The autonomic nervous system (ANS) is a component of the peripheral nervous system that uses both afferent (sensory) and efferent (effector) neurons, which control the functioning of the internal organs and involuntary processes via connections with the CNS. The ANS consists of the sympathetic and parasympathetic nervous systems, as well as the enteric nervous system (ENS). The efferent Efferent Neurons which send impulses peripherally to activate muscles or secretory cells. Nervous System: Histology nerve fibers Nerve Fibers Slender processes of neurons, including the axons and their glial envelopes (myelin sheath). Nerve fibers conduct nerve impulses to and from the central nervous system. Nervous System: Histology that terminate in the endocrine, vascular, and visceral structures coordinate the inner workings of the body in response to several afferent Afferent Neurons which conduct nerve impulses to the central nervous system. Nervous System: Histology inputs. The sympathetic and parasympathetic neural circuits coordinate stress responses and relaxation responses, respectively. The enteric nervous system Nervous system The nervous system is a small and complex system that consists of an intricate network of neural cells (or neurons) and even more glial cells (for support and insulation). It is divided according to its anatomical components as well as its functional characteristics. The brain and spinal cord are referred to as the central nervous system, and the branches of nerves from these structures are referred to as the peripheral nervous system. Nervous System: Anatomy, Structure, and Classification regulates visceral organ function. A balance between these systems results in homeostasis Homeostasis The processes whereby the internal environment of an organism tends to remain balanced and stable. Cell Injury and Death, whereas an imbalance leads to pathological conditions.
Last updated: Dec 15, 2025
The ANS is responsible for controlling functions that do not require conscious thought.
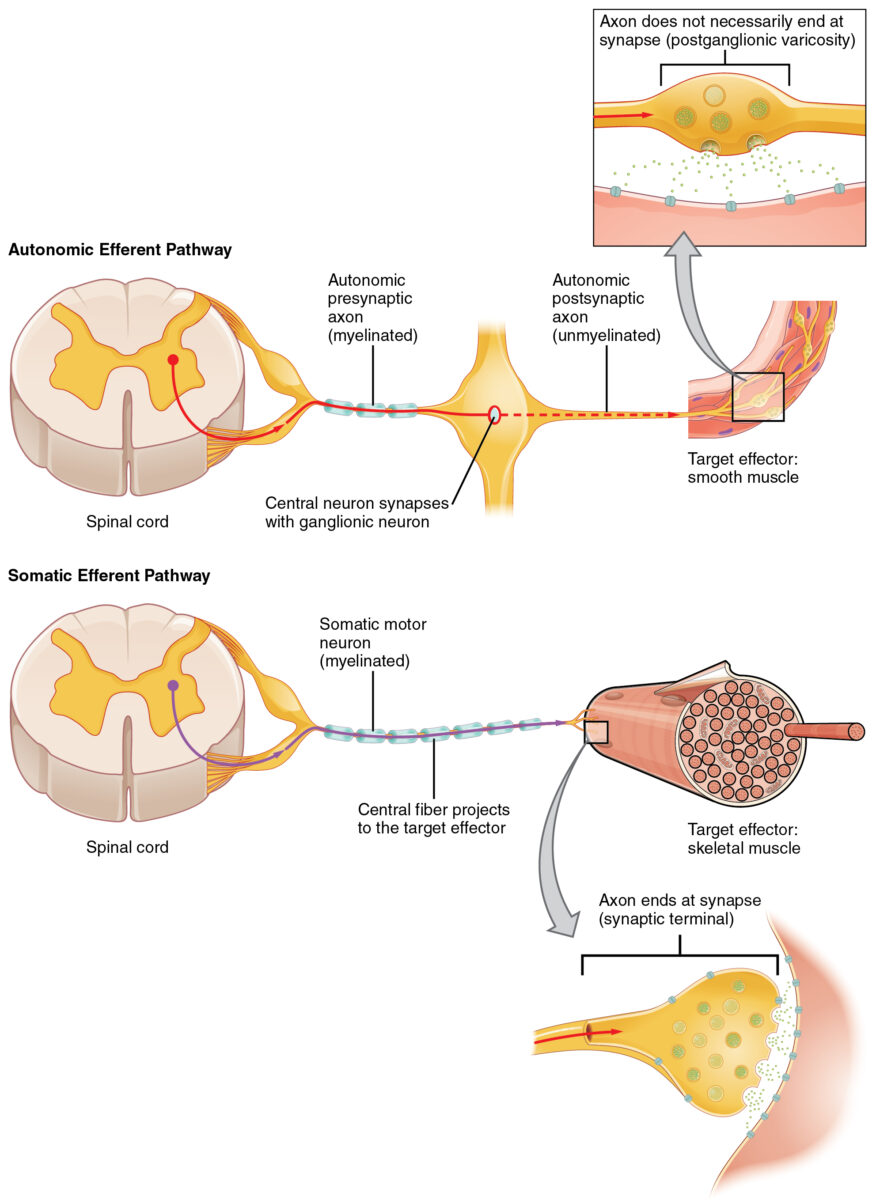
Autonomic efferent pathway vs. somatic efferent pathway
Image: “Comparison of Somatic and Visceral Reflexes” by Phil Schatz. License: CC BY 4.0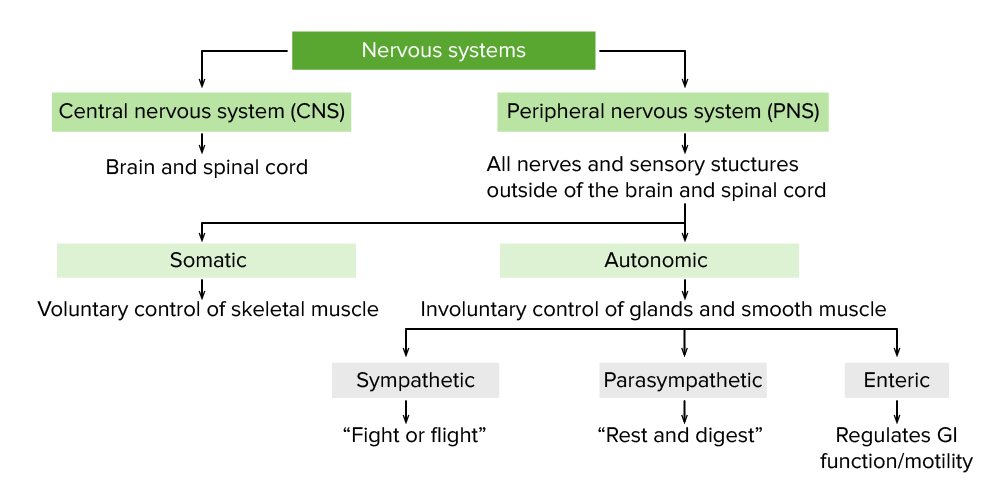
Schema showcasing the anatomical divisions of the nervous system
Image by Lecturio.The SNS is involved with many of the functions associated with the “fight-or-flight” response. Although this response is at the extreme end of the sympathetic physiology spectrum, it serves as a model to understand that the SNS enables our tissues to respond appropriately to varying degrees of physiologic stress.
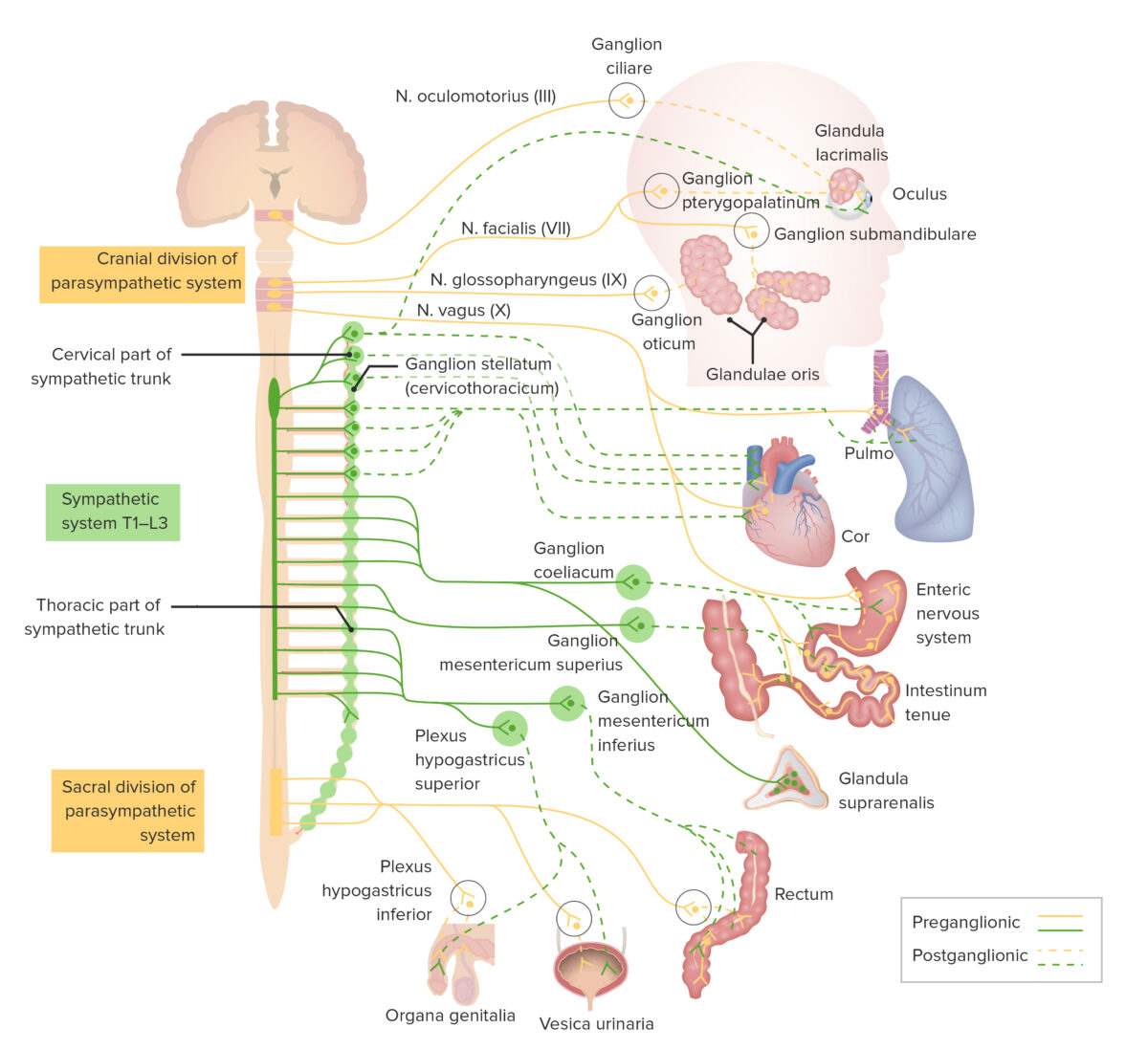
Pathways of the sympathetic nervous system
T: thoracic
L: lumbar
At its extreme, the SNS triggers the “fight-or-flight” response in reaction to physiologic stress.
In times of normal physiologic stress, the SNS is still constitutively active (but in balance with the PNS):
The SNS plays a complex role in immune regulation:
The dynamics of sympathetic outflow
Image by Lecturio.The PNS is involved with many of the functions associated with “rest and digest.” Although the “rest-and-digest” response is at the extreme end of the parasympathetic physiology spectrum, it serves as a model to understand that the PNS enables our tissues to recover appropriately from and/or balance varying degrees of physiologic stress.
At its extreme, the PNS triggers the “rest-and-digest” response in reaction to a need for physiologic recovery and balances the actions of the SNS.
The PNS plays a crucial role in immune regulation:

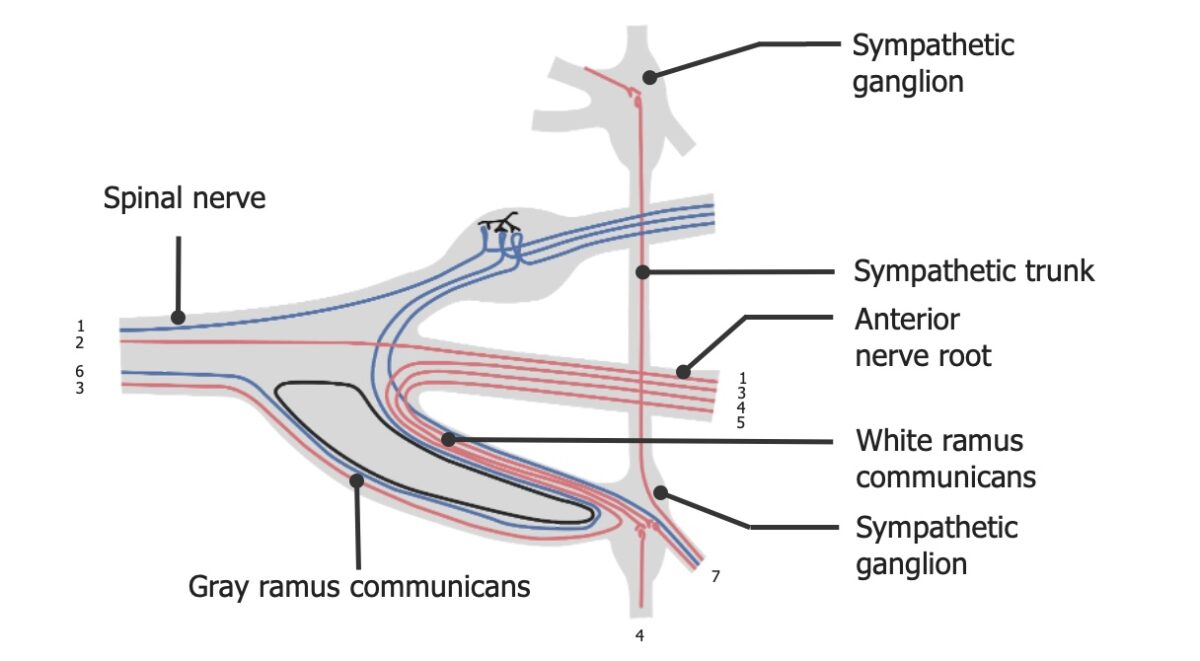
Pathways of the sympathetic and parasympathetic nervous systems
T: thoracic
L: lumbar
| Target | Sympathetic effects and receptors Receptors Receptors are proteins located either on the surface of or within a cell that can bind to signaling molecules known as ligands (e.g., hormones) and cause some type of response within the cell. Receptors | Parasympathetic effects and receptors Receptors Receptors are proteins located either on the surface of or within a cell that can bind to signaling molecules known as ligands (e.g., hormones) and cause some type of response within the cell. Receptors |
|---|---|---|
| Brain Brain The part of central nervous system that is contained within the skull (cranium). Arising from the neural tube, the embryonic brain is comprised of three major parts including prosencephalon (the forebrain); mesencephalon (the midbrain); and rhombencephalon (the hindbrain). The developed brain consists of cerebrum; cerebellum; and other structures in the brain stem. Nervous System: Anatomy, Structure, and Classification | α1: cerebral blood flow Blood flow Blood flow refers to the movement of a certain volume of blood through the vasculature over a given unit of time (e.g., mL per minute). Vascular Resistance, Flow, and Mean Arterial Pressure regulation | M1: ↑ memory Memory Complex mental function having four distinct phases: (1) memorizing or learning, (2) retention, (3) recall, and (4) recognition. Clinically, it is usually subdivided into immediate, recent, and remote memory. Psychiatric Assessment and attention Attention Focusing on certain aspects of current experience to the exclusion of others. It is the act of heeding or taking notice or concentrating. Psychiatric Assessment |
| Eye | α1: contraction (
mydriasis
Mydriasis
Dilation of pupils to greater than 6 mm combined with failure of the pupils to constrict when stimulated with light. This condition may occur due to injury of the pupillary fibers in the oculomotor nerve, in acute angle-closure glaucoma, and in adie syndrome.
Glaucoma) of the iris dilator muscle β2: focusing on distant objects |
M3: miosis Miosis Pupil: Physiology and Abnormalities and accommodation Accommodation Refractive Errors |
| Bladder Bladder A musculomembranous sac along the urinary tract. Urine flows from the kidneys into the bladder via the ureters, and is held there until urination. Pyelonephritis and Perinephric Abscess | α1: constriction of the
bladder
Bladder
A musculomembranous sac along the urinary tract. Urine flows from the kidneys into the bladder via the ureters, and is held there until urination.
Pyelonephritis and Perinephric Abscess sphincter control of micturition and urine flow Flow Blood flows through the heart, arteries, capillaries, and veins in a closed, continuous circuit. Flow is the movement of volume per unit of time. Flow is affected by the pressure gradient and the resistance fluid encounters between 2 points. Vascular resistance is the opposition to flow, which is caused primarily by blood friction against vessel walls. Vascular Resistance, Flow, and Mean Arterial Pressure β2: bladder Bladder A musculomembranous sac along the urinary tract. Urine flows from the kidneys into the bladder via the ureters, and is held there until urination. Pyelonephritis and Perinephric Abscess relaxation |
M3: relaxation of vesical sphincter muscle; contraction of the detrusor muscle Detrusor muscle Urinary Incontinence |
| Prostate Prostate The prostate is a gland in the male reproductive system. The gland surrounds the bladder neck and a portion of the urethra. The prostate is an exocrine gland that produces a weakly acidic secretion, which accounts for roughly 20% of the seminal fluid. and reproductive organs | α1: cause ejaculation by prostate Prostate The prostate is a gland in the male reproductive system. The gland surrounds the bladder neck and a portion of the urethra. The prostate is an exocrine gland that produces a weakly acidic secretion, which accounts for roughly 20% of the seminal fluid. contraction | M1: erection Erection The state of the penis when the erectile tissue becomes filled or swollen (tumid) with blood and causes the penis to become rigid and elevated. It is a complex process involving central nervous system; peripheral nervous systems; hormones; smooth muscles; and vascular functions. Penis: Anatomy |
| Kidney | α1: ↓
renin
Renin
A highly specific (leu-leu) endopeptidase that generates angiotensin I from its precursor angiotensinogen, leading to a cascade of reactions which elevate blood pressure and increase sodium retention by the kidney in the renin-angiotensin system.
Renal Sodium and Water Regulation
secretion
Secretion
Coagulation Studies β2: ↑ renin Renin A highly specific (leu-leu) endopeptidase that generates angiotensin I from its precursor angiotensinogen, leading to a cascade of reactions which elevate blood pressure and increase sodium retention by the kidney in the renin-angiotensin system. Renal Sodium and Water Regulation secretion Secretion Coagulation Studies |
None |
| Veins Veins Veins are tubular collections of cells, which transport deoxygenated blood and waste from the capillary beds back to the heart. Veins are classified into 3 types: small veins/venules, medium veins, and large veins. Each type contains 3 primary layers: tunica intima, tunica media, and tunica adventitia. Veins: Histology and arterioles Arterioles The smallest divisions of the arteries located between the muscular arteries and the capillaries. Arteries: Histology | α1: contraction of
smooth muscles
Smooth muscles
Unstriated and unstriped muscle, one of the muscles of the internal organs, blood vessels, hair follicles, etc. Contractile elements are elongated, usually spindle-shaped cells with centrally located nuclei. Smooth muscle fibers are bound together into sheets or bundles by reticular fibers and frequently elastic nets are also abundant.
Muscle Tissue: Histology of the peripheral blood vessels β2: promote dilation of arterioles Arterioles The smallest divisions of the arteries located between the muscular arteries and the capillaries. Arteries: Histology and veins Veins Veins are tubular collections of cells, which transport deoxygenated blood and waste from the capillary beds back to the heart. Veins are classified into 3 types: small veins/venules, medium veins, and large veins. Each type contains 3 primary layers: tunica intima, tunica media, and tunica adventitia. Veins: Histology; consequently, a decrease in total peripheral resistance Resistance Physiologically, the opposition to flow of air caused by the forces of friction. As a part of pulmonary function testing, it is the ratio of driving pressure to the rate of air flow. Ventilation: Mechanics of Breathing, blood pressure, and afterload Afterload Afterload is the resistance in the aorta that prevents blood from leaving the heart. Afterload represents the pressure the LV needs to overcome to eject blood into the aorta. Cardiac Mechanics |
Most vessels do not possess parasympathetic innervation. |
| Platelets Platelets Platelets are small cell fragments involved in hemostasis. Thrombopoiesis takes place primarily in the bone marrow through a series of cell differentiation and is influenced by several cytokines. Platelets are formed after fragmentation of the megakaryocyte cytoplasm. Platelets: Histology | α2: ↑ platelet aggregation Platelet aggregation The attachment of platelets to one another. This clumping together can be induced by a number of agents (e.g., thrombin; collagen) and is part of the mechanism leading to the formation of a thrombus. Hemostasis | None |
| Heart | β1: ↑ HR (positive chronotropic); ↑ conduction velocity (positive dromotropic); ↑ contractibility Contractibility Skeletal Muscle Contraction (positive inotropic) |
M2: ↓ HR (negative chronotropic); ↓ conduction velocity (negative dromotropic); ↓ contractibility Contractibility Skeletal Muscle Contraction (negative inotropic) |
| Bronchioles Bronchioles The small airways branching off the tertiary bronchi. Terminal bronchioles lead into several orders of respiratory bronchioles which in turn lead into alveolar ducts and then into pulmonary alveoli. Bronchial Tree: Anatomy | β2: bronchiolar smooth muscle relaxation | M3: bronchoconstriction |
| Liver Liver The liver is the largest gland in the human body. The liver is found in the superior right quadrant of the abdomen and weighs approximately 1.5 kilograms. Its main functions are detoxification, metabolism, nutrient storage (e.g., iron and vitamins), synthesis of coagulation factors, formation of bile, filtration, and storage of blood. Liver: Anatomy | β2: ↑ glycogenolysis Glycogenolysis The release of glucose from glycogen by glycogen phosphorylase (phosphorolysis). The released glucose-1-phosphate is then converted to glucose-6-phosphate by phosphoglucomutase before entering glycolysis. Glycogenolysis is stimulated by glucagon or epinephrine via the activation of phosphorylase kinase. Glycogen Metabolism | M3: ↑ gluconeogenesis Gluconeogenesis Gluconeogenesis is the process of making glucose from noncarbohydrate precursors. This metabolic pathway is more than just a reversal of glycolysis. Gluconeogenesis provides the body with glucose not obtained from food, such as during a fasting period. The production of glucose is critical for organs and cells that cannot use fat for fuel. Gluconeogenesis |
| Adipose tissue Adipose tissue Adipose tissue is a specialized type of connective tissue that has both structural and highly complex metabolic functions, including energy storage, glucose homeostasis, and a multitude of endocrine capabilities. There are three types of adipose tissue, white adipose tissue, brown adipose tissue, and beige or “brite” adipose tissue, which is a transitional form. Adipose Tissue: Histology | α2: ↓
lipolysis
Lipolysis
The metabolic process of breaking down lipids to release free fatty acids, the major oxidative fuel for the body. Lipolysis may involve dietary lipids in the digestive tract, circulating lipids in the blood, and stored lipids in the adipose tissue or the liver. A number of enzymes are involved in such lipid hydrolysis, such as lipase and lipoprotein lipase from various tissues.
Metabolic Dysfunction-associated Steatotic Liver Disease (MASLD) β1, β2: ↑ lipolysis Lipolysis The metabolic process of breaking down lipids to release free fatty acids, the major oxidative fuel for the body. Lipolysis may involve dietary lipids in the digestive tract, circulating lipids in the blood, and stored lipids in the adipose tissue or the liver. A number of enzymes are involved in such lipid hydrolysis, such as lipase and lipoprotein lipase from various tissues. Metabolic Dysfunction-associated Steatotic Liver Disease (MASLD) |
None |
| Skeletal muscle | β3: thermogenesis | M3: contraction of the detrusor muscle Detrusor muscle Urinary Incontinence |
The enteric portion of the ANS is associated with functions of digestion Digestion Digestion refers to the process of the mechanical and chemical breakdown of food into smaller particles, which can then be absorbed and utilized by the body. Digestion and Absorption and regulation of the GI secretions and the smooth muscle.