Lambert-Eaton myasthenic syndrome (LEMS) is an autoimmune disorder Autoimmune Disorder Septic Arthritis affecting the neuromuscular junction Neuromuscular junction The synapse between a neuron and a muscle. Skeletal Muscle Contraction and has a strong association with small cell lung carcinoma. Lambert-Eaton myasthenic syndrome affects the voltage-gated calcium Calcium A basic element found in nearly all tissues. It is a member of the alkaline earth family of metals with the atomic symbol ca, atomic number 20, and atomic weight 40. Calcium is the most abundant mineral in the body and combines with phosphorus to form calcium phosphate in the bones and teeth. It is essential for the normal functioning of nerves and muscles and plays a role in blood coagulation (as factor IV) and in many enzymatic processes. Electrolytes channels Channels The Cell: Cell Membrane at the presynaptic membrane Presynaptic membrane Synapses and Neurotransmission. Presentation includes proximal muscle weakness and symptoms of autonomic dysfunction Autonomic Dysfunction Anterior Cord Syndrome such as dry mouth and sluggish pupillary reflexes. Diagnosis includes nerve conduction studies/ electromyography Electromyography Recording of the changes in electric potential of muscle by means of surface or needle electrodes. Becker Muscular Dystrophy (EMG) and serum detection of antibodies Antibodies Immunoglobulins (Igs), also known as antibodies, are glycoprotein molecules produced by plasma cells that act in immune responses by recognizing and binding particular antigens. The various Ig classes are IgG (the most abundant), IgM, IgE, IgD, and IgA, which differ in their biologic features, structure, target specificity, and distribution. Immunoglobulins: Types and Functions. Management is mainly symptomatic with the use of potassium Potassium An element in the alkali group of metals with an atomic symbol k, atomic number 19, and atomic weight 39. 10. It is the chief cation in the intracellular fluid of muscle and other cells. Potassium ion is a strong electrolyte that plays a significant role in the regulation of fluid volume and maintenance of the water-electrolyte balance. Hyperkalemia channel blockers and immunosuppressants Immunosuppressants Immunosuppressants are a class of drugs widely used in the management of autoimmune conditions and organ transplant rejection. The general effect is dampening of the immune response. Immunosuppressants. Prognosis Prognosis A prediction of the probable outcome of a disease based on a individual's condition and the usual course of the disease as seen in similar situations. Non-Hodgkin Lymphomas is good for nonparaneoplastic LEMS but usually poor for paraneoplastic LEMS secondary to underlying malignancy Malignancy Hemothorax.
Last updated: Nov 17, 2022
Lambert-Eaton myasthenic syndrome (LEMS) is an autoimmune disorder Autoimmune Disorder Septic Arthritis affecting the neuromuscular junction Neuromuscular junction The synapse between a neuron and a muscle. Skeletal Muscle Contraction and has a strong association with small cell lung carcinoma.
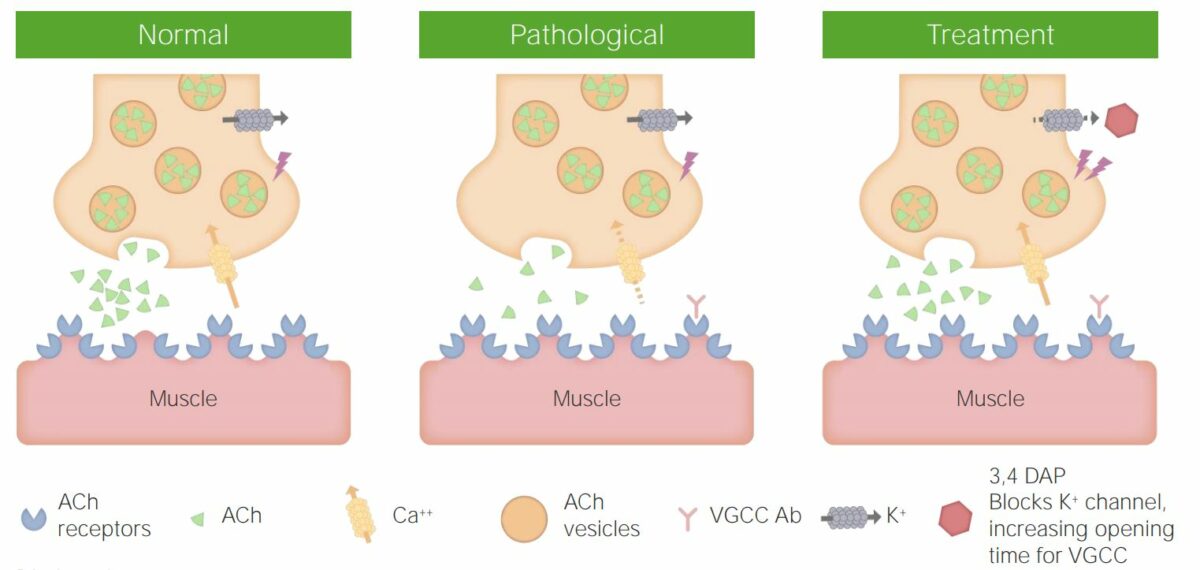
Illustrations showing normal activity at the neuromuscular junction, pathogenesis of LEMS, and activity at the neuromuscular junction after LEMS treatment
Image by Lecturio.Muscle weakness:
Bulbar involvement:
Autonomic dysfunction Autonomic Dysfunction Anterior Cord Syndrome:
Respiratory failure Respiratory failure Respiratory failure is a syndrome that develops when the respiratory system is unable to maintain oxygenation and/or ventilation. Respiratory failure may be acute or chronic and is classified as hypoxemic, hypercapnic, or a combination of the two. Respiratory Failure:
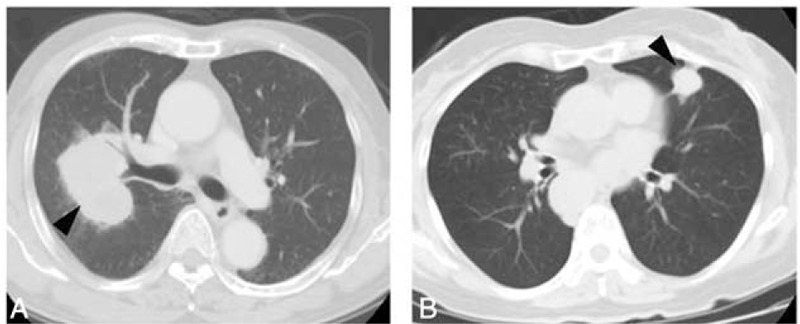
CT scan of the thorax showing central (A) and peripheral (B) small cell lung carcinoma (black arrow)
Image: “Distinct Characteristics of Small Cell Lung Cancer Correlate With Central or Peripheral Origin: Subtyping Based on Location and Expression of Transcription Factor TTF-1” by Miyauchi, E. et al. License: CC BY 4.0General:
Pharmacotherapy:
Immunotherapy:
Surgery: Where possible, tumor Tumor Inflammation resection can be considered and produce positive results (small number of cases).
Paraneoplastic LEMS:
Nonparaneoplastic LEMS: