Ehrlichiosis and anaplasmosis are tick-borne bacterial infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease. The most common causative species include Ehrlichia chaffeensis and Anaplasma phagocytophilum, which infect and multiply within monocytes Monocytes Large, phagocytic mononuclear leukocytes produced in the vertebrate bone marrow and released into the blood; contain a large, oval or somewhat indented nucleus surrounded by voluminous cytoplasm and numerous organelles. Innate Immunity: Phagocytes and Antigen Presentation and granulocytes Granulocytes Leukocytes with abundant granules in the cytoplasm. They are divided into three groups according to the staining properties of the granules: neutrophilic, eosinophilic, and basophilic. Mature granulocytes are the neutrophils; eosinophils; and basophils. White Myeloid Cells: Histology, respectively. The clinical presentation can vary widely, but often includes fever Fever Fever is defined as a measured body temperature of at least 38°C (100.4°F). Fever is caused by circulating endogenous and/or exogenous pyrogens that increase levels of prostaglandin E2 in the hypothalamus. Fever is commonly associated with chills, rigors, sweating, and flushing of the skin. Fever, malaise Malaise Tick-borne Encephalitis Virus, headache Headache The symptom of pain in the cranial region. It may be an isolated benign occurrence or manifestation of a wide variety of headache disorders. Brain Abscess, myalgia Myalgia Painful sensation in the muscles. Ion Channel Myopathy, and arthralgias. A maculopapular Maculopapular Dermatologic Examination or petechial rash Rash Rocky Mountain Spotted Fever occurs in some patients Patients Individuals participating in the health care system for the purpose of receiving therapeutic, diagnostic, or preventive procedures. Clinician–Patient Relationship. Gastrointestinal, neurologic, and respiratory symptoms are also possible. The diagnosis is based on clinical suspicion and confirmed PCR PCR Polymerase chain reaction (PCR) is a technique that amplifies DNA fragments exponentially for analysis. The process is highly specific, allowing for the targeting of specific genomic sequences, even with minuscule sample amounts. The PCR cycles multiple times through 3 phases: denaturation of the template DNA, annealing of a specific primer to the individual DNA strands, and synthesis/elongation of new DNA molecules. Polymerase Chain Reaction (PCR) or antibody testing. Management is with doxycycline.
Last updated: Oct 21, 2022
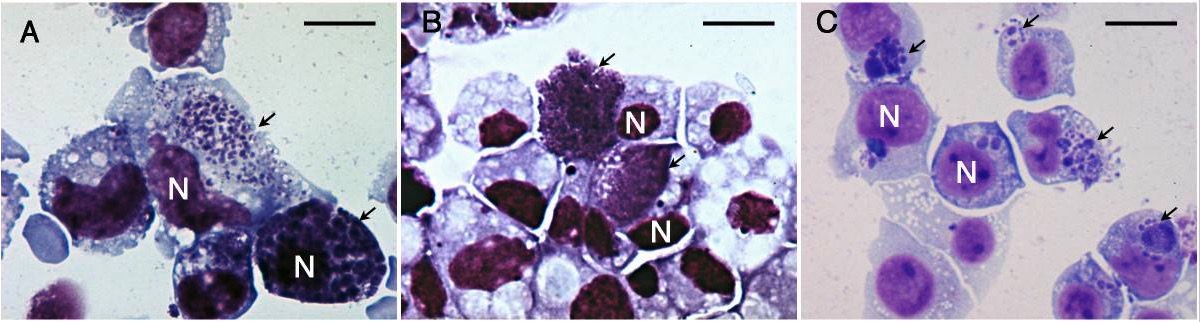
Microscopic images of Giemsa-stained cells infected with anaplasmosis:
Cell nuclei are labeled “N” and the arrows point to Anaplasma morulae.
The most notable species are:
Rare causes of ehrlichiosis in humans:
For ehrlichiosis/anaplasmosis:
For severe disease:

A Lone Star tick, which can transmit E. chaffeensis
Image: “Amblyomma americanum tick” by James Gathany. License: Public Domain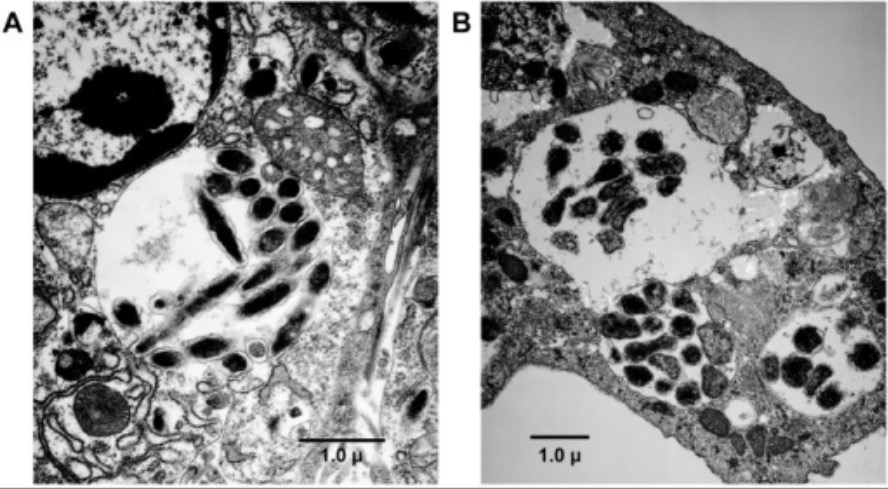
Transmission electron microscopy analysis of E. chaffeensis infected ticks:
E. chaffeensis organisms appear in cytoplasmic vacuoles of a tick’s midgut cell.
A wide range of wild and domestic animals Animals Unicellular or multicellular, heterotrophic organisms, that have sensation and the power of voluntary movement. Under the older five kingdom paradigm, animalia was one of the kingdoms. Under the modern three domain model, animalia represents one of the many groups in the domain eukaryota. Cell Types: Eukaryotic versus Prokaryotic can serve as reservoirs. The most notable are:
Human monocytic ehrlichiosis:
Human granulocytic anaplasmosis:
The incubation Incubation The amount time between exposure to an infectious agent and becoming symptomatic. Rabies Virus period is typically 1–2 weeks, and the clinical presentation can vary greatly.
Features of both HME and HGA:
Findings more commonly seen in HME:
Definitive studies:
Supporting laboratory studies:
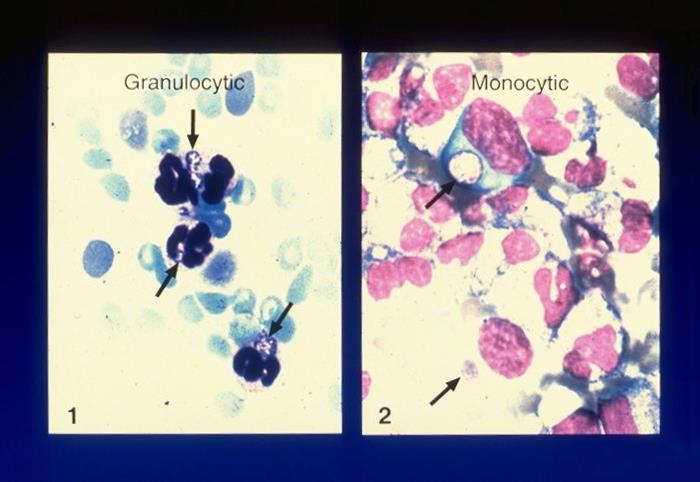
Peripheral blood smear evaluations for ehrlichiosis and anaplasmosis:
These blood smears show intracellular morulae of A. phagocytophilum (1) and E. chaffeensis (2).
Doxycycline is the antibiotic of choice.
Avoiding tick bites is key to preventing these diseases.
| Organism | Ehrlichia chaffeensis | Anaplasma phagocytophilum | Rickettsia rickettsii Rickettsia rickettsii A species of gram-negative, aerobic bacteria that is the etiologic agent of rocky mountain spotted fever. Its cells are slightly smaller and more uniform in size than those of rickettsia prowazekii. Rickettsia | Borrelia burgdorferi Borrelia burgdorferi A specific species of bacteria, part of the borrelia burgdorferi group, whose common name is lyme disease spirochete. Borrelia |
|---|---|---|---|---|
| Disease | HME | HGA | Rocky Mountain spotted fever Fever Fever is defined as a measured body temperature of at least 38°C (100.4°F). Fever is caused by circulating endogenous and/or exogenous pyrogens that increase levels of prostaglandin E2 in the hypothalamus. Fever is commonly associated with chills, rigors, sweating, and flushing of the skin. Fever | Lyme disease Lyme disease Lyme disease is a tick-borne infection caused by the gram-negative spirochete Borrelia burgdorferi. Lyme disease is transmitted by the black-legged Ixodes tick (known as a deer tick), which is only found in specific geographic regions. Patient presentation can vary depending on the stage of the disease and may include a characteristic erythema migrans rash. Lyme Disease |
| Micro |
|
|
|
|
| Vector | Lone Star tick Lone Star Tick Rocky Mountain Spotted Fever | Ixodes tick | Dermacentor Dermacentor A widely distributed genus of ticks, in the family ixodidae, including a number that infest humans and other mammals. Several are vectors of diseases such as tularemia; rocky mountain spotted fever; colorado tick fever; and anaplasmosis. Rickettsia tick | Ixodes tick |
| Reservoir Reservoir Animate or inanimate sources which normally harbor disease-causing organisms and thus serve as potential sources of disease outbreaks. Reservoirs are distinguished from vectors (disease vectors) and carriers, which are agents of disease transmission rather than continuing sources of potential disease outbreaks. Humans may serve both as disease reservoirs and carriers. Escherichia coli | White-tailed deer | White-footed mouse | Dermacentor Dermacentor A widely distributed genus of ticks, in the family ixodidae, including a number that infest humans and other mammals. Several are vectors of diseases such as tularemia; rocky mountain spotted fever; colorado tick fever; and anaplasmosis. Rickettsia tick |
|
| Geographical distribution in the United States | Southeast and South Central states | Northeast and upper Midwest states | Southeast and South Central states | Northeast and Midwest states |
| Diagnosis |
|
|
|
|
| Management | Doxycycline |
|
||