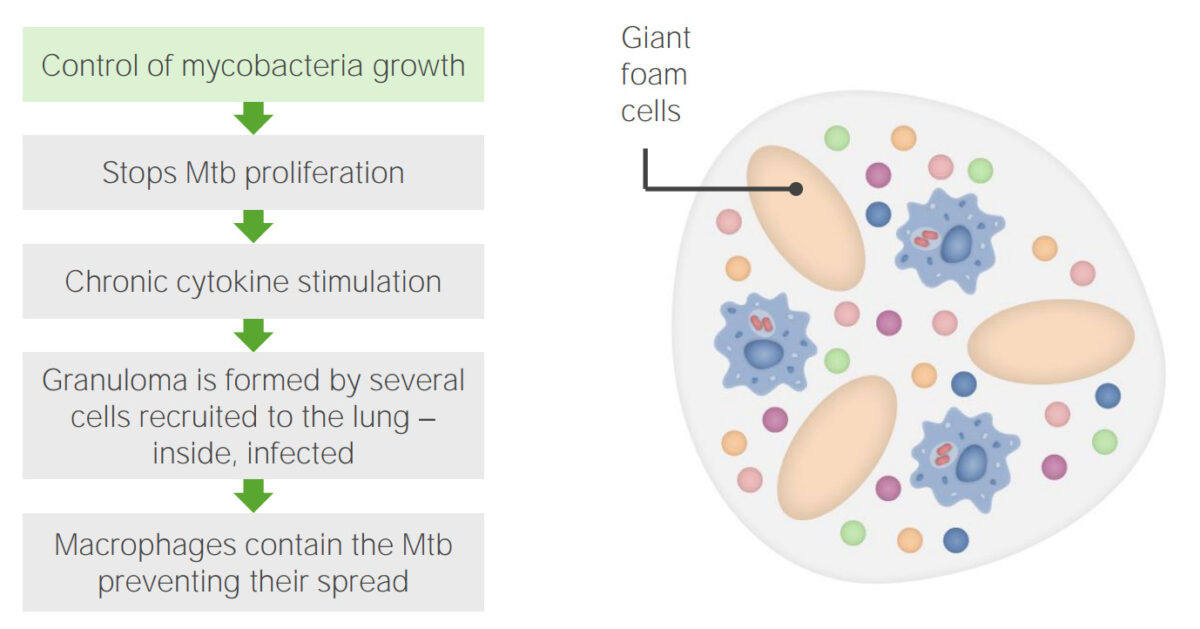
Tuberculosis infection step 3: Granuloma formation
Image by Lecturio. License: CC BY-NC-SA 4.0| Primary TB TB Tuberculosis (TB) is an infectious disease caused by Mycobacterium tuberculosis complex bacteria. The bacteria usually attack the lungs but can also damage other parts of the body. Approximately 30% of people around the world are infected with this pathogen, with the majority harboring a latent infection. Tuberculosis spreads through the air when a person with active pulmonary infection coughs or sneezes. Tuberculosis | Secondary TB TB Tuberculosis (TB) is an infectious disease caused by Mycobacterium tuberculosis complex bacteria. The bacteria usually attack the lungs but can also damage other parts of the body. Approximately 30% of people around the world are infected with this pathogen, with the majority harboring a latent infection. Tuberculosis spreads through the air when a person with active pulmonary infection coughs or sneezes. Tuberculosis | Extrapulmonary (miliary) TB TB Tuberculosis (TB) is an infectious disease caused by Mycobacterium tuberculosis complex bacteria. The bacteria usually attack the lungs but can also damage other parts of the body. Approximately 30% of people around the world are infected with this pathogen, with the majority harboring a latent infection. Tuberculosis spreads through the air when a person with active pulmonary infection coughs or sneezes. Tuberculosis | |
|---|---|---|---|
| Presentation | Within 2 years of infection in 5%–10% of cases | Reactivation Reactivation Herpes Simplex Virus 1 and 2 of infection in immunocompromised immunocompromised A human or animal whose immunologic mechanism is deficient because of an immunodeficiency disorder or other disease or as the result of the administration of immunosuppressive drugs or radiation. Gastroenteritis patients Patients Individuals participating in the health care system for the purpose of receiving therapeutic, diagnostic, or preventive procedures. Clinician–Patient Relationship | Immunocompromised immunocompromised A human or animal whose immunologic mechanism is deficient because of an immunodeficiency disorder or other disease or as the result of the administration of immunosuppressive drugs or radiation. Gastroenteritis individuals |
| Location |
|
|
|
| Symptoms |
|
Recurrence of symptoms |
|
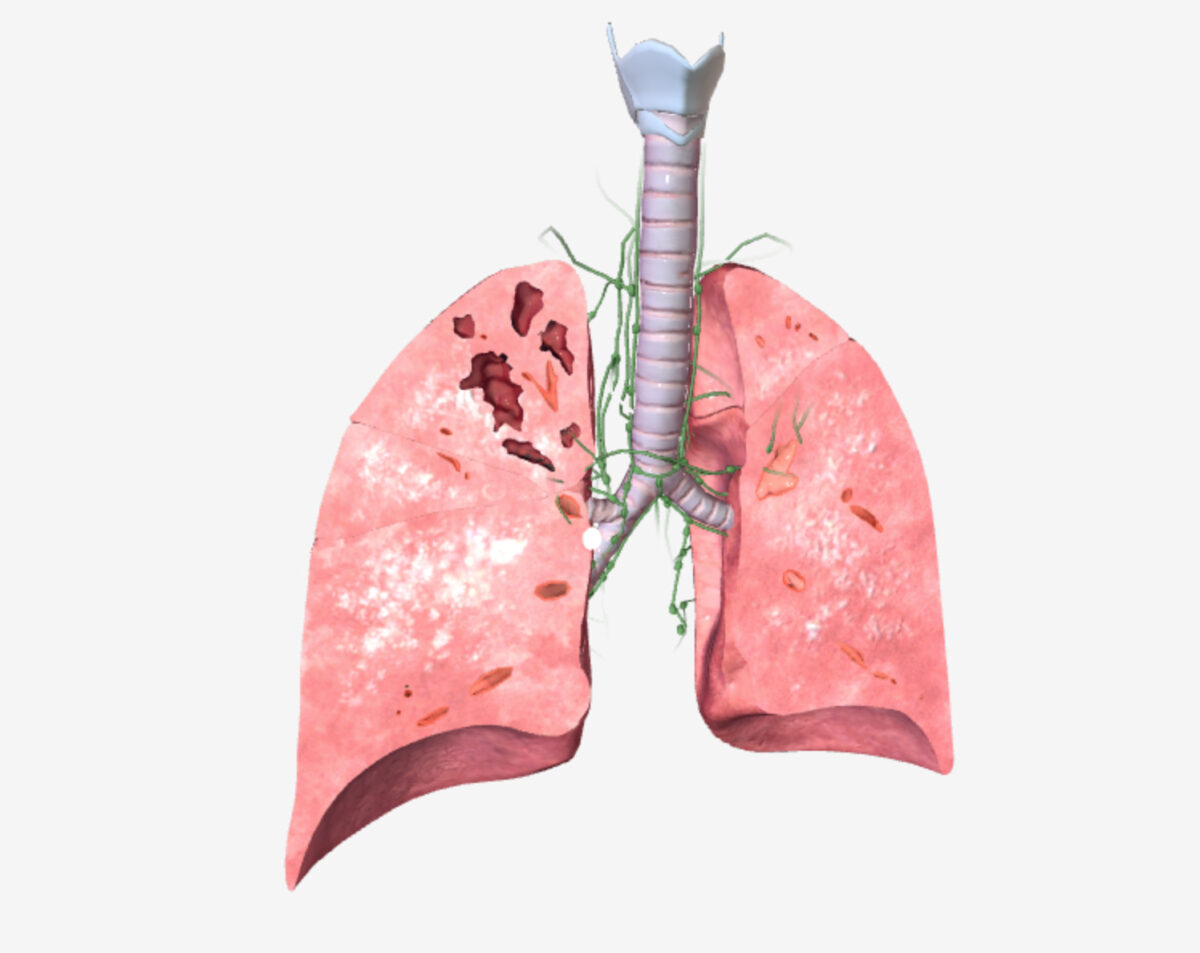
Pulmonary tuberculosis (TB): primary and secondary disease manifestations
Image by BioDigital, edited by Lecturio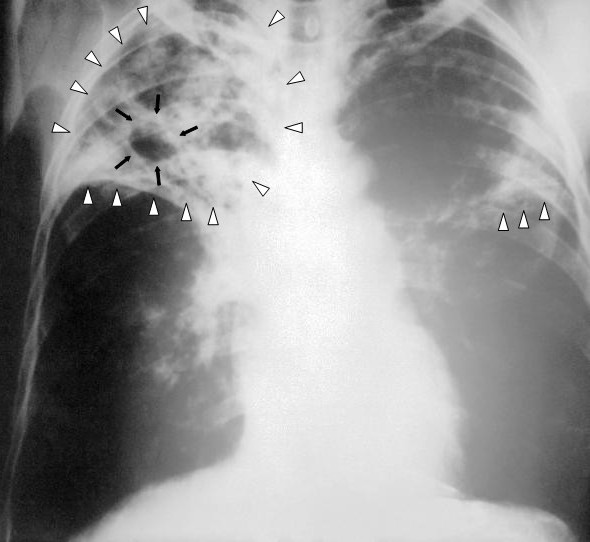
X-ray of a patient with tuberculosis:
Chest X-ray of bilateral reticular infiltrates (white triangles) and cavitary lesion (black arrows) in the right upper lobe
Sputum:
Blood or urine mycobacterial culture: in patients Patients Individuals participating in the health care system for the purpose of receiving therapeutic, diagnostic, or preventive procedures. Clinician–Patient Relationship with HIV HIV Anti-HIV Drugs or immunocompromised immunocompromised A human or animal whose immunologic mechanism is deficient because of an immunodeficiency disorder or other disease or as the result of the administration of immunosuppressive drugs or radiation. Gastroenteritis individuals
Tuberculin Tuberculin A protein extracted from boiled culture of tubercle bacilli (Mycobacterium tuberculosis). It is used in the tuberculin skin test (tuberculin test) for the diagnosis of tuberculosis infection in asymptomatic persons. Type IV Hypersensitivity Reaction skin Skin The skin, also referred to as the integumentary system, is the largest organ of the body. The skin is primarily composed of the epidermis (outer layer) and dermis (deep layer). The epidermis is primarily composed of keratinocytes that undergo rapid turnover, while the dermis contains dense layers of connective tissue. Skin: Structure and Functions test (TST; PPD (purified protein derivative) or Mantoux test):
lnterferon-γ release assay ( IGRA IGRA The assay of interferon-gamma released from lymphocytes after their exposure to a specific test antigen, to check for immunologic memory resulting from a previous exposure to the antigen. The amount of interferon-gamma released is usually assayed by an enzyme-linked immunosorbent assay. Tuberculosis): no distinction between active and inactive TB TB Tuberculosis (TB) is an infectious disease caused by Mycobacterium tuberculosis complex bacteria. The bacteria usually attack the lungs but can also damage other parts of the body. Approximately 30% of people around the world are infected with this pathogen, with the majority harboring a latent infection. Tuberculosis spreads through the air when a person with active pulmonary infection coughs or sneezes. Tuberculosis

Immune response to leprosy
Image by Lecturio. License: CC BY-NC-SA 4.0| Tuberculoid | Lepromatous | |
|---|---|---|
| Presentation | Immunocompetent individuals | Immunocompromised immunocompromised A human or animal whose immunologic mechanism is deficient because of an immunodeficiency disorder or other disease or as the result of the administration of immunosuppressive drugs or radiation. Gastroenteritis individuals |
| Location | Skin Skin The skin, also referred to as the integumentary system, is the largest organ of the body. The skin is primarily composed of the epidermis (outer layer) and dermis (deep layer). The epidermis is primarily composed of keratinocytes that undergo rapid turnover, while the dermis contains dense layers of connective tissue. Skin: Structure and Functions and nerves | Skin Skin The skin, also referred to as the integumentary system, is the largest organ of the body. The skin is primarily composed of the epidermis (outer layer) and dermis (deep layer). The epidermis is primarily composed of keratinocytes that undergo rapid turnover, while the dermis contains dense layers of connective tissue. Skin: Structure and Functions and nerves |
| Symptoms |
|
|
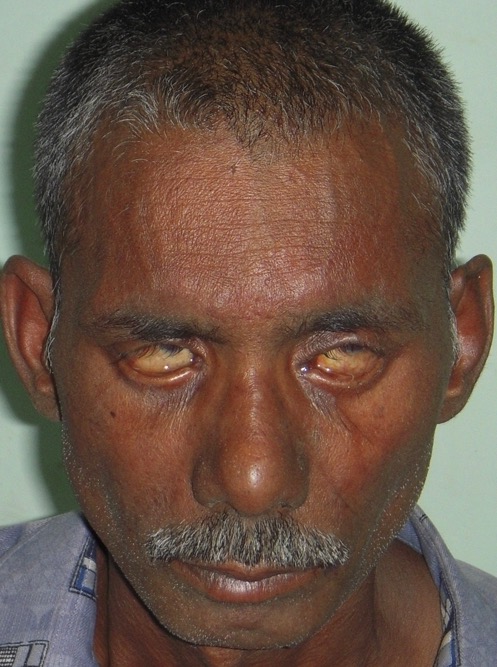
Lagophthalmos in a patient with lepromatous leprosy:
Patient is attempting to close his eyelids but is unable to do so.
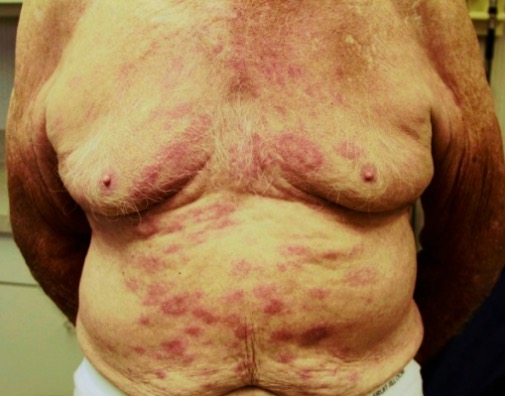
Leprosy:
Erythematous skin patches
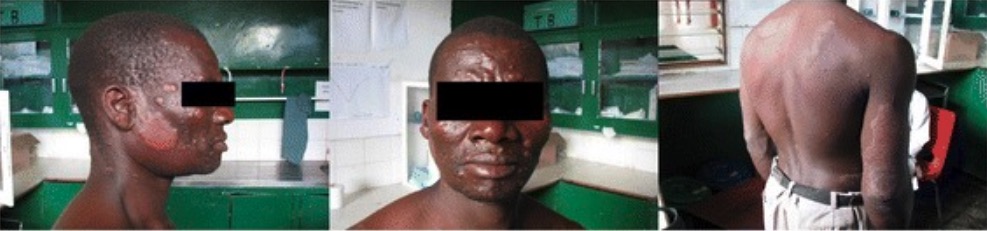
Widespread skin lesions consistent with lepromatous leprosy:
The patient is beginning to develop leonine facies, with a large number of papules and nodules afflicting the face, chest, back, legs, and groin. Large well-circumscribed plaques appear on the triceps aspect of the right arm and over his left shoulder and right flank.
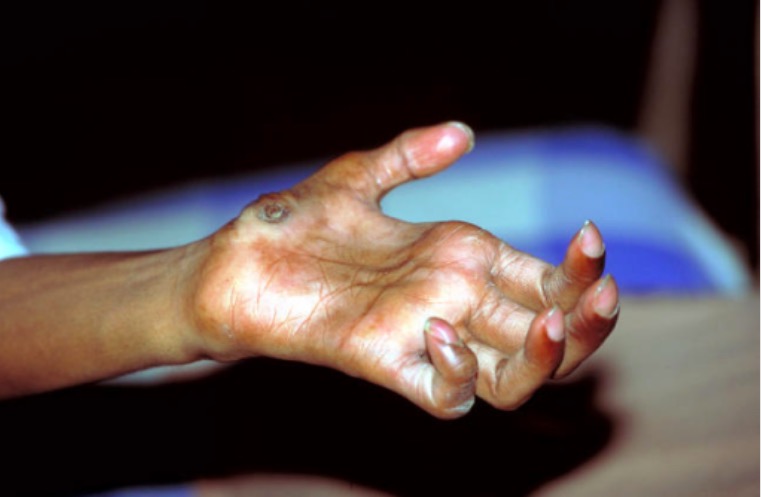
Clawed fingers:
Typical deformity in a patient with advanced leprosy
The organism is identified by skin biopsy Skin Biopsy Secondary Skin Lesions:
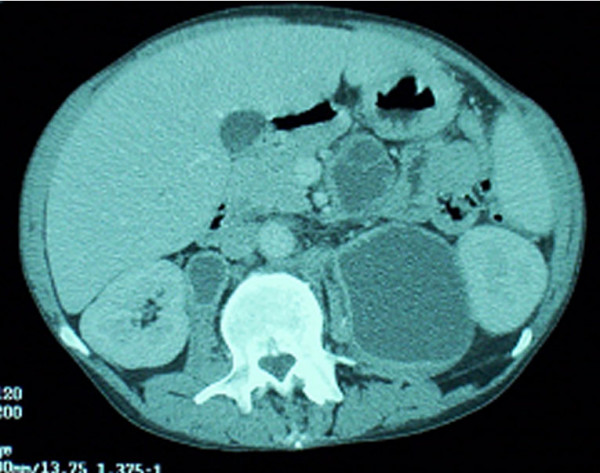
MAC infection: abdominal CT demonstrating multiple abscessed intra-abdominal lymph nodes
Image: “Mycobacterium avium complex immune reconstitution inflammatory syndrome: long term outcomes” by Riddell J, Kaul DR, Karakousis PC, Gallant JE, Mitty J, Kazanjian PH. License: CC BY 2.0