The pancreas Pancreas The pancreas lies mostly posterior to the stomach and extends across the posterior abdominal wall from the duodenum on the right to the spleen on the left. This organ has both exocrine and endocrine tissue. Pancreas: Anatomy is a composite organ containing a distinctive combination of cell lineages. The exocrine tissue comprises acinar cells Acinar cells Cells lining the saclike dilatations known as acini of various glands or the lungs. Gastrointestinal Secretions, which secrete digestive enzymes Enzymes Enzymes are complex protein biocatalysts that accelerate chemical reactions without being consumed by them. Due to the body's constant metabolic needs, the absence of enzymes would make life unsustainable, as reactions would occur too slowly without these molecules. Basics of Enzymes into the intestine. The endocrine function is performed by the islets of Langerhans, which consist of distinct cell types secreting 4 different hormones Hormones Hormones are messenger molecules that are synthesized in one part of the body and move through the bloodstream to exert specific regulatory effects on another part of the body. Hormones play critical roles in coordinating cellular activities throughout the body in response to the constant changes in both the internal and external environments. Hormones: Overview and Types into the circulation Circulation The movement of the blood as it is pumped through the cardiovascular system. ABCDE Assessment (α-cells, glucagon Glucagon A 29-amino acid pancreatic peptide derived from proglucagon which is also the precursor of intestinal glucagon-like peptides. Glucagon is secreted by pancreatic alpha cells and plays an important role in regulation of blood glucose concentration, ketone metabolism, and several other biochemical and physiological processes. Gastrointestinal Secretions; β-cells, insulin Insulin Insulin is a peptide hormone that is produced by the beta cells of the pancreas. Insulin plays a role in metabolic functions such as glucose uptake, glycolysis, glycogenesis, lipogenesis, and protein synthesis. Exogenous insulin may be needed for individuals with diabetes mellitus, in whom there is a deficiency in endogenous insulin or increased insulin resistance. Insulin; δ-cells, somatostatin Somatostatin A 14-amino acid peptide named for its ability to inhibit pituitary growth hormone release, also called somatotropin release-inhibiting factor. It is expressed in the central and peripheral nervous systems, the gut, and other organs. SRIF can also inhibit the release of thyroid-stimulating hormone; prolactin; insulin; and glucagon besides acting as a neurotransmitter and neuromodulator. In a number of species including humans, there is an additional form of somatostatin, srif-28 with a 14-amino acid extension at the n-terminal. Gastrointestinal Secretions; and γ-cells, pancreatic polypeptide). Endocrine hormones Endocrine hormones Released from specialized cells and travel through the blood to a distant site of action Hormones: Overview and Types, as well as some exocrine enzymes Enzymes Enzymes are complex protein biocatalysts that accelerate chemical reactions without being consumed by them. Due to the body's constant metabolic needs, the absence of enzymes would make life unsustainable, as reactions would occur too slowly without these molecules. Basics of Enzymes, can be measured in body fluids and provide important diagnostic information in acute and chronic pancreatic disease.
Last updated: May 17, 2024
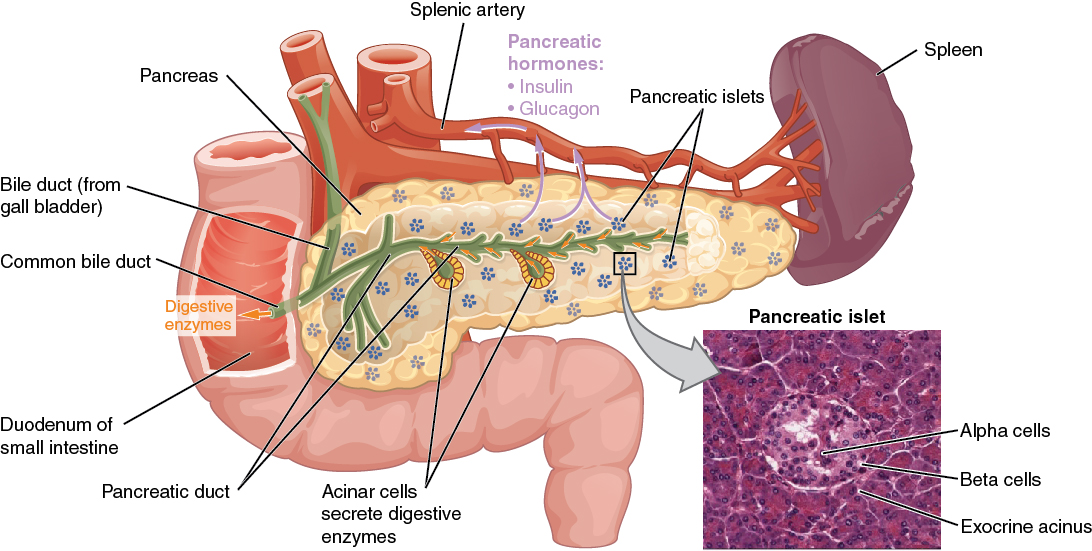
The pancreas Pancreas The pancreas lies mostly posterior to the stomach and extends across the posterior abdominal wall from the duodenum on the right to the spleen on the left. This organ has both exocrine and endocrine tissue. Pancreas: Anatomy is an organ positioned across the posterior aspect of the abdomen, behind the stomach Stomach The stomach is a muscular sac in the upper left portion of the abdomen that plays a critical role in digestion. The stomach develops from the foregut and connects the esophagus with the duodenum. Structurally, the stomach is C-shaped and forms a greater and lesser curvature and is divided grossly into regions: the cardia, fundus, body, and pylorus. Stomach: Anatomy and has 2 major functions:
The pancreas
Image: “The pancreas” by CDC. License: CC BY 3.0The islets of Langerhans, scattered throughout the pancreas Pancreas The pancreas lies mostly posterior to the stomach and extends across the posterior abdominal wall from the duodenum on the right to the spleen on the left. This organ has both exocrine and endocrine tissue. Pancreas: Anatomy, have different types of cells, which correspond to the following hormones Hormones Hormones are messenger molecules that are synthesized in one part of the body and move through the bloodstream to exert specific regulatory effects on another part of the body. Hormones play critical roles in coordinating cellular activities throughout the body in response to the constant changes in both the internal and external environments. Hormones: Overview and Types:
Functions and stimuli differ with each hormone.
| Parameter | Associated tests | Conditions |
|---|---|---|
| Insulin Insulin Insulin is a peptide hormone that is produced by the beta cells of the pancreas. Insulin plays a role in metabolic functions such as glucose uptake, glycolysis, glycogenesis, lipogenesis, and protein synthesis. Exogenous insulin may be needed for individuals with diabetes mellitus, in whom there is a deficiency in endogenous insulin or increased insulin resistance. Insulin |
|
Increased in:
|
| Somatostatin Somatostatin A 14-amino acid peptide named for its ability to inhibit pituitary growth hormone release, also called somatotropin release-inhibiting factor. It is expressed in the central and peripheral nervous systems, the gut, and other organs. SRIF can also inhibit the release of thyroid-stimulating hormone; prolactin; insulin; and glucagon besides acting as a neurotransmitter and neuromodulator. In a number of species including humans, there is an additional form of somatostatin, srif-28 with a 14-amino acid extension at the n-terminal. Gastrointestinal Secretions | Plasma Plasma The residual portion of blood that is left after removal of blood cells by centrifugation without prior blood coagulation. Transfusion Products somatostatin Somatostatin A 14-amino acid peptide named for its ability to inhibit pituitary growth hormone release, also called somatotropin release-inhibiting factor. It is expressed in the central and peripheral nervous systems, the gut, and other organs. SRIF can also inhibit the release of thyroid-stimulating hormone; prolactin; insulin; and glucagon besides acting as a neurotransmitter and neuromodulator. In a number of species including humans, there is an additional form of somatostatin, srif-28 with a 14-amino acid extension at the n-terminal. Gastrointestinal Secretions | Increased in:
|
| Glucagon Glucagon A 29-amino acid pancreatic peptide derived from proglucagon which is also the precursor of intestinal glucagon-like peptides. Glucagon is secreted by pancreatic alpha cells and plays an important role in regulation of blood glucose concentration, ketone metabolism, and several other biochemical and physiological processes. Gastrointestinal Secretions | Plasma Plasma The residual portion of blood that is left after removal of blood cells by centrifugation without prior blood coagulation. Transfusion Products glucagon Glucagon A 29-amino acid pancreatic peptide derived from proglucagon which is also the precursor of intestinal glucagon-like peptides. Glucagon is secreted by pancreatic alpha cells and plays an important role in regulation of blood glucose concentration, ketone metabolism, and several other biochemical and physiological processes. Gastrointestinal Secretions | Increased in:
|
| Pancreatic polypeptide | Plasma Plasma The residual portion of blood that is left after removal of blood cells by centrifugation without prior blood coagulation. Transfusion Products pancreatic polypeptide | Increased in:
|
| Pancreatic amylase Amylase A group of amylolytic enzymes that cleave starch, glycogen, and related alpha-1, 4-glucans. Digestion and Absorption | Serum/ plasma Plasma The residual portion of blood that is left after removal of blood cells by centrifugation without prior blood coagulation. Transfusion Products amylase Amylase A group of amylolytic enzymes that cleave starch, glycogen, and related alpha-1, 4-glucans. Digestion and Absorption | Increased in:
|
| Pancreatic lipase Lipase An enzyme of the hydrolase class that catalyzes the reaction of triacylglycerol and water to yield diacylglycerol and a fatty acid anion. It is produced by glands on the tongue and by the pancreas and initiates the digestion of dietary fats. Malabsorption and Maldigestion | Serum lipase Lipase An enzyme of the hydrolase class that catalyzes the reaction of triacylglycerol and water to yield diacylglycerol and a fatty acid anion. It is produced by glands on the tongue and by the pancreas and initiates the digestion of dietary fats. Malabsorption and Maldigestion | Increased in:
|
| Pancreatic elastase Elastase A protease of broad specificity, obtained from dried pancreas. Molecular weight is approximately 25, 000. The enzyme breaks down elastin, the specific protein of elastic fibers, and digests other proteins such as fibrin, hemoglobin, and albumin. Proteins and Peptides | Elastase-1 (feces) | Increased in: acute
pancreatitis
Pancreatitis
Inflammation of the pancreas. Pancreatitis is classified as acute unless there are computed tomographic or endoscopic retrograde cholangiopancreatographic findings of chronic pancreatitis. The two most common forms of acute pancreatitis are alcoholic pancreatitis and gallstone pancreatitis.
Acute Pancreatitis Decreased in:
|
| Trypsinogen | Serum trypsinogen | Increased in: acute pancreatitis Pancreatitis Inflammation of the pancreas. Pancreatitis is classified as acute unless there are computed tomographic or endoscopic retrograde cholangiopancreatographic findings of chronic pancreatitis. The two most common forms of acute pancreatitis are alcoholic pancreatitis and gallstone pancreatitis. Acute Pancreatitis |
| Chymotrypsin | Chymotrypsin (feces) | Pancreatic insufficiency: negative test |