Epidemiologic data obtained by clinical studies allow researchers to determine the likelihood of developing a certain outcome of interest within a studied population. This likelihood, or risk, can be quantified through what are known as measures of risk, which are mathematical formulas derived from contingency tables. These measures of risk include absolute risk ( AR AR Aortic regurgitation (AR) is a cardiac condition characterized by the backflow of blood from the aorta to the left ventricle during diastole. Aortic regurgitation is associated with an abnormal aortic valve and/or aortic root stemming from multiple causes, commonly rheumatic heart disease as well as congenital and degenerative valvular disorders. Aortic Regurgitation), relative risk (RR), attributable risk, and odds ratios (OR), each of which offers different kinds of information according to the needs of the researchers.
Last updated: Dec 15, 2025
A contingency table lists the frequency distributions of variables from a study and is a convenient way to look at any relationships between variables.
Below is an example of a 2×2 contingency table. The cells show the frequencies of distribution (A, B, C, D) for different combinations of the two variables (outcome, exposure), for a population of size N.
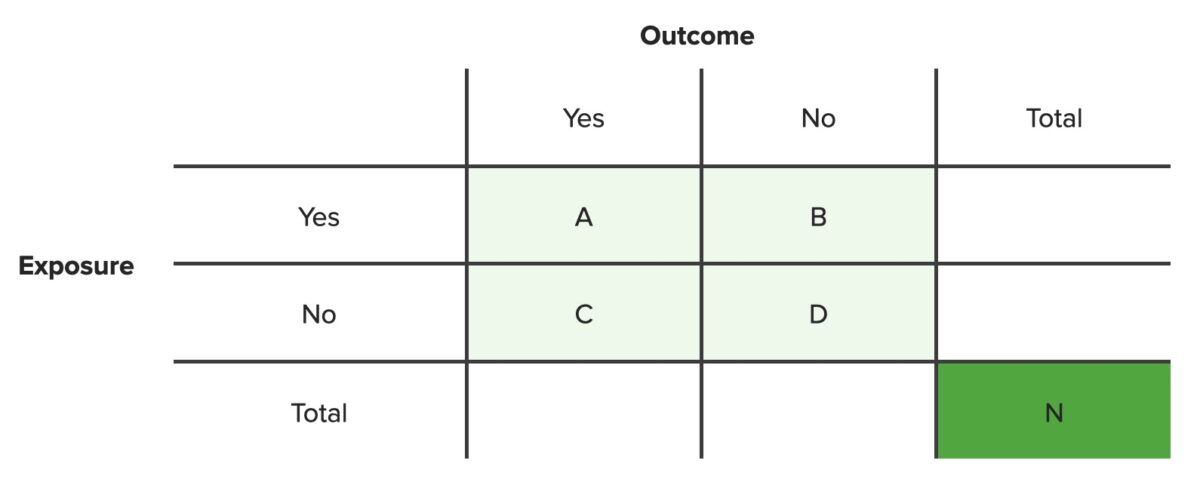
Contingency table
N = total number of people in set population
The AR AR Aortic regurgitation (AR) is a cardiac condition characterized by the backflow of blood from the aorta to the left ventricle during diastole. Aortic regurgitation is associated with an abnormal aortic valve and/or aortic root stemming from multiple causes, commonly rheumatic heart disease as well as congenital and degenerative valvular disorders. Aortic Regurgitation is the risk of developing a disease or condition after an exposure.
The AR AR Aortic regurgitation (AR) is a cardiac condition characterized by the backflow of blood from the aorta to the left ventricle during diastole. Aortic regurgitation is associated with an abnormal aortic valve and/or aortic root stemming from multiple causes, commonly rheumatic heart disease as well as congenital and degenerative valvular disorders. Aortic Regurgitation is calculated as the number of people who have a particular outcome divided by the total number of people with the same exposure (or the same nonexposure). This risk can be calculated for both exposed and unexposed populations.
Steps:
Start by setting up a contingency table:

Contingency table
N = total number of people in set population
Using the contingency table, the AR AR Aortic regurgitation (AR) is a cardiac condition characterized by the backflow of blood from the aorta to the left ventricle during diastole. Aortic regurgitation is associated with an abnormal aortic valve and/or aortic root stemming from multiple causes, commonly rheumatic heart disease as well as congenital and degenerative valvular disorders. Aortic Regurgitation in the exposed group is calculated as:
$$ Absolute\ risk\ of\ the\ exposed\ group = \frac{A}{A + B} $$where A = a patient was exposed to the risk factor and developed the outcome and B = a patient was exposed to the risk factor but did not develop the outcome.
Using the contingency table, the AR AR Aortic regurgitation (AR) is a cardiac condition characterized by the backflow of blood from the aorta to the left ventricle during diastole. Aortic regurgitation is associated with an abnormal aortic valve and/or aortic root stemming from multiple causes, commonly rheumatic heart disease as well as congenital and degenerative valvular disorders. Aortic Regurgitation in the unexposed group is calculated as:
$$ Absolute\ risk\ of\ the\ unexposed\ group = \frac{C}{C + D} $$where C = a patient who was not exposed to the risk factor but developed the outcome anyway and D = a patient who was not exposed to the risk factor and did not develop the outcome.
Example 1: In a population of 100 smokers, 75 developed lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer and 25 did not. What is the AR AR Aortic regurgitation (AR) is a cardiac condition characterized by the backflow of blood from the aorta to the left ventricle during diastole. Aortic regurgitation is associated with an abnormal aortic valve and/or aortic root stemming from multiple causes, commonly rheumatic heart disease as well as congenital and degenerative valvular disorders. Aortic Regurgitation of developing lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer if you are a smoker?
Example 2: In a population of 100 nonsmokers, 10 developed lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer and 90 did not. What is the AR AR Aortic regurgitation (AR) is a cardiac condition characterized by the backflow of blood from the aorta to the left ventricle during diastole. Aortic regurgitation is associated with an abnormal aortic valve and/or aortic root stemming from multiple causes, commonly rheumatic heart disease as well as congenital and degenerative valvular disorders. Aortic Regurgitation of developing lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer if you are not a smoker?
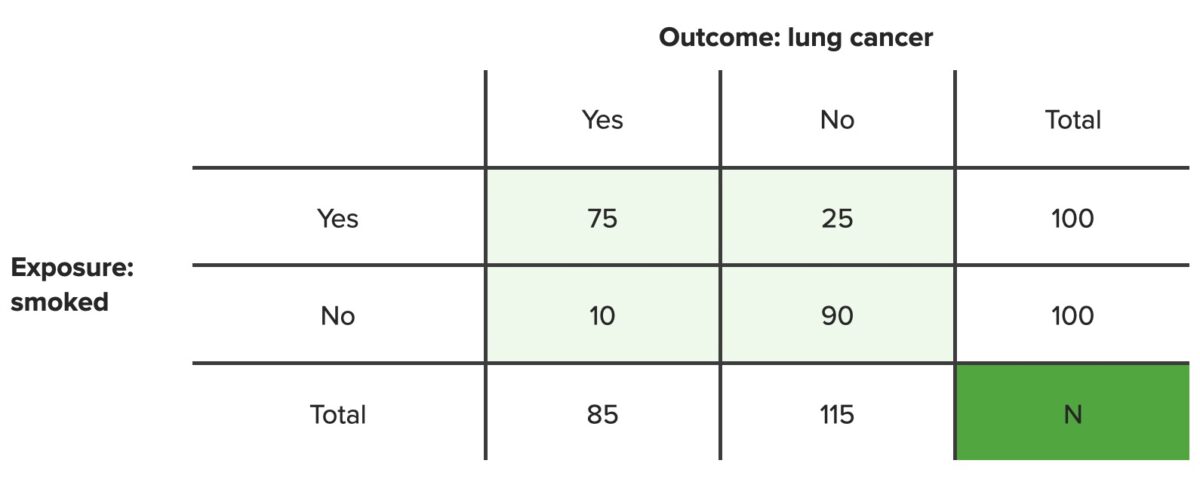
Contingency table
N = total number of people in set population
ARR or ARI is a measure of the reduction or increase in risk of developing a disease or condition as the result of an exposure.
Other ways to conceptualize ARR:
ARR can be interpreted as the health “gained” or ”lost” as a result of the exposure. For example, if you don’t smoke, by what percent can you reduce your risk of lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer?
The ARR between the exposed and unexposed groups can be calculated as:
$$ ARR = I_{Exposed} – I_{Unexposed} $$where I = incidence Incidence The number of new cases of a given disease during a given period in a specified population. It also is used for the rate at which new events occur in a defined population. It is differentiated from prevalence, which refers to all cases in the population at a given time. Measures of Disease Frequency rate. Because I is the same as AR AR Aortic regurgitation (AR) is a cardiac condition characterized by the backflow of blood from the aorta to the left ventricle during diastole. Aortic regurgitation is associated with an abnormal aortic valve and/or aortic root stemming from multiple causes, commonly rheumatic heart disease as well as congenital and degenerative valvular disorders. Aortic Regurgitation, this formula can be calculated from a contingency table:
$$ ARR = \frac{A}{A + B} – \frac{C}{C + D} $$In a population of 100 smokers, 75 developed lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer and 25 did not. In a population of 100 nonsmokers, 10 developed lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer and 90 did not. By how much does not smoking Smoking Willful or deliberate act of inhaling and exhaling smoke from burning substances or agents held by hand. Interstitial Lung Diseases reduce the AR AR Aortic regurgitation (AR) is a cardiac condition characterized by the backflow of blood from the aorta to the left ventricle during diastole. Aortic regurgitation is associated with an abnormal aortic valve and/or aortic root stemming from multiple causes, commonly rheumatic heart disease as well as congenital and degenerative valvular disorders. Aortic Regurgitation of developing lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer?

Contingency table
N = total number of people in set population
These are numbers typically reported when testing new therapeutic options. In these cases:
Number needed to treat Number needed to treat The number needed to treat (NNT) is the number of patients that are needed to treat to prevent 1 additional adverse outcome (e.g., stroke, death). For example, if a drug has an NNT of 10, it means 10 people must be treated with the drug to prevent 1 additional adverse outcome. Number Needed to Treat ( NNT NNT The number needed to treat (NNT) is the number of patients that are needed to treat to prevent 1 additional adverse outcome (e.g., stroke, death). For example, if a drug has an nnt of 10, it means 10 people must be treated with the drug to prevent 1 additional adverse outcome. Number Needed to Treat):
Number needed to harm Number needed to harm The NNH is the additional number of individuals who need to be exposed to risk (harmful exposure or treatment) to have 1 extra person develop the disease compared to that in the unexposed group. Number Needed to Treat ( NNH NNH The NNH is the additional number of individuals who need to be exposed to risk (harmful exposure or treatment) to have 1 extra person develop the disease compared to that in the unexposed group. Number Needed to Treat):
Relative risk (RR) is the risk of a disease or condition occurring in a group or population with a particular exposure relative to a control (unexposed) group.
RR is typically among the most important numbers calculated. Cohort studies Cohort studies Studies in which subsets of a defined population are identified. These groups may or may not be exposed to factors hypothesized to influence the probability of the occurrence of a particular disease or other outcome. Cohorts are defined populations which, as a whole, are followed in an attempt to determine distinguishing subgroup characteristics. Epidemiological Studies are the only type of observational study that can determine the RR.
The relative risk is calculated as the frequency of a disease or condition in the exposed group (IE) divided by the frequency in an unexposed control group (IO), which is represented by the formula:
$$ RR = \frac{I_{E}}{I_{O}} $$Again, since the incidence Incidence The number of new cases of a given disease during a given period in a specified population. It also is used for the rate at which new events occur in a defined population. It is differentiated from prevalence, which refers to all cases in the population at a given time. Measures of Disease Frequency rates are the same as the AR AR Aortic regurgitation (AR) is a cardiac condition characterized by the backflow of blood from the aorta to the left ventricle during diastole. Aortic regurgitation is associated with an abnormal aortic valve and/or aortic root stemming from multiple causes, commonly rheumatic heart disease as well as congenital and degenerative valvular disorders. Aortic Regurgitation, the RR can be calculated from a contingency table by using the following expanded formula:
$$ RR = \frac{\frac{A}{A + B}}{\frac{C}{C + D}} $$In a population of 100 smokers, 75 developed lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer and 25 did not. In a population of 100 nonsmokers, 10 developed lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer and 90 did not. What is the risk of getting lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer if you smoke compared to the risk of getting lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer if you do not smoke?

Contingency table
N = total number of people in set population
Definition:
Calculation:
The RRR is calculated as the difference between the incidence Incidence The number of new cases of a given disease during a given period in a specified population. It also is used for the rate at which new events occur in a defined population. It is differentiated from prevalence, which refers to all cases in the population at a given time. Measures of Disease Frequency of a disease in an exposed (IE) and an unexposed (IO ) group divided by the incidence Incidence The number of new cases of a given disease during a given period in a specified population. It also is used for the rate at which new events occur in a defined population. It is differentiated from prevalence, which refers to all cases in the population at a given time. Measures of Disease Frequency in the unexposed group, which is calculated with the following formula:
$$ RRR = \frac{ARR}{I_{O}} = (\frac{I_{E} – I_{O}}{I_{O}}) = \frac{\frac{A}{A + B} – \frac{C}{C + D}}{\frac{C}{C + D}} $$In a population of 100 smokers, 75 developed lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer and 25 did not after 10 years in a cohort study. In a population of 100 nonsmokers, 10 developed lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer and 90 did not. If people didn’t smoke, how much less lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer can be expected in the population?

Contingency table
N = total number of people in set population
The attributable risk is a measure of the risk of developing an outcome associated with a particular exposure.
The attributable risk in the exposed group is the difference in the rate of a disease between the exposed and the unexposed groups. For example, what percentage of
lung cancer
Lung cancer
Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort.
Lung Cancer cases are likely due to
smoking
Smoking
Willful or deliberate act of inhaling and exhaling smoke from burning substances or agents held by hand.
Interstitial Lung Diseases?
The attributable risk is calculated by subtracting the
incidence
Incidence
The number of new cases of a given disease during a given period in a specified population. It also is used for the rate at which new events occur in a defined population. It is differentiated from prevalence, which refers to all cases in the population at a given time.
Measures of Disease Frequency in the unexposed group (IO) from the
incidence
Incidence
The number of new cases of a given disease during a given period in a specified population. It also is used for the rate at which new events occur in a defined population. It is differentiated from prevalence, which refers to all cases in the population at a given time.
Measures of Disease Frequency of the exposed group (IE) and dividing by the
incidence
Incidence
The number of new cases of a given disease during a given period in a specified population. It also is used for the rate at which new events occur in a defined population. It is differentiated from prevalence, which refers to all cases in the population at a given time.
Measures of Disease Frequency in the exposed group, which is expressed as:
Example: In a population of 100 smokers, 75 developed lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer and 25 did not after 10 years in a cohort study. In a population of 100 nonsmokers, 10 developed lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer and 90 did not. What percentage of lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer cases are likely due to smoking Smoking Willful or deliberate act of inhaling and exhaling smoke from burning substances or agents held by hand. Interstitial Lung Diseases?

Contingency table
N = total number of people in set population
The PAR is the attributable risk for an entire population. It represents the fraction of cases that would not occur in a population if the exposure was eliminated.
For example, what percentage of lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer cases could be prevented if nobody smoked?
The PAR is calculated by subtracting the incidence Incidence The number of new cases of a given disease during a given period in a specified population. It also is used for the rate at which new events occur in a defined population. It is differentiated from prevalence, which refers to all cases in the population at a given time. Measures of Disease Frequency rate in the unexposed population from the incidence Incidence The number of new cases of a given disease during a given period in a specified population. It also is used for the rate at which new events occur in a defined population. It is differentiated from prevalence, which refers to all cases in the population at a given time. Measures of Disease Frequency rate in the entire population:
$$ PAR = \frac{(I_{T} – I_{O})}{I_{T}} = \frac{(\frac{A + C}{N} – \frac{C}{C + D})}{\frac{A + C}{N}} $$Example: In a population of 100 smokers, 75 developed lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer and 25 did not after 10 years in a cohort study. In a population of 100 nonsmokers, 10 developed lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer and 90 did not. What percentage of lung cancer Lung cancer Lung cancer is the malignant transformation of lung tissue and the leading cause of cancer-related deaths. The majority of cases are associated with long-term smoking. The disease is generally classified histologically as either small cell lung cancer or non-small cell lung cancer. Symptoms include cough, dyspnea, weight loss, and chest discomfort. Lung Cancer cases could be prevented if nobody smoked?
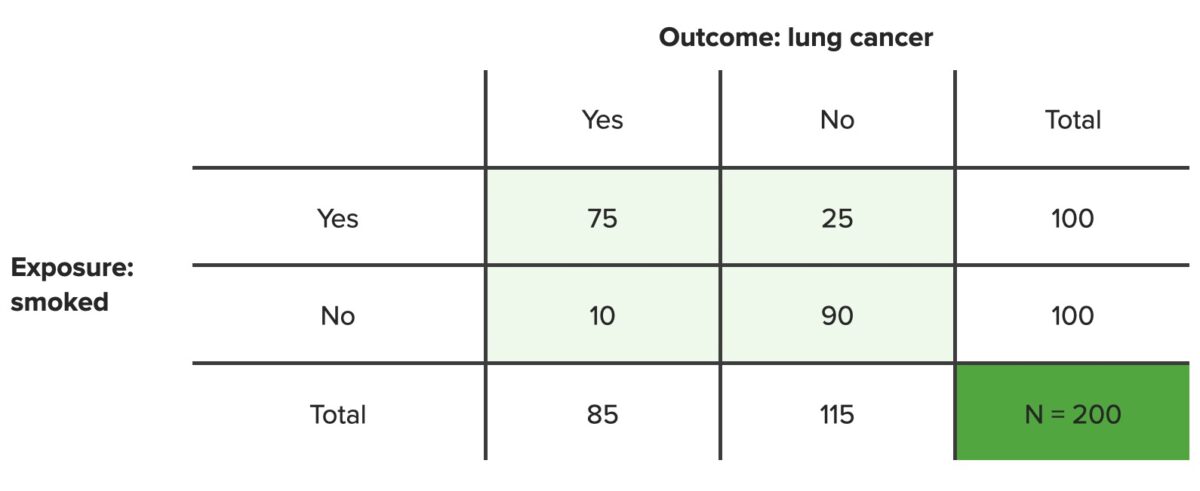
Contingency table
N = total number of people in set population
An odds ratio (OR) is a statistic that quantifies the strength of association between 2 variables or events.
An OR is used as an estimation of RR in case–control studies. OR is calculated by determining the odds of exposure among the diseased divided by the odds of exposure among the undiseased. This is represented as:
$$ OR = \frac{(Odds\ of\ exposure\ among\ diseased)}{(Odds\ of\ exposure\ among\ undiseased)} = \frac{A \div C}{B \div D} $$where (A ÷ C) represents the number of exposed cases divided by the number of unexposed cases among those with the disease and (B ÷ D) is the number of exposed undiseased divided by the number of unexposed undiseased.
Rearranging the formula gives the simplified equation:
$$ OR = ( AD AD The term advance directive (AD) refers to treatment preferences and/or the designation of a surrogate decision-maker in the event that a person becomes unable to make medical decisions on their own behalf. Advance directives represent the ethical principle of autonomy and may take the form of a living will, health care proxy, durable power of attorney for health care, and/or a physician’s order for life-sustaining treatment. Advance Directives) \div (BC) $$OR is interpreted in the same way as RR:
In this example, 6 people developed Creutzfeldt-Jakob disease Creutzfeldt-Jakob Disease A rare transmissible encephalopathy most prevalent between the ages of 50 and 70 years. Affected individuals may present with sleep disturbances, personality changes, ataxia; aphasia, visual loss, weakness, muscle atrophy, myoclonus, progressive dementia, and death within one year of disease onset. A familial form exhibiting autosomal dominant inheritance and a new variant cjd (potentially associated with bovine spongiform encephalopathy) have been described. Pathological features include prominent cerebellar and cerebral cortical spongiform degeneration and the presence of prions. Transmissible Spongiform Encephalopathies (CJD); 3 of them ate a significant amount of beef and 3 of them did not. These patients Patients Individuals participating in the health care system for the purpose of receiving therapeutic, diagnostic, or preventive procedures. Clinician–Patient Relationship were compared to a control population in a case–control study; in the control population of 10 people, 4 of them ate a significant amount of beef and 6 of them did not. What are the odds of developing CJD after eating a significant amount of beef?
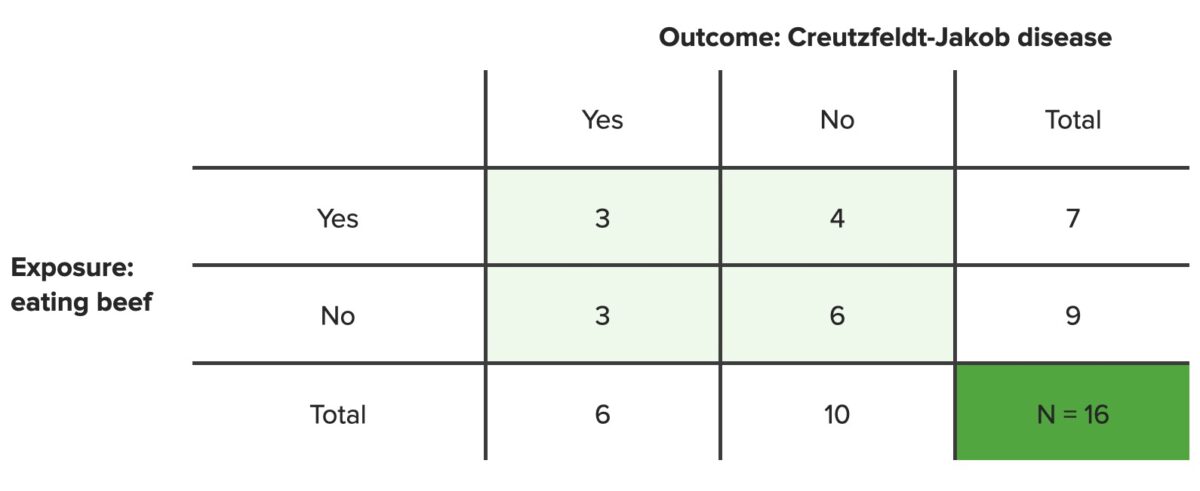
Contingency table
N = total number of people in set population
Note: These are hypothetical data; the true incidence rates of Creutzfeldt-Jakob disease are much lower than these data suggest.