In order to cope with their environment, cells undergo structural and functional changes. These cellular adaptations are reversible responses that allow cells to survive and continue to adequately function. Adaptive processes consist of increased cellular size and function (hypertrophy), increase in cell number (hyperplasia), decrease in cell size and metabolic activity (atrophy), or a change in the phenotype Phenotype The complete genetic complement contained in the DNA of a set of chromosomes in a human. The length of the human genome is about 3 billion base pairs. Basic Terms of Genetics of the cells (metaplasia). If the stress or stimulus is removed, the cell can return to its original state. However, when the limits of the adaptive responses are exceeded, cellular function is adversely affected, leading to cellular injury.
Last updated: May 17, 2024
Adaptation:
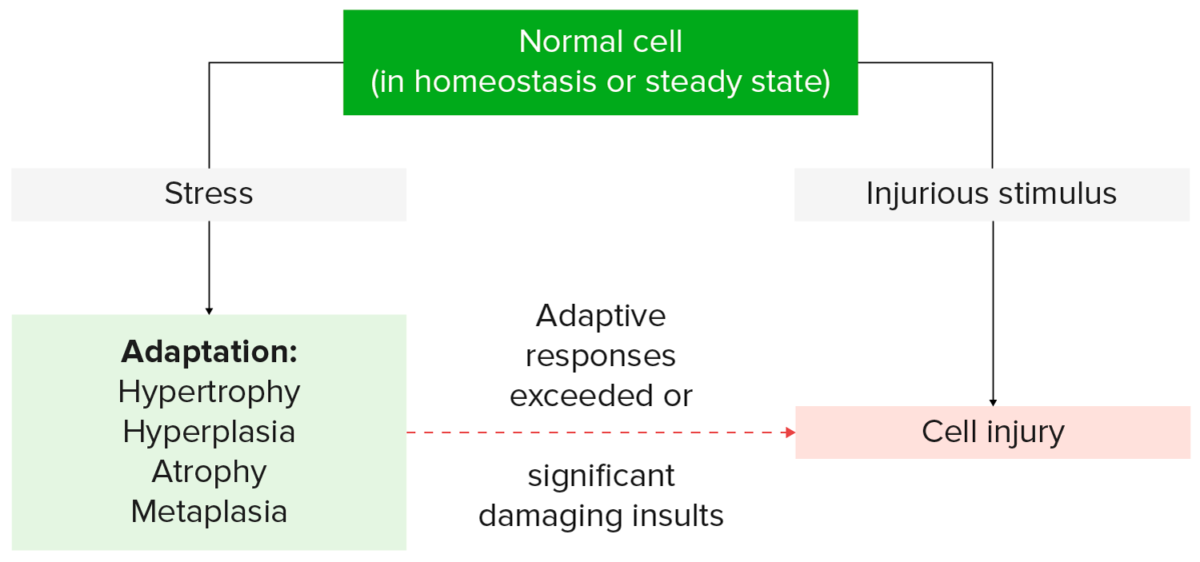
Cellular response to stress and stimuli
Image by Lecturio.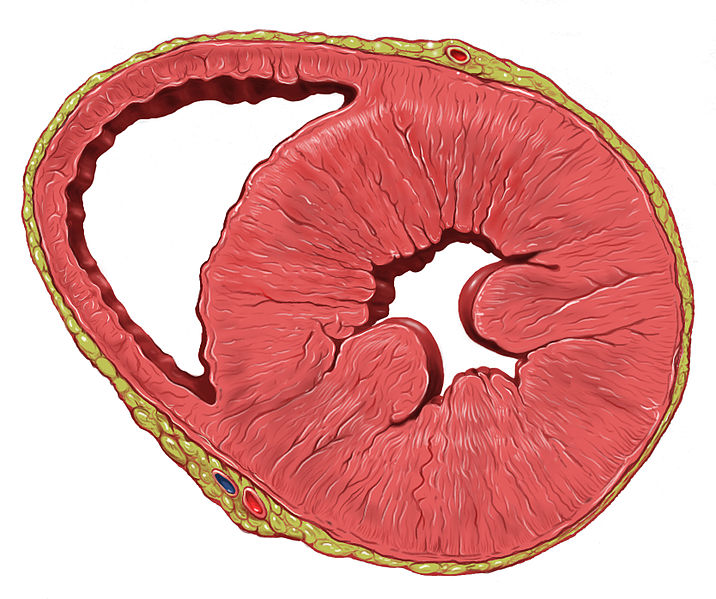
Cross-section of a heart with left ventricular hypertrophy: Note the thickened ventricular walls and associated reduced ventricular cavity.
Image: “Heart left ventricular hypertrophy short axis view” by Patrick J. Lynch, medical illustrator. License: CC BY 2.5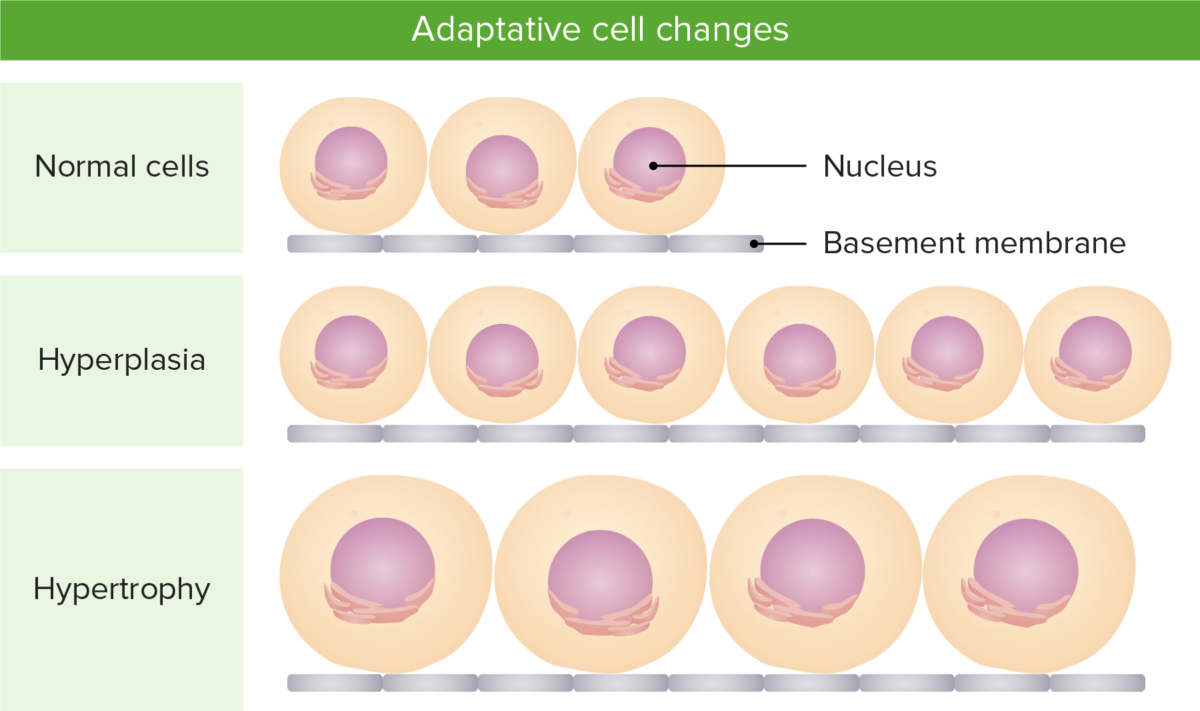
Illustration of the difference between hypertrophy (increase in size of cells) and hyperplasia (increase in number of cells)
Image by Lecturio.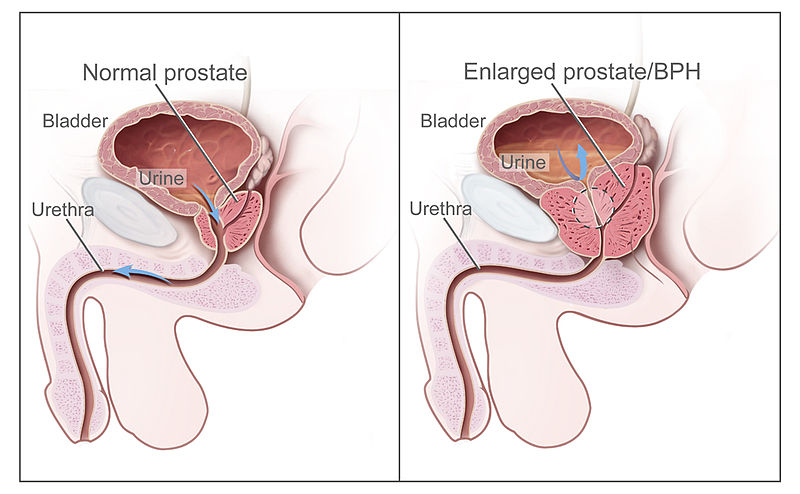
Normal prostate (left) and an enlarged prostate or benign prostatic hyperplasia (right), which is associated with bladder outlet obstruction
Image: “Benign Prostatic Hyperplasia” by National Cancer Institute. License: Public Domain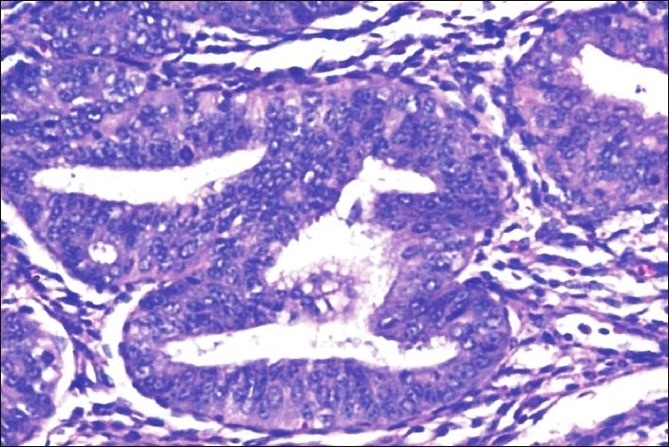
Histopathology of complex hyperplasia with atypia: closely packed endometrial glands with sparse intervening stroma and stratification of the lining epithelium. Epithelial cells show cytological atypia with high nucleocytoplasmic ratio, irregular clumping of nuclear chromatin, and mitotic figures (hematoxylin and eosin stain, × 200).
Image: “Histopathology of complex hyperplasia with atypia” by Shalinee Rao. License: CC BY 2.0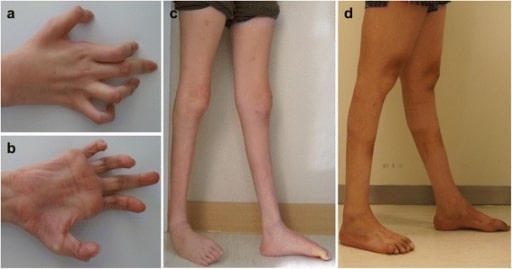
Photos showing the hands and legs of patients with axonal sensorimotor polyneuropathy:
Hands, patient 1 (a) and 2 (b). Right hand shows severe, atrophied, intrinsic muscle and flexion deformities of interphalangeal joints.
Lower extremities, patient 1 (c) and 2 (d). Severe bilateral distal muscle atrophies with ankle joint deformities are observed in both patients.
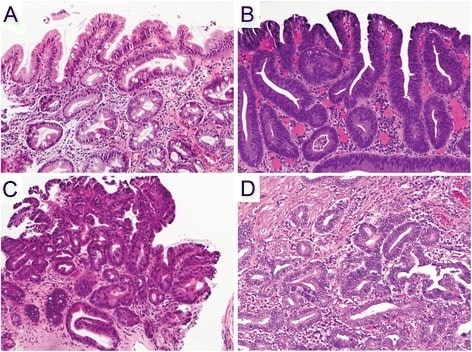
Hematoxylin and eosin staining of the histopathology of Barrett’s esophagus, dysplasia, and adenocarcinoma
A. Non-dysplastic Barrett’s mucosa (columnar cells) characterized by uniform, bland nuclei arranged in a surface monolayer
B. Low-grade dysplasia (nuclear hyperchromasia, elongation, and stratification extending up to the surface epithelium)
C. High-grade dysplasia (increased architectural and cytologic complexity)
D. Intramucosal adenocarcinoma (severe architectural distortion, including angulated glands).