Benign Benign Fibroadenoma breast epithelial lesions are grouped histologically as nonproliferative, proliferative without atypia Atypia Fibrocystic Change, and atypical hyperplasia Hyperplasia An increase in the number of cells in a tissue or organ without tumor formation. It differs from hypertrophy, which is an increase in bulk without an increase in the number of cells. Cellular Adaptation. The classifications are based on subsequent cancer risk in either breast. The nonproliferative type carries no risk, while fibroadenoma Fibroadenoma Fibroadenomas are the most common benign tumor of the female breast and the most common breast tumor in adolescent and young women. The tumors are well-circumscribed, mobile, and unencapsulated, with a rubbery or firm consistency. Fibroadenoma, the most common benign Benign Fibroadenoma tumor Tumor Inflammation, is a proliferative breast lesion (i.e., may have a slight increase in malignancy Malignancy Hemothorax risk if complex). Because atypical hyperplasia Hyperplasia An increase in the number of cells in a tissue or organ without tumor formation. It differs from hypertrophy, which is an increase in bulk without an increase in the number of cells. Cellular Adaptation shares some features with breast carcinoma in situ Carcinoma in situ A lesion with cytological characteristics associated with invasive carcinoma but the tumor cells are confined to the epithelium of origin, without invasion of the basement membrane. Leukoplakia, future cancer potential is increased. Management ranges from frequent monitoring to surgical excision, depending on certain factors, including the inherent risk of the pathologic diagnosis. Other breast disorders without malignant possibility are associated with underlying infection or systemic disease, so treatment differs. Benign Benign Fibroadenoma breast diseases are common but present diversely. It is important to distinguish between them to determine the likelihood of cancer and the best course of treatment.
Last updated: Dec 15, 2025
Nonproliferative breast lesions are conditions generally not associated with an increased risk of breast cancer Breast cancer Breast cancer is a disease characterized by malignant transformation of the epithelial cells of the breast. Breast cancer is the most common form of cancer and 2nd most common cause of cancer-related death among women. Breast Cancer.
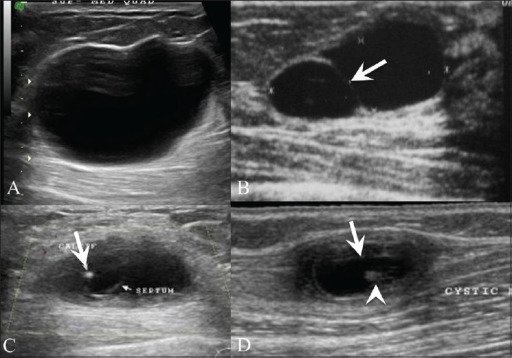
Breast US shows a simple cyst (A) presenting as an anechoic lesion with posterior enhancement and a cyst (B) with a septum (arrow). Breast US (C) in a 26-year-old woman with a painless palpable lump in her left breast shows a complex cyst with an eccentrically placed echogenic focus (arrow) representing the scolex of a cysticercus granuloma. The patient also had a similar swelling in the right upper arm, a US (D) of which revealed a cyst (arrow) with an echogenic scolex (arrowhead), within.
Image: “Breast USG” by Advanced Radiology Centre, Mumbai, India. License: CC BY 2.0In proliferative breast lesions without atypia Atypia Fibrocystic Change, the development of breast cancer Breast cancer Breast cancer is a disease characterized by malignant transformation of the epithelial cells of the breast. Breast cancer is the most common form of cancer and 2nd most common cause of cancer-related death among women. Breast Cancer is 1.5–2 times the risk of the general population.
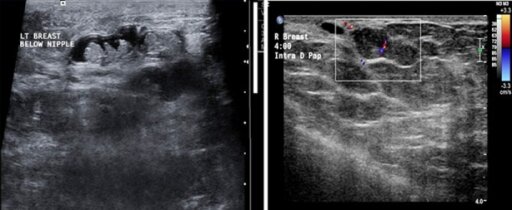
A: Multiple intraductal papillomas within the same dilated duct.
B: A papilloma arising from the wall of the duct, nearly filling the lumen of the duct. A vascular stalk is seen.
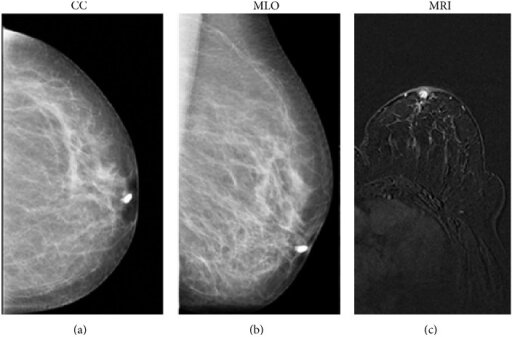
Craniocaudal (a) and mediolateral oblique (b) mammographic images show the filling stops a few millimeters from the nipple of the cannulated duct; imaging finding is suggestive of papilloma.
Magnetic resonance imaging (MRI) (T1-weighted 3D flash acquired in the axial plane) shows a mass intraductal enhancement in the retroareolar area (c) with round and sharp margins, indicative of a benign proliferation of the ductal epithelium (papilloma). Histology later confirmed the papilloma diagnosis.
Image: “Galactography” by Department of Radiological Sciences, Sapienza University of Rome, Umberto I Hospital, Viale Regina Elena 324, 00161 Rome, Italy. License: CC BY 3.0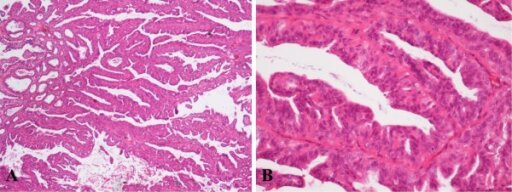
Histological appearance of Intraductal papilloma (IDP) of the left breast in low-power field, ×40 (A). Two-cell pattern lined by luminal cuboidal cells and a distinct outer layer of myoepithelial cells under higher magnification, ×200 (B). Image: “Histological appearance of IDP” by Institute for Oncology and Radiology of Serbia, 14 Pasterova, Belgrade 11000, Serbia. License: CC BY 2.0
Intraductal papillomas
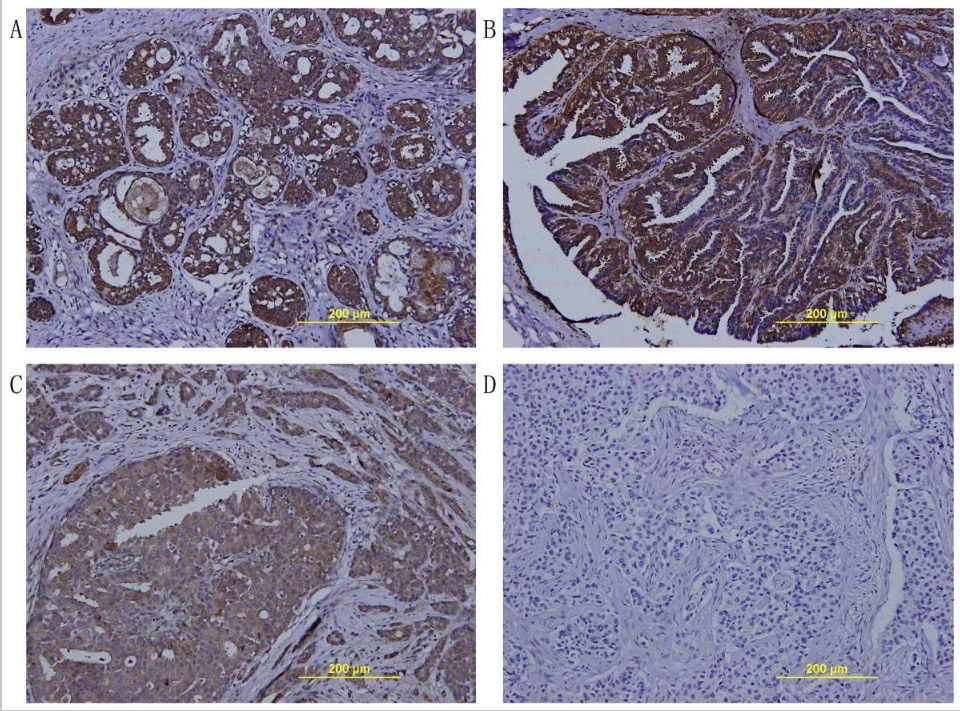
Image: “Immunohistochemical staining for TFPI-2 expression in breast tumors” by Department of Breast Surgery, Yangpu Hospital, Tongji University, Shanghai 200090, China. License: CC BY 2.0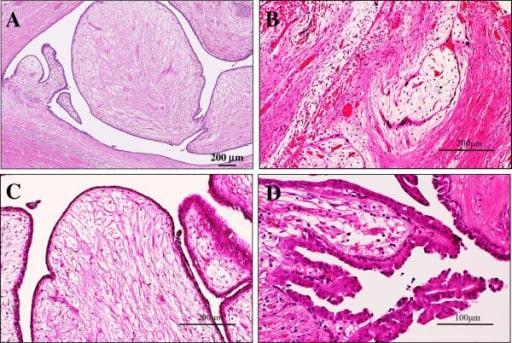
Microscopic examination of intraductal fibroadenoma or intraductal phyllodes tumour of the breast
(A) Low power view showed that its polypoid parts were composed of leaf-like processes with a hypocellular and prominent myxoid stroma, protruding into cystic spaces, reminiscent of intracanalicular type fibroadenoma or benign phyllodes tumour features (H&E stains). Bar = 200 μm.
(B) In some areas, there were foci of typical intracanalicular variant of fibroadenoma, in which duct lumens were compressed by the proliferating myxoid stroma (H&E stains). Bar = 200 μm.
(C) On high-power view, those stromal cells showed no significant atypia, but the covering hyperplastic epithelial components were also bland-looking in 2 cell layers. Mitotic figures were very rarely seen (H&E stains). Bar = 200 μm.
(D) In others, tiny foci of benign intraductal papilloma with characteristic delicate fibrovascular stalks were rarely seen (H&E stains). Bar = 100 μm.
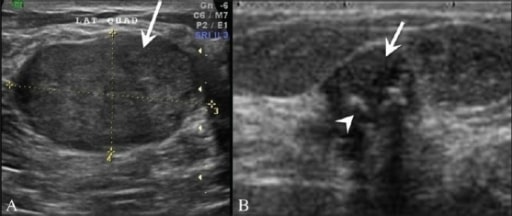
A: Breast ultrasonography in a 25-year-old patient shows a homogenous, hypoechoic, gently lobulated lesion (arrow), suggestive of a fibroadenoma.
B: Ultrasonography shows a degenerating fibroadenoma (arrow) with coarse calcifications (arrowhead) and posterior shadowing from the calcific foci.
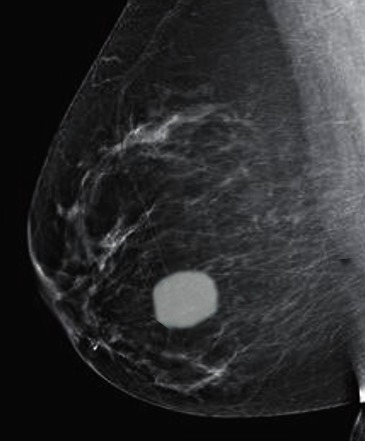
Fibroadenoma on a mammogram
Image: “Fibroadenoma on mammogram” by S. Bhimji MD. License: CC BY 4.0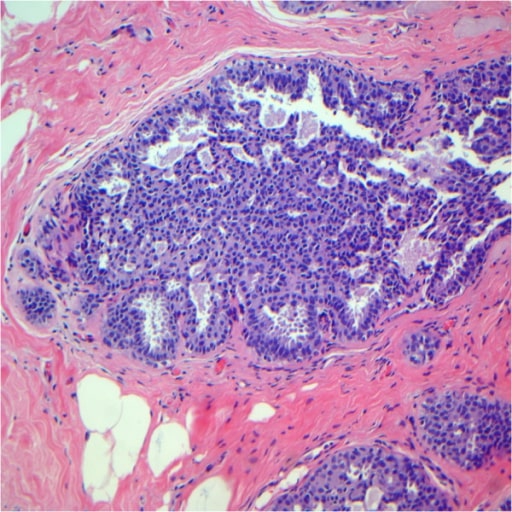
Usual ductal hyperplasia after review. Note the epithelial cells displaying a haphazard orientation and the presence of slit-like secondary lumina peripherally located.
Image: “Usual ductal hyperplasia” by Breast Pathology Laboratory, School of Medicine, Federal University of Minas Gerais (UFMG), Av, Professor Alfredo Balena, 190, Belo Horizonte, Minas Gerais 30130-100, Brazil. License: CC BY 2.0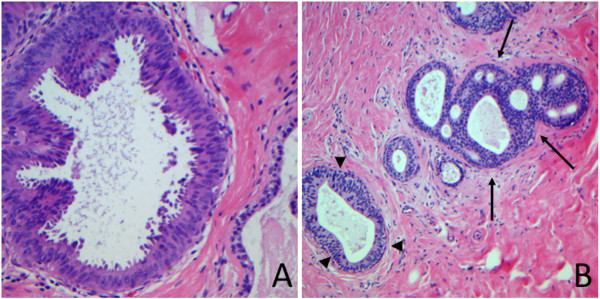
Atypical ductal hyperplasia (arrows)
Image: “Atypical ductal hyperplasia” by Breast Pathology Laboratory, School of Medicine, Federal University of Minas Gerais (UFMG), Av, Professor Alfredo Balena, 190, Belo Horizonte, Minas Gerais 30130-100, Brazil. License: CC BY 2.0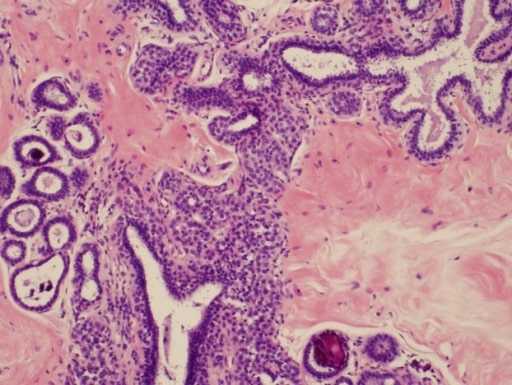
Photomicrograph of atypical lobular hyperplasia associated with columnar cell changes and incidental to targeted microcalcifications
Image: “Atypical lobular hyperplasia” by Department of Pathology, The University of Texas MD Anderson Cancer Center, Houston, Texas. License: CC BY 3.0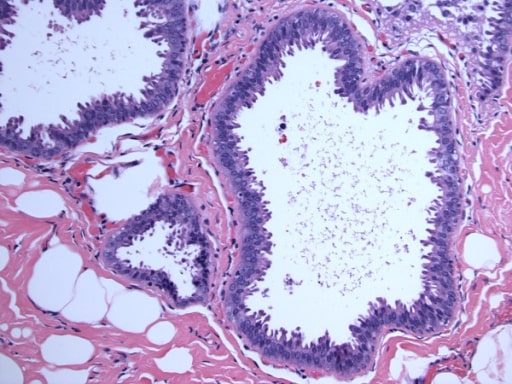
Columnar cell lesions with atypia (flat epithelial atypia)
Image: “Flat epithelial atypia” by Department of Pathology, Wilford Hall Medical Center, Lackland AFB, TX, USA. License: CC BY 2.0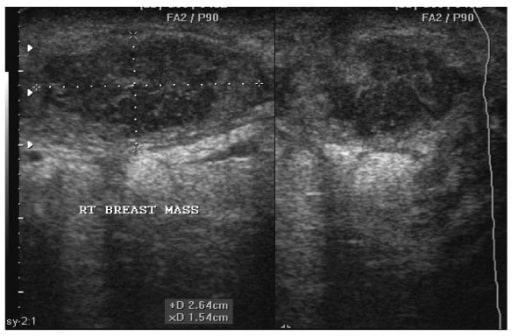
Sonogram of a 22-year-old woman showing a right breast abscess. Note the oval shape of the abscess, which measures 2.64 cm by 1.54 cm before aspiration under ultrasound guidance.
Image: “Breast abscess” by Department of Surgery, Weil Bugando University College of Health Sciences, Mwanza, Tanzania. License: CC BY 2.0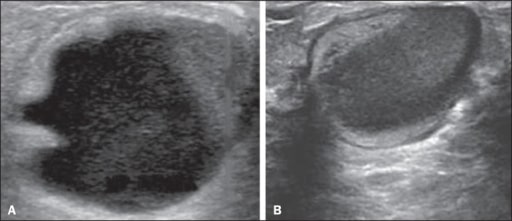
Galactocele:
A: Lesion parallel to the skin with well-defined borders showing anechoic (cystic) and echogenic (solid) components, with discrete posterior acoustic enhancement and well-defined borders.
B: Predominantly hypoechoic lesion parallel to the skin with well-defined borders, peripheral areas of hyperechogenicity, and posterior acoustic enhancement.
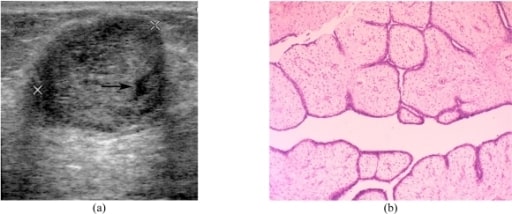
Phyllodes tumor of the breast
(a) Transverse US image shows a circumscribed heterogenous echo with a small cystic space (arrow) and a slight posterior acoustic enhancement.
(b) Photomicrograph shows leaf-like processes containing cellular stroma lined with benign ductal epithelial cells projecting into the cystic space (haematoxylin & eosin stain; x100).