Class 1 antiarrhythmics inhibit the fast Na channels Channels The Cell: Cell Membrane in non-nodal myocardial tissues and are subdivided into 3 categories (A, B, and C) on the basis of their speed of dissociation Dissociation Defense Mechanisms from the Na channels Channels The Cell: Cell Membrane and electrophysiologic effects. All drugs in class 1 reduce cardiac conduction velocity to some degree by slowing phase 0 depolarization Depolarization Membrane Potential. Indications vary between subgroups but generally include atrial and ventricular arrhythmias. Contraindications Contraindications A condition or factor associated with a recipient that makes the use of a drug, procedure, or physical agent improper or inadvisable. Contraindications may be absolute (life threatening) or relative (higher risk of complications in which benefits may outweigh risks). Noninvasive Ventilation, adverse effects, and warnings are category- and drug-dependent factors.
Last updated: Oct 13, 2022
Class 1 antiarrhythmics bind BIND Hyperbilirubinemia of the Newborn to and block the fast Na channels Channels The Cell: Cell Membrane in non-nodal tissue (e.g., myocytes Myocytes Mature contractile cells, commonly known as myocytes, that form one of three kinds of muscle. The three types of muscle cells are skeletal, cardiac, and smooth. They are derived from embryonic (precursor) muscle cells called myoblasts. Muscle Tissue: Histology of the atria and ventricles, His-Purkinje system):
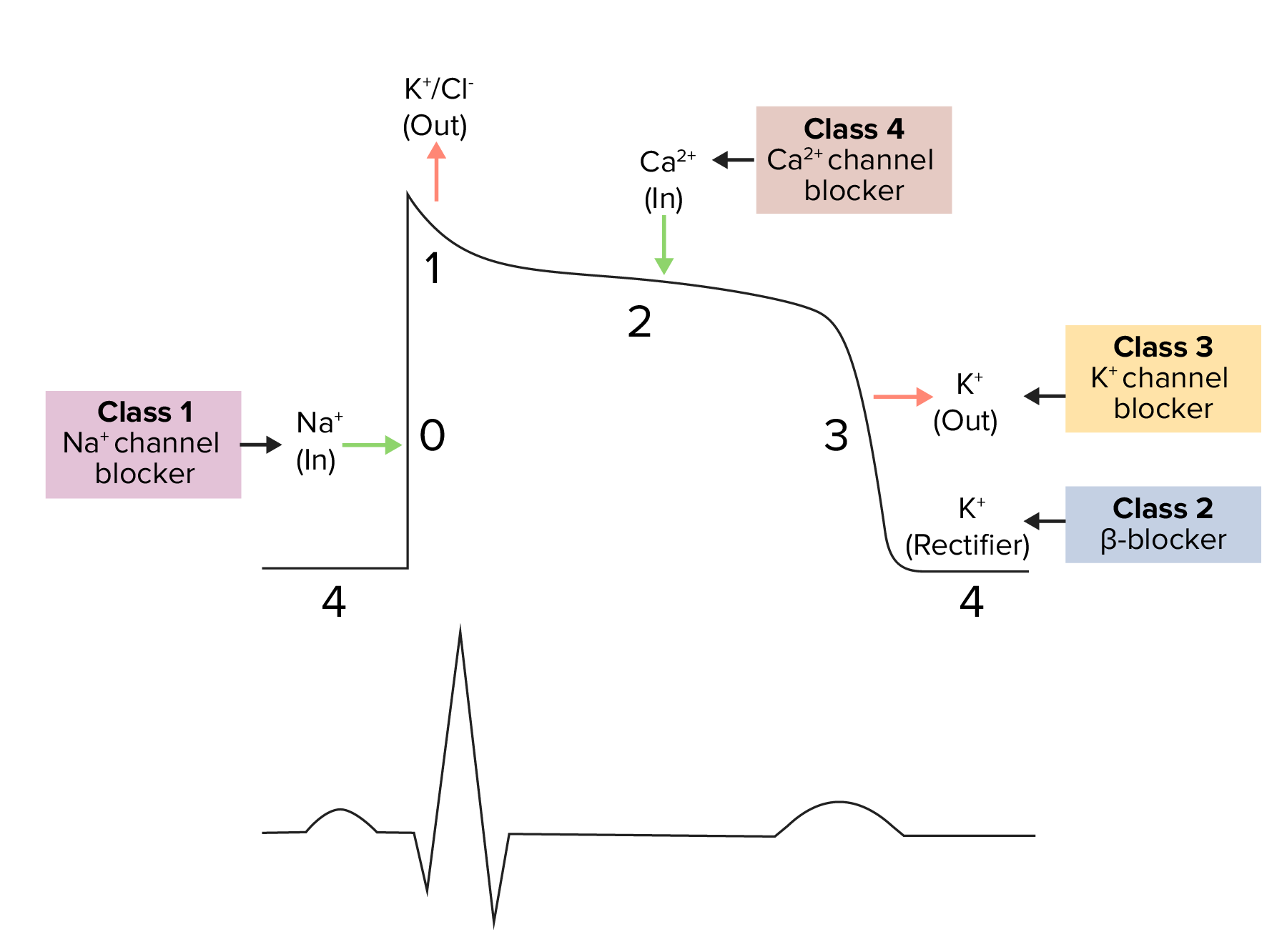
Diagram demonstrating a cardiac action potential and the phases of action for different antiarrhythmic drug classes:
The cycle starts with phase 4, the resting potential. Phase 0 is when rapid depolarization occurs due to an influx of Na ions into the cell and where class 1 (Na channel blockers) antiarrhythmics work.
Repolarization follows, with an efflux of K through fast K channels in phase 1, Ca influx in phase 2, and efflux of K through delayed K channels in phase 3.
| Class 1 subgroup | Strength of Na channel blockade | Effect on action potential Action Potential Abrupt changes in the membrane potential that sweep along the cell membrane of excitable cells in response to excitation stimuli. Membrane Potential duration | Effect on ERP | Effect seen on ECG ECG An electrocardiogram (ECG) is a graphic representation of the electrical activity of the heart plotted against time. Adhesive electrodes are affixed to the skin surface allowing measurement of cardiac impulses from many angles. The ECG provides 3-dimensional information about the conduction system of the heart, the myocardium, and other cardiac structures. Electrocardiogram (ECG) |
|---|---|---|---|---|
| 1A | Intermediate | ↑ | ↑ |
|
| 1B | Weak | ↓ | ↓ |
|
| 1C | Strong | Minimal to none | Minimal to none* |
|
To recall all class I antiarrhythmic medications, think of ordering a burger at a restaurant: “Double Quarter Pounder with Lettuce, Mayo, Pickles, and Fries, Please!”
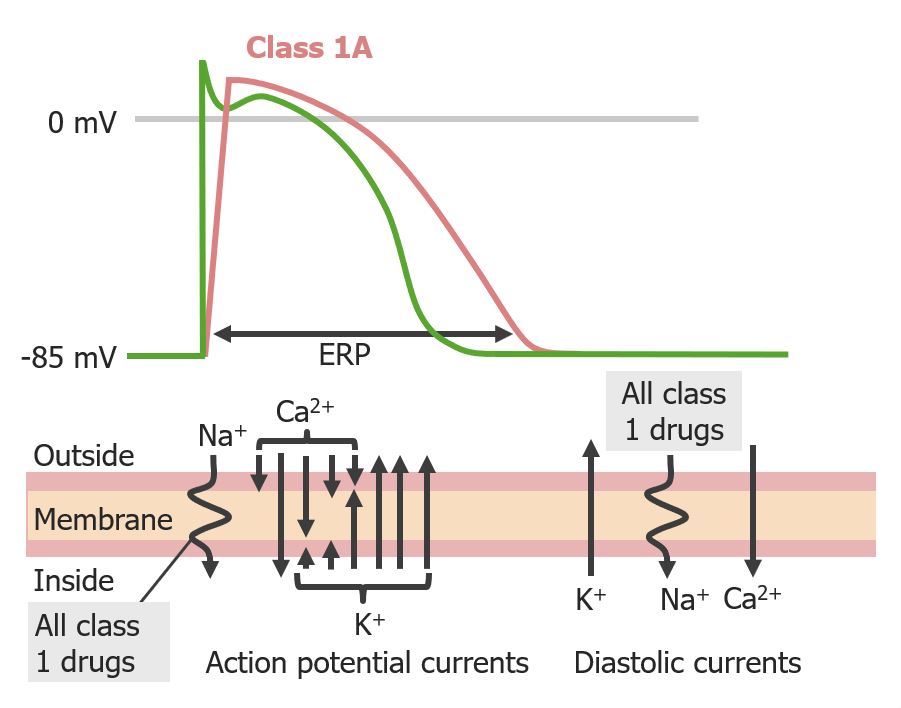
Effect of group 1A antiarrhythmics on the cardiac action potential:
Notice the delayed phase 0 upstroke, as well as increased effective refractory period and action potential duration
Due to severe side effects, such as their proarrhythmic effect, class 1A medications are usually reserved for life-threatening arrhythmias such as:
General:
Disopyramide:
Quinidine:
Procainamide:
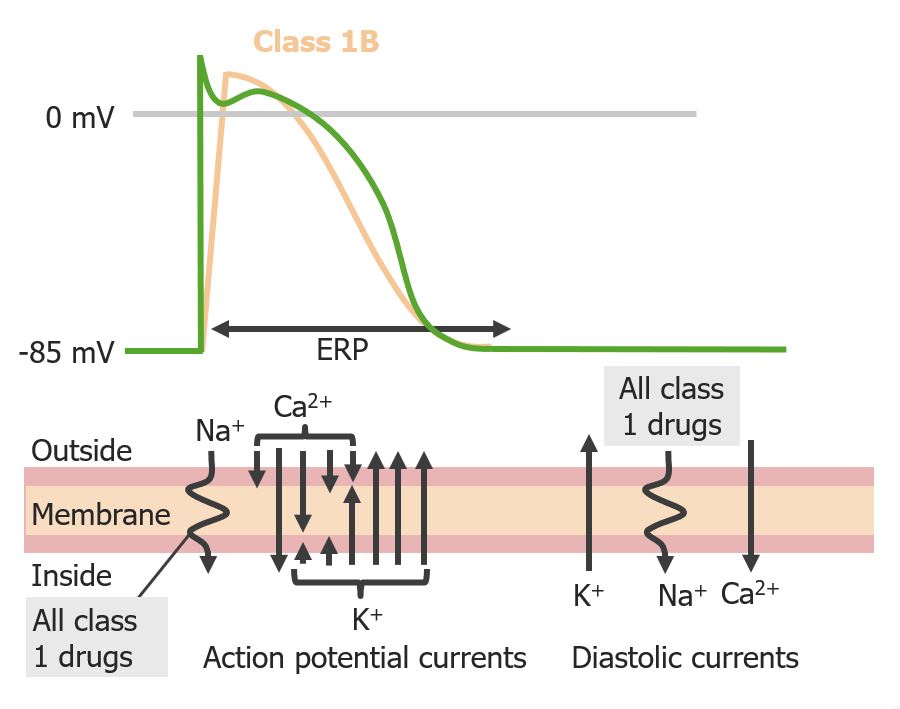
Effect of Group 1B antiarrhythmics on the cardiac action potential. Notice the decrease in action potential duration
Image by Lecturio. License: CC BY-NC-SA 4.0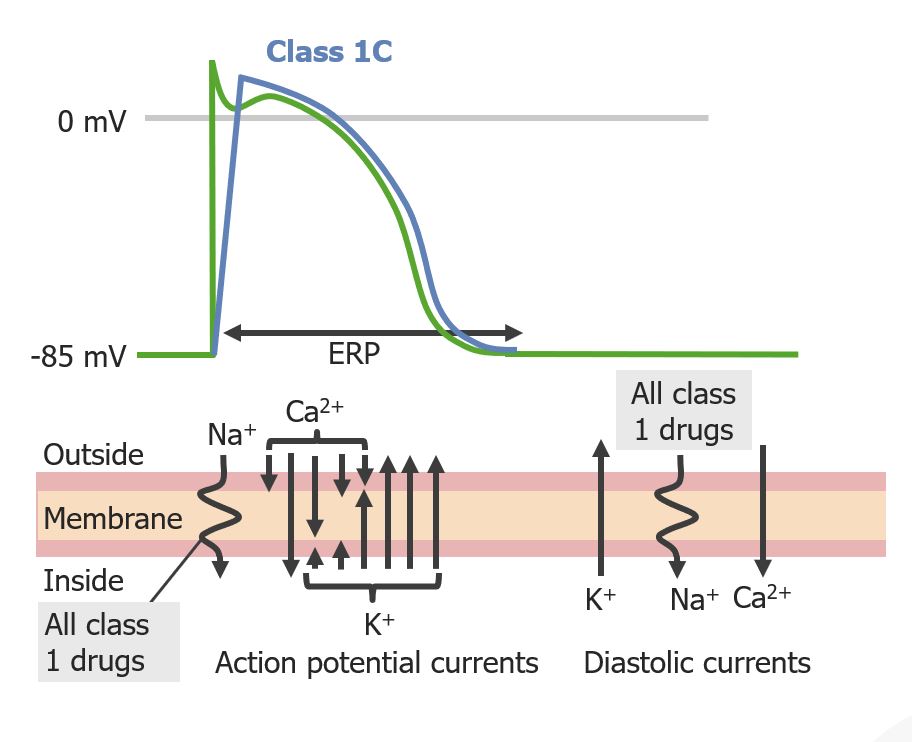
Effect of group 1C antiarrhythmics on the cardiac action potential:
Notice that this group has the greatest effect on phase 0, while the action potential duration remains about the same.
General:
Flecainide:
Propafenone:
| Class | Mechanism of action | Effects | Arrhythmia indications | |
|---|---|---|---|---|
| 1 | 1A |
|
|
|
| 1B | Ventricular | |||
| 1C | Mostly atrial | |||
| 2 |
|
|
Atrial and ventricular | |
| 3 |
|
|
Atrial and ventricular | |
| 4 |
|
|
Atrial | |