Ependymomas are glial cell tumors arising from CSF-producing ependymal cells Ependymal Cells The macroglial cells of ependyma. They are characterized by bipolar cell body shape and processes that contact basal lamina around blood vessels and/or the pia mater and the cerebral ventricles. Muller cells of the retina are included based on similar microenvironmental contacts and morphology. Nervous System: Histology lining the ventricular system Ventricular System The ventricular system is an extension of the subarachnoid space into the brain consisting of a series of interconnecting spaces and channels. Four chambers are filled with cerebrospinal fluid (CSF): the paired lateral ventricles, the unpaired 3rd ventricle, and the unpaired 4th ventricle. Ventricular System: Anatomy. Ependymomas most commonly occur within the posterior fossa in contact with the 4th ventricle, or within the intramedullary spinal cord Spinal cord The spinal cord is the major conduction pathway connecting the brain to the body; it is part of the CNS. In cross section, the spinal cord is divided into an H-shaped area of gray matter (consisting of synapsing neuronal cell bodies) and a surrounding area of white matter (consisting of ascending and descending tracts of myelinated axons). Spinal Cord: Anatomy. The clinical presentation of ependymomas varies depending on the location of the tumor Tumor Inflammation. Magnetic resonance imaging is the imaging modality of choice, but histologic confirmation is required for diagnosis. The mainstay of treatment for intracranial ependymoma is surgical resection and adjuvant Adjuvant Substances that augment, stimulate, activate, potentiate, or modulate the immune response at either the cellular or humoral level. The classical agents (freund's adjuvant, bcg, corynebacterium parvum, et al.) contain bacterial antigens. Some are endogenous (e.g., histamine, interferon, transfer factor, tuftsin, interleukin-1). Their mode of action is either non-specific, resulting in increased immune responsiveness to a wide variety of antigens, or antigen-specific, i.e., affecting a restricted type of immune response to a narrow group of antigens. The therapeutic efficacy of many biological response modifiers is related to their antigen-specific immunoadjuvanticity. Vaccination radiation Radiation Emission or propagation of acoustic waves (sound), electromagnetic energy waves (such as light; radio waves; gamma rays; or x-rays), or a stream of subatomic particles (such as electrons; neutrons; protons; or alpha particles). Osteosarcoma therapy; young patients Patients Individuals participating in the health care system for the purpose of receiving therapeutic, diagnostic, or preventive procedures. Clinician–Patient Relationship receive chemotherapy Chemotherapy Osteosarcoma instead. Spinal cord Spinal cord The spinal cord is the major conduction pathway connecting the brain to the body; it is part of the CNS. In cross section, the spinal cord is divided into an H-shaped area of gray matter (consisting of synapsing neuronal cell bodies) and a surrounding area of white matter (consisting of ascending and descending tracts of myelinated axons). Spinal Cord: Anatomy ependymomas are treated with maximal surgical resection.
Last updated: Mar 25, 2025
Ependymomas are glial cell tumors in the CNS arising from ependymal cells Ependymal Cells The macroglial cells of ependyma. They are characterized by bipolar cell body shape and processes that contact basal lamina around blood vessels and/or the pia mater and the cerebral ventricles. Muller cells of the retina are included based on similar microenvironmental contacts and morphology. Nervous System: Histology.
| Categories | Specific tumors |
|---|---|
| Neuroepithelial tumors in the CNS |
|
| Meningeal tumors |
|
| Sellar-region tumors |
|
| Primary CNS lymphoma Lymphoma A general term for various neoplastic diseases of the lymphoid tissue. Imaging of the Mediastinum | Primary CNS lymphoma Lymphoma A general term for various neoplastic diseases of the lymphoid tissue. Imaging of the Mediastinum |
| Metastasis Metastasis The transfer of a neoplasm from one organ or part of the body to another remote from the primary site. Grading, Staging, and Metastasis to the brain Brain The part of central nervous system that is contained within the skull (cranium). Arising from the neural tube, the embryonic brain is comprised of three major parts including prosencephalon (the forebrain); mesencephalon (the midbrain); and rhombencephalon (the hindbrain). The developed brain consists of cerebrum; cerebellum; and other structures in the brain stem. Nervous System: Anatomy, Structure, and Classification (5x more common than primary brain Brain The part of central nervous system that is contained within the skull (cranium). Arising from the neural tube, the embryonic brain is comprised of three major parts including prosencephalon (the forebrain); mesencephalon (the midbrain); and rhombencephalon (the hindbrain). The developed brain consists of cerebrum; cerebellum; and other structures in the brain stem. Nervous System: Anatomy, Structure, and Classification tumors) | Most commonly arising from: |
| Peripheral tumors |
|
Intracranial ependymoma:
Spinal ependymoma:
The clinical presentation depends on the location of the ependymoma and the age of the patient. Most patients Patients Individuals participating in the health care system for the purpose of receiving therapeutic, diagnostic, or preventive procedures. Clinician–Patient Relationship have symptoms for approximately 3–6 months prior to diagnosis.
Imaging is obtained 1st as part of the diagnostic work-up, but histologic evaluation is required for diagnosis. Imaging findings may suggest an ependymoma and guide surgery.
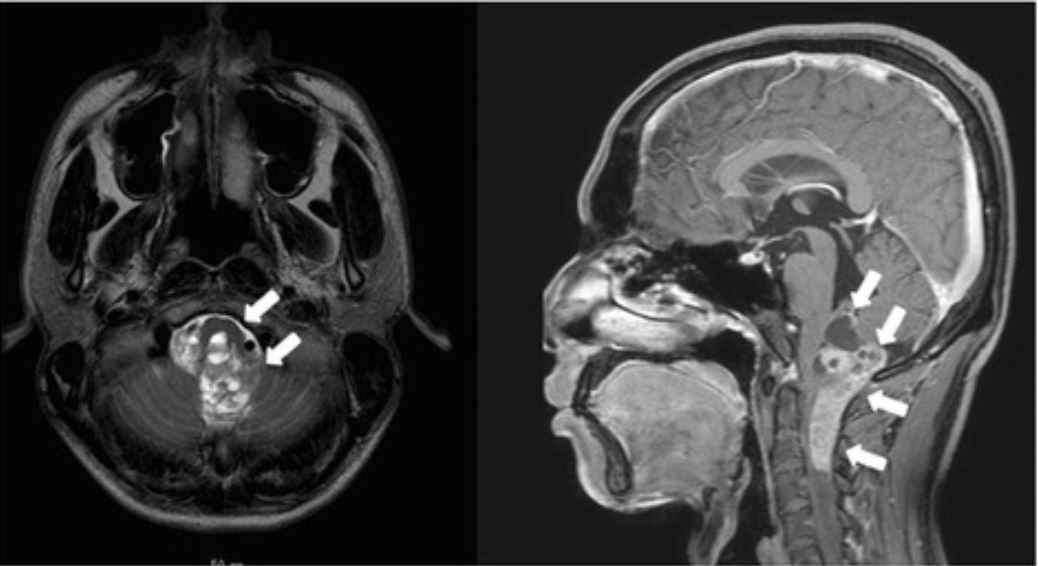
Magnetic resonance imaging scan reveals an anaplastic ependymoma extending from the brainstem to the 4th ventricle.
Image: “Giant duodenal ulcers after neurosurgery for brainstem tumors that required reoperation for gastric disconnection: a report of two cases” by BMC Surgery. License: CC BY 4.0Ependymoma is based on a histologic diagnosis; therefore, the gold standard for diagnosis is pathologic evaluation of a tissue specimen. Ependymomas are classified by histologic grades according to the WHO.
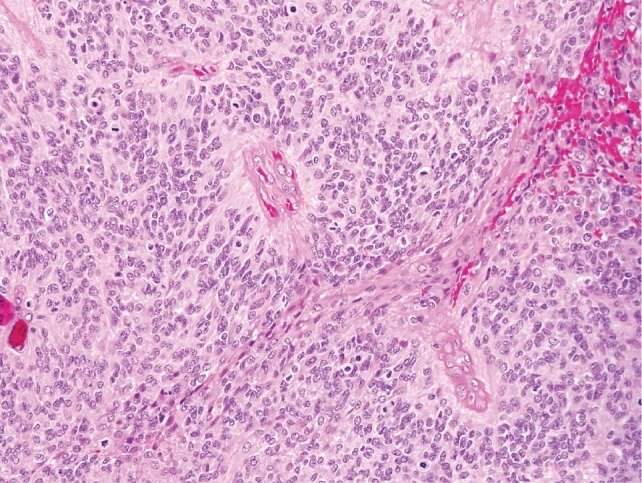
World Health Organization grade III intracranial ependymoma:
Perivascular pseudorosettes are seen as well as brisk, mitotic activity, which classifies the tumor as an anaplastic ependymoma.