Gastritis is inflammation of the gastric mucosa and can be classified by time course (acute or chronic), histologic features, and etiology. The predominant causes are Helicobacter pylori infection and aspirin/NSAID use. Chronic (metaplastic) gastritis may be due to autoimmune or environmental causes and is a risk factor for gastric cancer. Individuals with gastritis may be asymptomatic or may have abdominal pain, dyspepsia, and nausea. Treatment of acute and chronic gastritis due to H. pylori infection involves acid suppression and antibiotics. Other causes are managed by avoiding offending agents and replacing associated deficiencies.
Last updated: Dec 15, 2025
Gastritis is the inflammation Inflammation Inflammation is a complex set of responses to infection and injury involving leukocytes as the principal cellular mediators in the body’s defense against pathogenic organisms. Inflammation is also seen as a response to tissue injury in the process of wound healing. The 5 cardinal signs of inflammation are pain, heat, redness, swelling, and loss of function. Inflammation of gastric mucosa Gastric mucosa Lining of the stomach, consisting of an inner epithelium, a middle lamina propria, and an outer muscularis mucosae. The surface cells produce mucus that protects the stomach from attack by digestive acid and enzymes. When the epithelium invaginates into the lamina propria at various region of the stomach (cardia; gastric fundus; and pylorus), different tubular gastric glands are formed. These glands consist of cells that secrete mucus, enzymes, hydrochloric acid, or hormones. Stomach: Anatomy associated with mucosal injury.
Gastritis is usually due to an infection or an immune-mediated process.
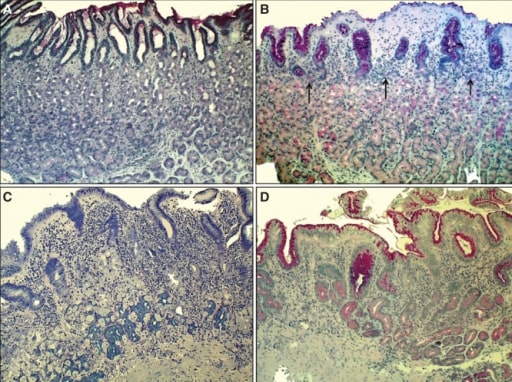
Chronic H. pylori gastritis:
A: Normal mucosa of the corpus
B: Non-atrophic gastritis: mild mononuclear inflammation seen in upper layer of the mucosa (superficial gastritis as indicated by arrows). Gland layer is still intact.
C: Moderate atrophic gastritis: intense chronic mononuclear inflammation occurs in the lower layers, accompanied by atrophy of the oxyntic glands (indicating stomach with hypochlorhydria). Acid secretion is impaired due to loss of parietal cells (in the oxyntic glands).
D: Severe atrophic gastritis, showing mild inflammation but there is loss of oxyntic glands.
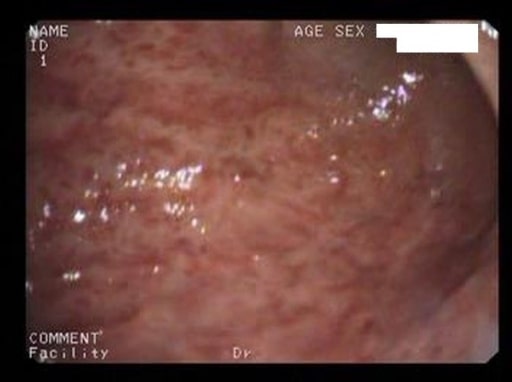
Acute erosive gastritis changes (endoscopic view) in a patient who has had prolonged COX-2 NSAID therapy
Image: “Acute erosive gastritis” by Department of Medical Sciences II, Medical Rehabilitation, University of Medicine and Pharmacy of Craiova, Romania. License: CC BY 2.0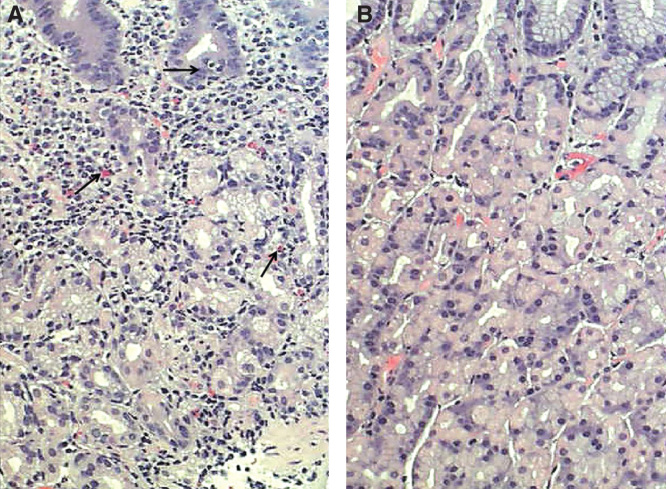
Chronic gastritis. A: mucosal inflammation with many mononuclear cells (arrows); B: normal mucosa.
Image: “Corpus mucosa” by Informa Healthcare. License: CC BY 4.0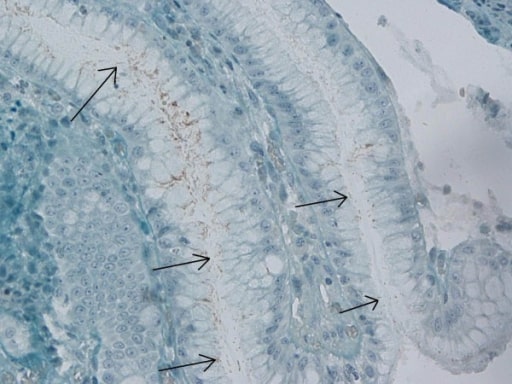
Immunohistochemical stain for H. pylori in the gastric antrum
Image: “Immunohistochemical H. pylori” by Department of Gastroenterology, Bakirkoy Dr Sadi Konuk Training and Research Hospital, Istanbul, Turkey. License: CC BY 2.0