Aortic dissection occurs due to shearing stress from pulsatile pressure causing a tear in the tunica intima of the aortic wall. This tear allows blood to flow into the media, creating a “false lumen.” Aortic dissection is most commonly caused by uncontrolled hypertension. Complications arise due to partial occlusion of vital branches off the aorta and reduced blood flow to the brain, visceral organs, and extremities. Patients often present with acute, tearing chest or back pain. Computed tomography is the diagnostic modality of choice. All type A dissections (ascending aorta) are a surgical emergency due to the risk of imminent rupture. Type B dissections (descending aorta) can be managed medically with impulse control using beta-blockers and calcium channel blockers. If there is evidence of malperfusion to visceral organs or extremities, aneurysm dilation to > 5 cm, retrograde extension into the ascending aorta, or intractable pain, the patient will need evaluation for endovascular or open repair.
Last updated: May 16, 2024
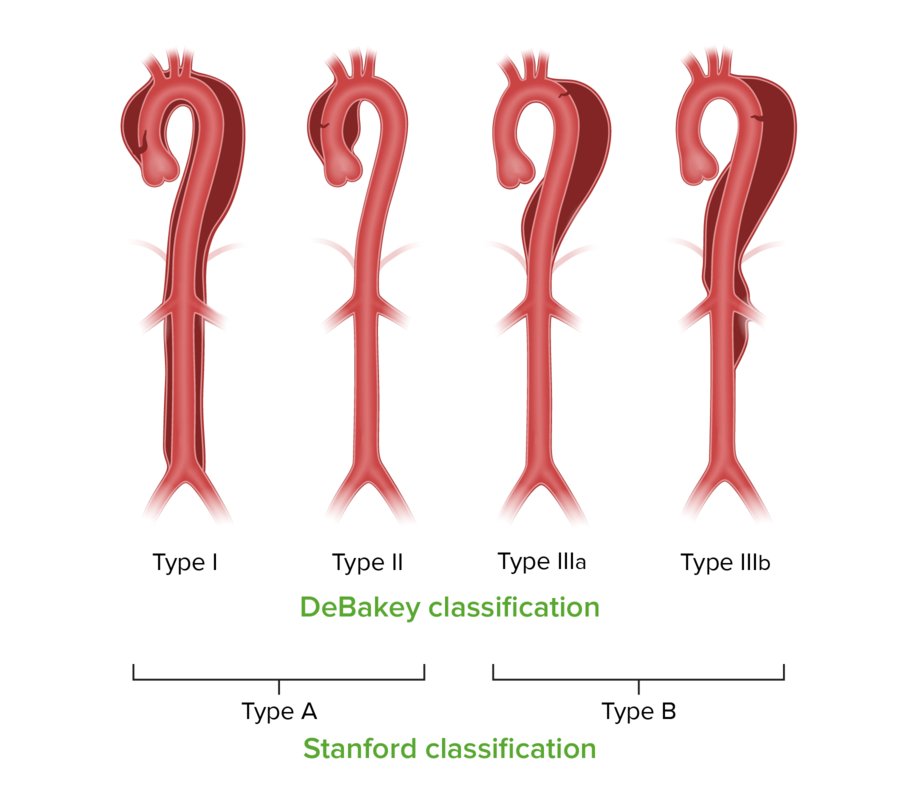
Stanford and DeBakey classifications for aortic dissection
Image by Lecturio.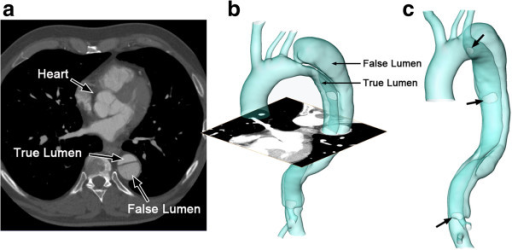
False versus true lumen in aortic dissection
The reconstructed computed tomography (CT) image of an aortic dissection:
1 axial slice of the CT scan (a); reconstructed surface of the aortic dissection (b); positions of the entries along the intimal flap (c; indicated by arrows).
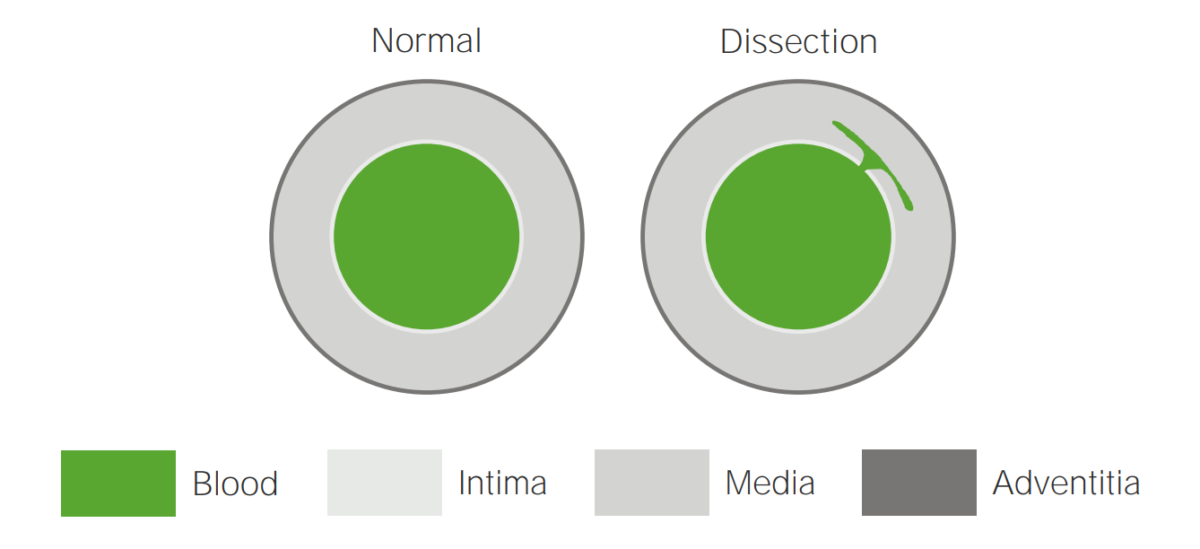
Illustration of an aortic dissection
A tear in the intima allows blood to flow into the media.

Mechanisms of aortic dissections
A tear in the intima, rupture of the vasa vasorum, and ulceration of an atherosclerotic plaque
TL: true lumen
FL: false lumen
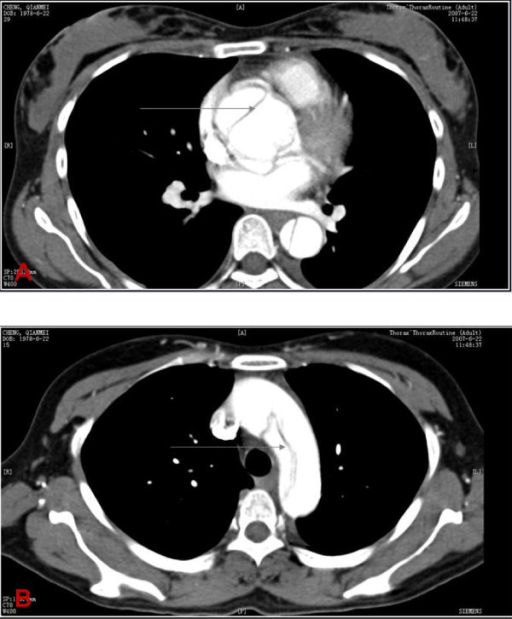
Computed tomography findings in aortic dissection
A: CT scan showing an ascending aortic dissection (gray arrow)
B: CT scan demonstrating an aortic arch dissection (gray arrow)
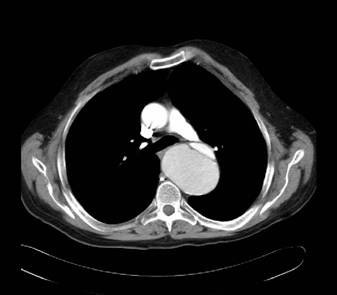
Computed tomography findings in aortic dissection
CT scan demonstrating the dissection at the level of the aortic valve
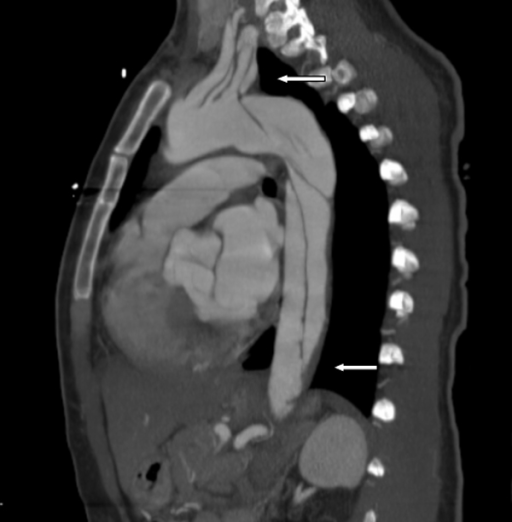
Computed tomography findings in aortic dissection
Aortic dissection extending into the left subclavian and axillary arteries, and to the level of bifurcation of the common iliac artery (thick arrows)
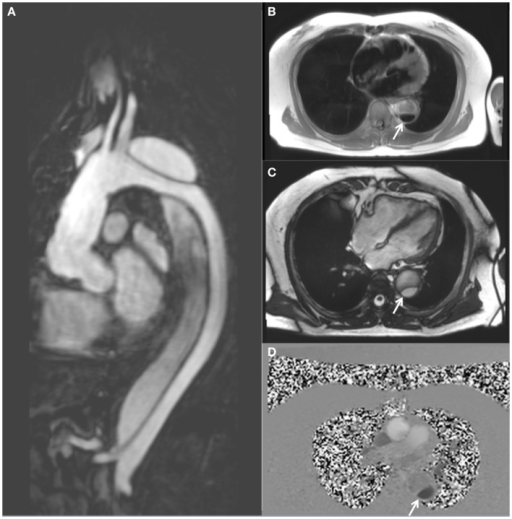
Magnetic resonance imaging demonstrating a descending thoracic aortic dissection:
A: sagittal gadolinium-contrast-enhanced MRA view
B: axial black blood view of the proximal descending thoracic aorta
C: axial true FISP (steady state-free precession) cine view
D: axial phase-contrast view, showing flow patterns in the true and false lumens of the descending aorta (the true lumen is indicated by the white arrow)
Type A dissections require emergent surgical intervention for replacement of the involved ascending aorta Ascending aorta Mediastinum and Great Vessels: Anatomy or the entire ascending aorta Ascending aorta Mediastinum and Great Vessels: Anatomy with Dacron graft Graft A piece of living tissue that is surgically transplanted Organ Transplantation.