Amebiasis, or amoebic dysentery Dysentery Acute inflammation of the intestine associated with infectious diarrhea of various etiologies, generally acquired by eating contaminated food containing toxins, biological derived from bacteria or other microorganisms. Dysentery is characterized initially by watery feces then by bloody mucoid stools. It is often associated with abdominal pain; fever; and dehydration. Gastroenteritis, is an infection caused by the parasite Entamoeba histolytica Entamoeba Histolytica A species of parasitic protozoa causing entamoebiasis and amebic dysentery (dysentery, amebic). Characteristics include a single nucleus containing a small central karyosome and peripheral chromatin that is finely and regularly beaded. Amebicides. Transmission is through the fecal-oral route Fecal-oral route Echovirus or by consumption of contaminated food and water. Most patients Patients Individuals participating in the health care system for the purpose of receiving therapeutic, diagnostic, or preventive procedures. Clinician–Patient Relationship infected with E. histolytica are asymptomatic, but about 10% may develop dysentery Dysentery Acute inflammation of the intestine associated with infectious diarrhea of various etiologies, generally acquired by eating contaminated food containing toxins, biological derived from bacteria or other microorganisms. Dysentery is characterized initially by watery feces then by bloody mucoid stools. It is often associated with abdominal pain; fever; and dehydration. Gastroenteritis. Invasive infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease are characterized by abdominal pain Abdominal Pain Acute Abdomen, fever Fever Fever is defined as a measured body temperature of at least 38°C (100.4°F). Fever is caused by circulating endogenous and/or exogenous pyrogens that increase levels of prostaglandin E2 in the hypothalamus. Fever is commonly associated with chills, rigors, sweating, and flushing of the skin. Fever, and bloody diarrhea Bloody diarrhea Diarrhea, and can lead to serious complications including liver Liver The liver is the largest gland in the human body. The liver is found in the superior right quadrant of the abdomen and weighs approximately 1.5 kilograms. Its main functions are detoxification, metabolism, nutrient storage (e.g., iron and vitamins), synthesis of coagulation factors, formation of bile, filtration, and storage of blood. Liver: Anatomy abscesses, intestinal fistulas, or fulminant colitis Fulminant colitis Pseudomembranous Colitis. Diagnosis is usually made based on stool studies or the detection of immunologic markers. Treatment consists of both an absorbable amebicidal agent such as metronidazole Metronidazole A nitroimidazole used to treat amebiasis; vaginitis; trichomonas infections; giardiasis; anaerobic bacteria; and treponemal infections. Pyogenic Liver Abscess and an intraluminal amebicidal agent such as paromomycin Paromomycin An aminoglycoside antibacterial and antiprotozoal agent produced by species of streptomyces. Amebicides.
Last updated: Dec 15, 2025
Causative organism: anaerobic protozoan parasites of the Entamoeba Entamoeba A genus of ameboid protozoa characterized by the presence of beaded chromatin on the inner surface of the nuclear membrane. Its organisms are parasitic in invertebrates and vertebrates, including humans. Nitroimidazoles genus
Transmission occurs through the ingestion of cysts Cysts Any fluid-filled closed cavity or sac that is lined by an epithelium. Cysts can be of normal, abnormal, non-neoplastic, or neoplastic tissues. Fibrocystic Change:
Risk factors and high-risk populations:
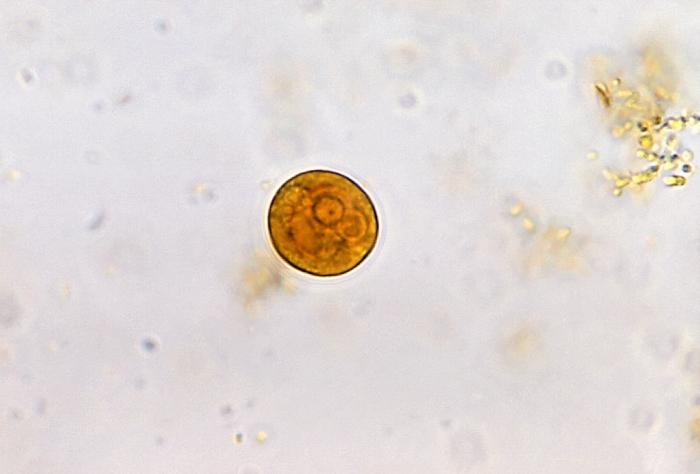
Entamoeba histolytica photomicrograph, cyst stage
Image: “Entamoeba histolytica cyst” by Yasser. License: CC BY 2.0
Trophozoites of Entamoeba histolytica with ingested erythrocytes stained with trichrome:
Ingested erythrocytes appear as dark inclusions. The parasites contain nuclei that have the typical small, centrally located karyosome and thin, uniform peripheral chromatin. Erythrophagocytosis, or ingestion of RBCs, is the distinguishing feature between E. histolytica and E. dispar.
The life cycle of Entamoeba Entamoeba A genus of ameboid protozoa characterized by the presence of beaded chromatin on the inner surface of the nuclear membrane. Its organisms are parasitic in invertebrates and vertebrates, including humans. Nitroimidazoles spp. is dependent on the infection of a host because transition through the life stages occurs within the host’s intestinal tract.
Cyst stage:
Trophozoite stage:
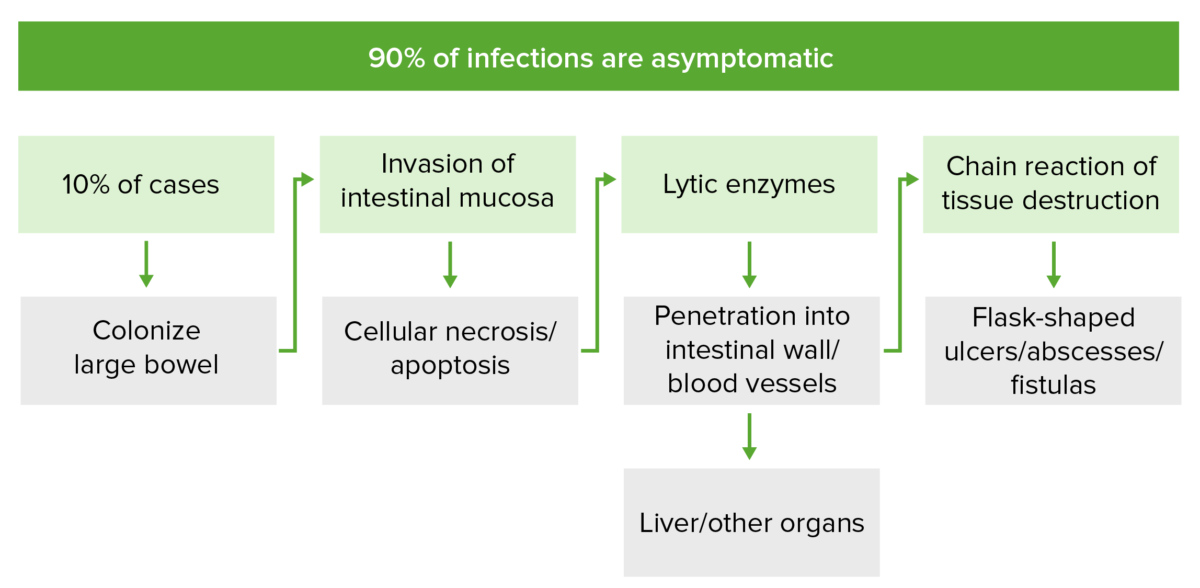
The pathogenesis of invasive Entamoeba histolytica infection:
In 10% of cases, E. histolytica colonizes the large intestine mucosa and invades via secretion of proteinases and lytic enzymes. This causes cellular necrosis and lysis of the membranes, respectively. This chain of events induces mucosal cell apoptosis and disrupts tight junctions between cells, allowing for flask-shaped ulcers, abscesses, and fistulas to form. Invasion may reach the portal venous system, through which E. histolytica can spread to other organs.
The incubation Incubation The amount time between exposure to an infectious agent and becoming symptomatic. Rabies Virus period is usually 2–4 weeks once ingested, but symptoms may develop up to 1 year after infection.
90% of all Entamoeba Entamoeba A genus of ameboid protozoa characterized by the presence of beaded chromatin on the inner surface of the nuclear membrane. Its organisms are parasitic in invertebrates and vertebrates, including humans. Nitroimidazoles infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease are asymptomatic:
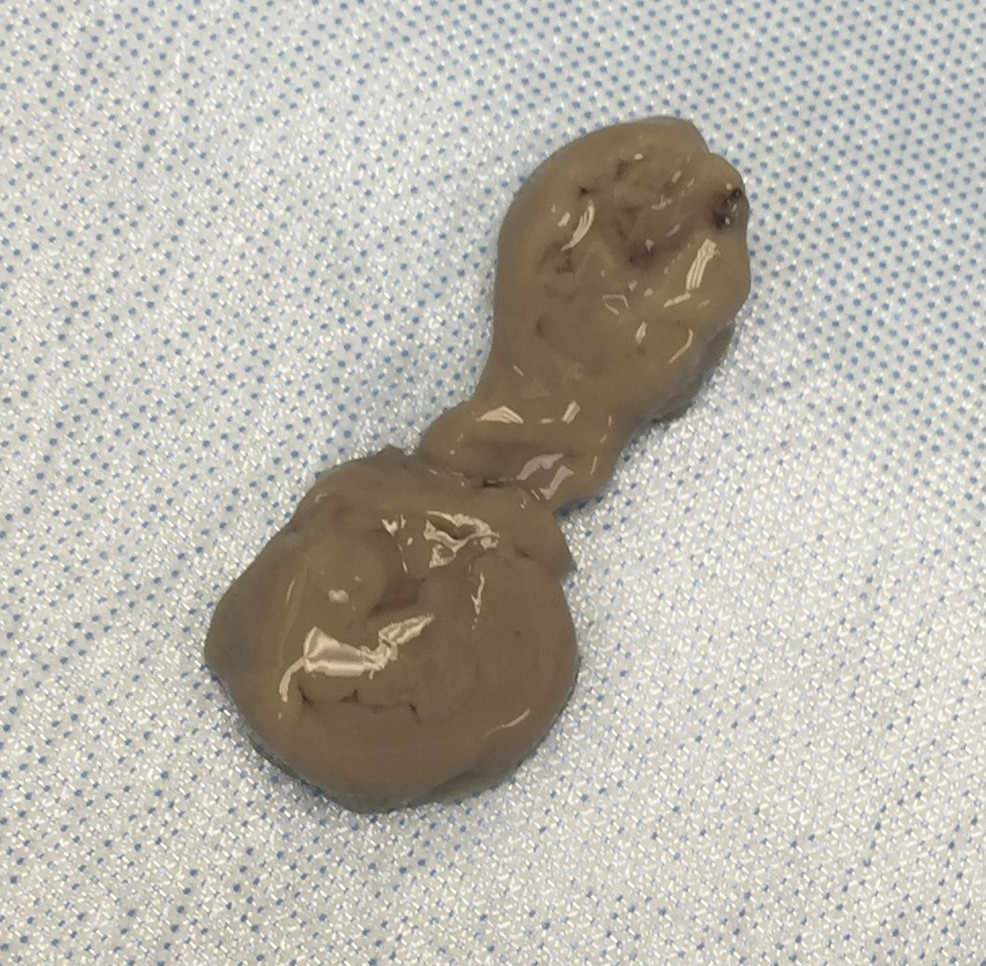
“Anchovy paste” aspirate from the liver abscess of a patient with invasive E. histolytica infection
Image: “Aspirate of “anchovy paste”” by Palak Patel et al. License: CC BY 4.0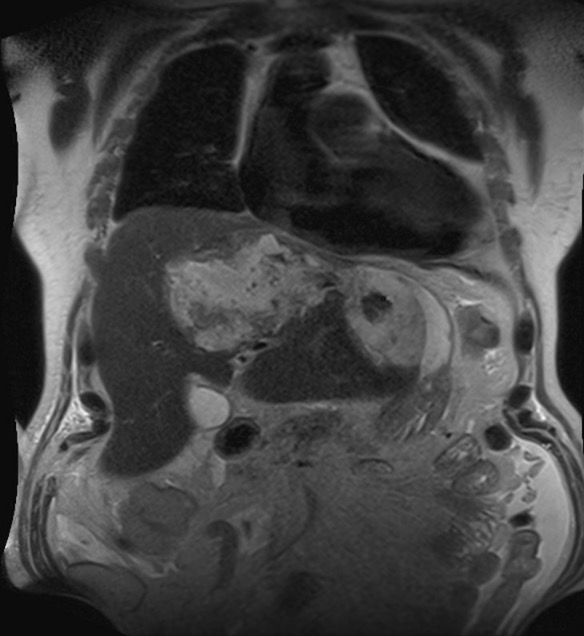
Amoebic abscess in the left lobe of the liver:
Coronal T2-weighted magnetic resonance cholangiopancreatography (MRC) showing a collection of heterogeneous material in the left lobe of the liver
The diagnosis of amebiasis is based on clinical suspicion as well as confirmatory testing. Several testing modalities may be used.
All E. histolytica infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease should be treated regardless of the absence or severity of symptoms; however, E. dispar infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease do not need to be treated.
All conditions that cause nonspecific or bloody diarrhea Bloody diarrhea Diarrhea are differential diagnoses for amebiasis.