Rotavirus belongs to the Reoviridae family and is a non-enveloped, double-stranded RNA RNA A polynucleotide consisting essentially of chains with a repeating backbone of phosphate and ribose units to which nitrogenous bases are attached. RNA is unique among biological macromolecules in that it can encode genetic information, serve as an abundant structural component of cells, and also possesses catalytic activity. RNA Types and Structure virus Virus Viruses are infectious, obligate intracellular parasites composed of a nucleic acid core surrounded by a protein capsid. Viruses can be either naked (non-enveloped) or enveloped. The classification of viruses is complex and based on many factors, including type and structure of the nucleoid and capsid, the presence of an envelope, the replication cycle, and the host range. Virology. Transmission occurs through the fecal-oral route Fecal-oral route Echovirus. Rotavirus is a common cause of severe gastroenteritis Gastroenteritis Gastroenteritis is inflammation of the stomach and intestines, commonly caused by infections from bacteria, viruses, or parasites. Transmission may be foodborne, fecal-oral, or through animal contact. Common clinical features include abdominal pain, diarrhea, vomiting, fever, and dehydration. Gastroenteritis in children. Severe infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease can result in dehydration Dehydration The condition that results from excessive loss of water from a living organism. Volume Depletion and Dehydration and death. Diagnostic testing is not usually required, but the virus Virus Viruses are infectious, obligate intracellular parasites composed of a nucleic acid core surrounded by a protein capsid. Viruses can be either naked (non-enveloped) or enveloped. The classification of viruses is complex and based on many factors, including type and structure of the nucleoid and capsid, the presence of an envelope, the replication cycle, and the host range. Virology can be detected in stool samples using ELISA ELISA An immunoassay utilizing an antibody labeled with an enzyme marker such as horseradish peroxidase. While either the enzyme or the antibody is bound to an immunosorbent substrate, they both retain their biologic activity; the change in enzyme activity as a result of the enzyme-antibody-antigen reaction is proportional to the concentration of the antigen and can be measured spectrophotometrically or with the naked eye. Many variations of the method have been developed. St. Louis Encephalitis Virus or PCR PCR Polymerase chain reaction (PCR) is a technique that amplifies DNA fragments exponentially for analysis. The process is highly specific, allowing for the targeting of specific genomic sequences, even with minuscule sample amounts. The PCR cycles multiple times through 3 phases: denaturation of the template DNA, annealing of a specific primer to the individual DNA strands, and synthesis/elongation of new DNA molecules. Polymerase Chain Reaction (PCR). Oral rehydration Rehydration Dengue Virus therapy is the mainstay of treatment. A live-attenuated vaccine Vaccine Suspensions of killed or attenuated microorganisms (bacteria, viruses, fungi, protozoa), antigenic proteins, synthetic constructs, or other bio-molecular derivatives, administered for the prevention, amelioration, or treatment of infectious and other diseases. Vaccination is available to prevent rotavirus infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease.
Last updated: Feb 18, 2025
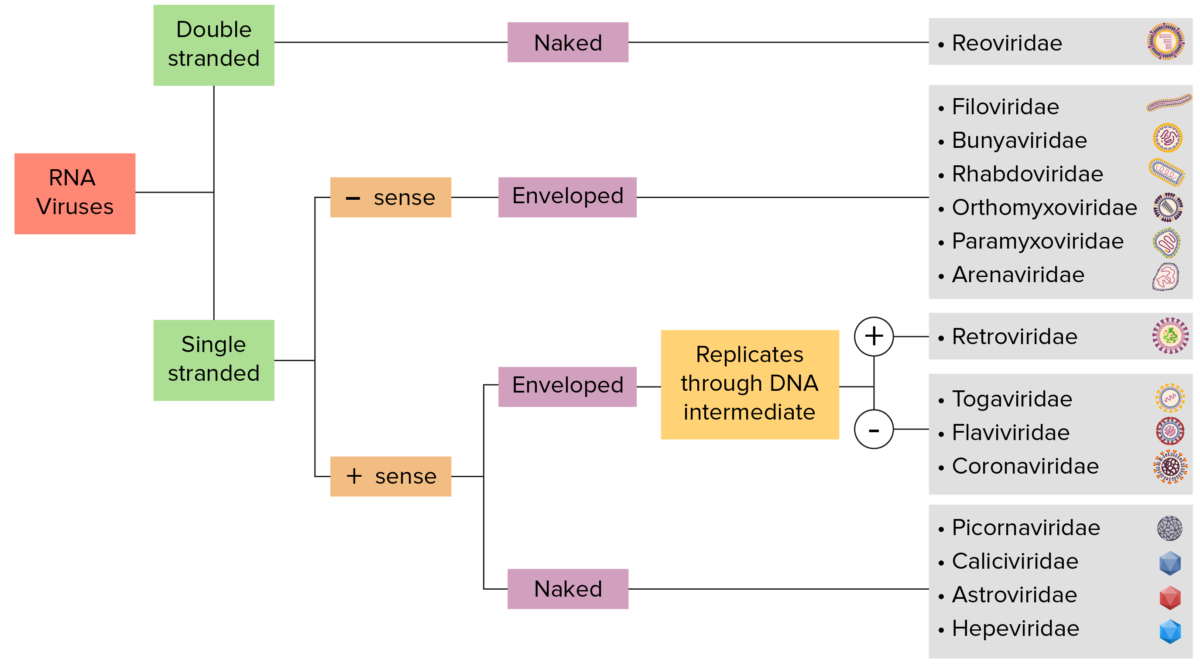
RNA virus identification:
Viruses can be classified in many ways. Most viruses, however, will have a genome formed by either DNA or RNA. RNA genome viruses can be further characterized by either a single- or double-stranded RNA. “Enveloped” viruses are covered by a thin coat of cell membrane (usually taken from the host cell). If the coat is absent, the viruses are called “naked” viruses. Viruses with single-stranded genomes are “positive-sense” viruses if the genome is directly employed as messenger RNA (mRNA), which is translated into proteins. “Negative-sense,” single-stranded viruses employ RNA dependent RNA polymerase, a viral enzyme, to transcribe their genome into messenger RNA.
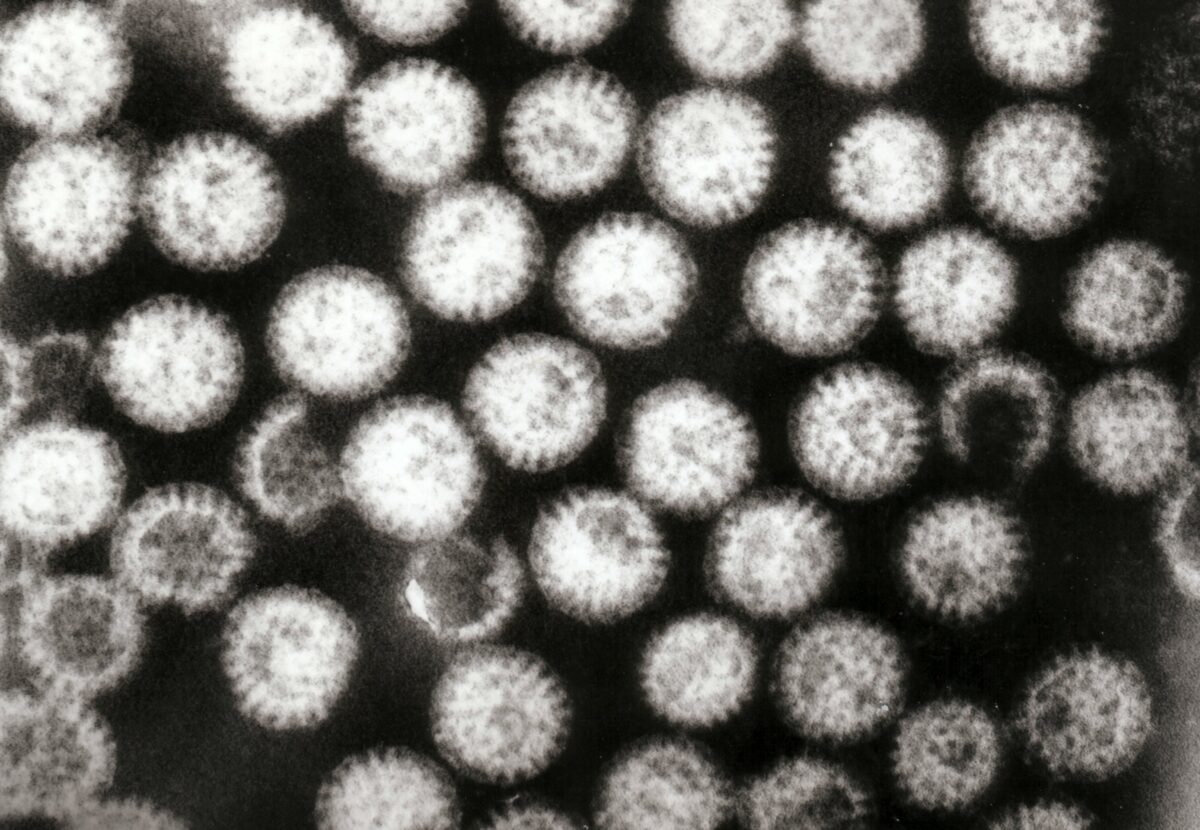
Transmission electron micrograph of multiple rotavirus virions
Image: “Transmission electron micrograph of multiple rotavirus particles” by Dr Graham Beards. License: CC BY 3.0Rotaviruses causes gastroenteritis Gastroenteritis Gastroenteritis is inflammation of the stomach and intestines, commonly caused by infections from bacteria, viruses, or parasites. Transmission may be foodborne, fecal-oral, or through animal contact. Common clinical features include abdominal pain, diarrhea, vomiting, fever, and dehydration. Gastroenteritis.
Humans are the primary reservoir Reservoir Animate or inanimate sources which normally harbor disease-causing organisms and thus serve as potential sources of disease outbreaks. Reservoirs are distinguished from vectors (disease vectors) and carriers, which are agents of disease transmission rather than continuing sources of potential disease outbreaks. Humans may serve both as disease reservoirs and carriers. Escherichia coli.
Transmission occurs through the fecal-oral route Fecal-oral route Echovirus:
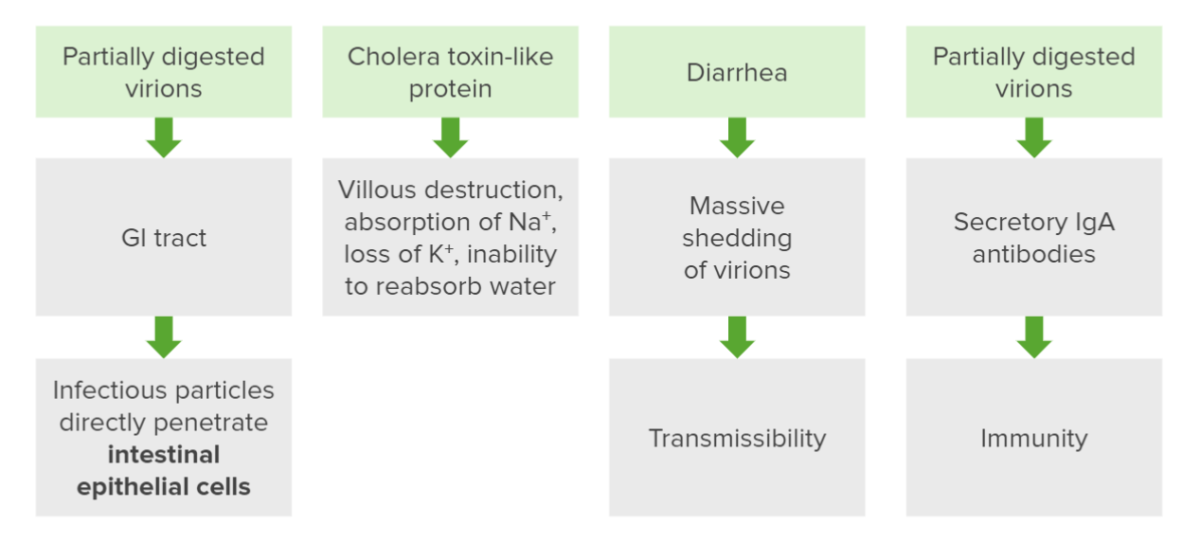
Flowchart summarizing the pathogenesis of rotavirus infection
Image by Lecturio. License: CC BY-NC-SA 4.0Timeline:
Symptoms:
Signs of dehydration Dehydration The condition that results from excessive loss of water from a living organism. Volume Depletion and Dehydration:
Rotavirus infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease are difficult to distinguish from other diarrheal diseases on the basis of clinical exam alone.
Live attenuated vaccines have been developed to prevent rotavirus infections Infections Invasion of the host organism by microorganisms or their toxins or by parasites that can cause pathological conditions or diseases. Chronic Granulomatous Disease: