Lymphatic filariasis, also known as elephantiasis, is a chronic mosquito-borne infection caused by Wuchereria bancrofti, Brugia malayi, and B. timori. The majority of causes are due to W. bancrofti. Mosquitos are the vectors, and humans are the primary reservoir Reservoir Animate or inanimate sources which normally harbor disease-causing organisms and thus serve as potential sources of disease outbreaks. Reservoirs are distinguished from vectors (disease vectors) and carriers, which are agents of disease transmission rather than continuing sources of potential disease outbreaks. Humans may serve both as disease reservoirs and carriers. Escherichia coli. Patients Patients Individuals participating in the health care system for the purpose of receiving therapeutic, diagnostic, or preventive procedures. Clinician–Patient Relationship with acute infection can present with fever Fever Fever is defined as a measured body temperature of at least 38°C (100.4°F). Fever is caused by circulating endogenous and/or exogenous pyrogens that increase levels of prostaglandin E2 in the hypothalamus. Fever is commonly associated with chills, rigors, sweating, and flushing of the skin. Fever, adenolymphangitis, dermatolymphangioadenitis, and tropical pulmonary eosinophilia Eosinophilia Abnormal increase of eosinophils in the blood, tissues or organs. Autosomal Dominant Hyperimmunoglobulin E Syndrome. Patients Patients Individuals participating in the health care system for the purpose of receiving therapeutic, diagnostic, or preventive procedures. Clinician–Patient Relationship with chronic infection present with lymphedema, which commonly affects the lower extremities (but can cause testicular swelling Swelling Inflammation or hydrocele Hydrocele Accumulation of serous fluid between the layers of membrane (tunica vaginalis) covering the testis in the scrotum. Varicocele, Hydrocele, and Spermatocele). Long-term effects also include renal manifestations. Thick and thin peripheral blood smears are the mainstay of diagnosis. Lymphatic filariasis without co-infection is generally managed with diethylcarbamazine Diethylcarbamazine An anthelmintic used primarily as the citrate in the treatment of filariasis, particularly infestations with wucheria bancrofti or loa loa. Anthelmintic Drugs. Prognosis Prognosis A prediction of the probable outcome of a disease based on a individual's condition and the usual course of the disease as seen in similar situations. Non-Hodgkin Lymphomas is good with early diagnosis and intervention. Elephantiasis, or late-stage lymphedema, is associated with significant disability Disability Determination of the degree of a physical, mental, or emotional handicap. The diagnosis is applied to legal qualification for benefits and income under disability insurance and to eligibility for social security and workman's compensation benefits. ABCDE Assessment and would require different methods (including surgery) to reduce swelling Swelling Inflammation and complications.
Last updated: Dec 15, 2025
Lymphatic filariasis is caused by nematodes.
Causative species:
General characteristics:
Stages of life:
Transmitted to mosquitoes:
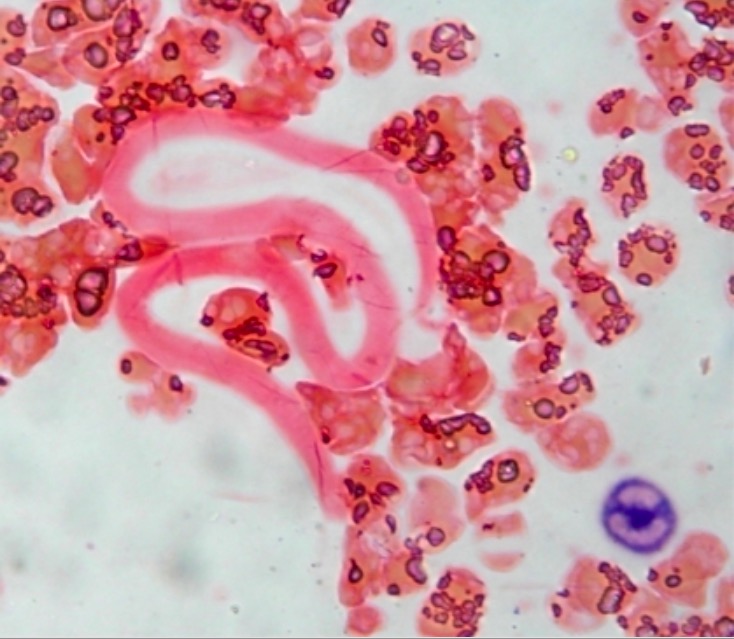
Wuchereria bancrofti microfilariae
Image: “Neutrophil Alkaline Phosphatase stained peripheral smear” by Department of Hematology, All India Institute of Medical Sciences, New Delhi. License: CC BY 2.0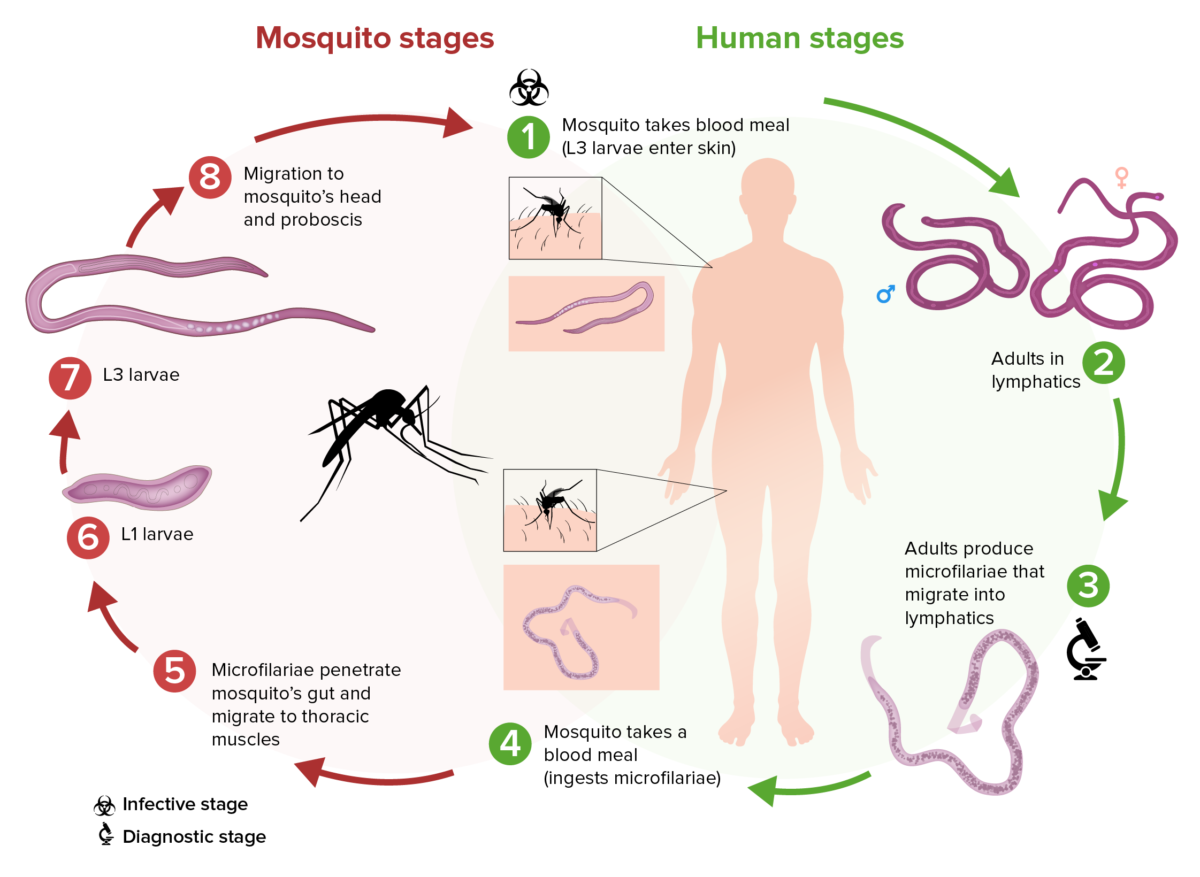
Schematic of the filarial life cycle of Wuchereria bancrofti:
(1) During a blood meal, an infected mosquito introduces L3 larvae into the skin of the human host.
(2) These larvae develop into adults that commonly reside in the lymphatics.
(3) Adults produce microfilariae that migrate actively through lymph and blood.
(4) A mosquito ingests the microfilariae during a blood meal.
(5) After ingestion, the microfilariae work their way through the wall of the mosquito’s midgut and reach the thoracic muscles.
(6) In the thoracic muscles, the microfilariae develop into L1 larvae.
(7) The L1 larvae subsequently develop into L3 infective larvae.
(8) The L3 larvae migrate to the mosquito’s proboscis.
(9) The L3 larvae can infect another human when the mosquito takes another blood meal.
Symptoms may take 9 months up to 1 year to manifest after the initial infection. Children or individuals in endemic areas often remain asymptomatic ( subclinical infection Subclinical infection Toxoplasma/Toxoplasmosis), while others show acute and/or chronic signs and symptoms.

Filariasis:
A patient with chronic lymphedema of the right lower extremity manifesting as elephantiasis
Filariasis without co-infection:
Filariasis with loiasis:
Filariasis with onchocerciasis:
Surgical treatment:
Long-term management to reduce lymphedema progression:
Occult filariasis is a filarial infection that extends into tissues, without any evidence in the blood. This leads to chronic complications, such as:
Prognosis Prognosis A prediction of the probable outcome of a disease based on a individual’s condition and the usual course of the disease as seen in similar situations. Non-Hodgkin Lymphomas: