The skull (cranium) is the skeletal structure of the head supporting the face and forming a protective cavity for the brain Brain The part of central nervous system that is contained within the skull (cranium). Arising from the neural tube, the embryonic brain is comprised of three major parts including prosencephalon (the forebrain); mesencephalon (the midbrain); and rhombencephalon (the hindbrain). The developed brain consists of cerebrum; cerebellum; and other structures in the brain stem. Nervous System: Anatomy, Structure, and Classification. The skull consists of 22 bones divided into the viscerocranium (facial skeleton) and the neurocranium. The neurocranium is further subdivided into the calvarium (the skullcap) and the base of the skull (the cranial fossa).
Last updated: Jan 18, 2024
The viscerocranium is responsible for maintaining the structure for the muscles of the face Muscles of the Face The facial muscles (also called mimetic muscles) control facial expression and are supplied by the facial nerve. Most of them originate from the skull and attach to the skin around the facial openings, which serve as a method to group or classify them. Facial Muscles: Anatomy. The muscles are arranged in a complex anatomical pattern to allow for a variety of facial functions such as mastication Mastication The act and process of chewing and grinding food in the mouth. Jaw and Temporomandibular Joint: Anatomy, smiling, frowning, eye movement, and patency of the nasal passage.
| Viscerocranium bone Bone Bone is a compact type of hardened connective tissue composed of bone cells, membranes, an extracellular mineralized matrix, and central bone marrow. The 2 primary types of bone are compact and spongy. Bones: Structure and Types | Number of bones | Facial features |
|---|---|---|
| Zygomatic | Paired | Cheekbone |
| Lacrimal | Paired | Anterior medial wall of the orbit |
| Nasal | Paired | Bridge of the nose Nose The nose is the human body’s primary organ of smell and functions as part of the upper respiratory system. The nose may be best known for inhaling oxygen and exhaling carbon dioxide, but it also contributes to other important functions, such as tasting. The anatomy of the nose can be divided into the external nose and the nasal cavity. Nose Anatomy (External & Internal) |
| Palatine | Paired | Posterior quarter of the hard palate Hard palate The anteriorly located rigid section of the palate. Palate: Anatomy |
| Maxilla | Paired | Upper jaw Jaw The jaw is made up of the mandible, which comprises the lower jaw, and the maxilla, which comprises the upper jaw. The mandible articulates with the temporal bone via the temporomandibular joint (TMJ). The 4 muscles of mastication produce the movements of the TMJ to ensure the efficient chewing of food. Jaw and Temporomandibular Joint: Anatomy and ¾ of the hard palate Hard palate The anteriorly located rigid section of the palate. Palate: Anatomy (the palatine process of the maxilla) |
| Vomer | Single | Posterior portion of the nasal septum Nasal septum The partition separating the two nasal cavities in the midplane. It is formed by the septal nasal cartilage, parts of skull bones, and membranous parts. Nose Anatomy (External & Internal) |
| Mandible Mandible The largest and strongest bone of the face constituting the lower jaw. It supports the lower teeth. Jaw and Temporomandibular Joint: Anatomy | Single | Lower jaw Jaw The jaw is made up of the mandible, which comprises the lower jaw, and the maxilla, which comprises the upper jaw. The mandible articulates with the temporal bone via the temporomandibular joint (TMJ). The 4 muscles of mastication produce the movements of the TMJ to ensure the efficient chewing of food. Jaw and Temporomandibular Joint: Anatomy and chin Chin The anatomical frontal portion of the mandible, also known as the mentum, that contains the line of fusion of the two separate halves of the mandible (symphysis menti). This line of fusion divides inferiorly to enclose a triangular area called the mental protuberance. On each side, inferior to the second premolar tooth, is the mental foramen for the passage of blood vessels and a nerve. Melasma |
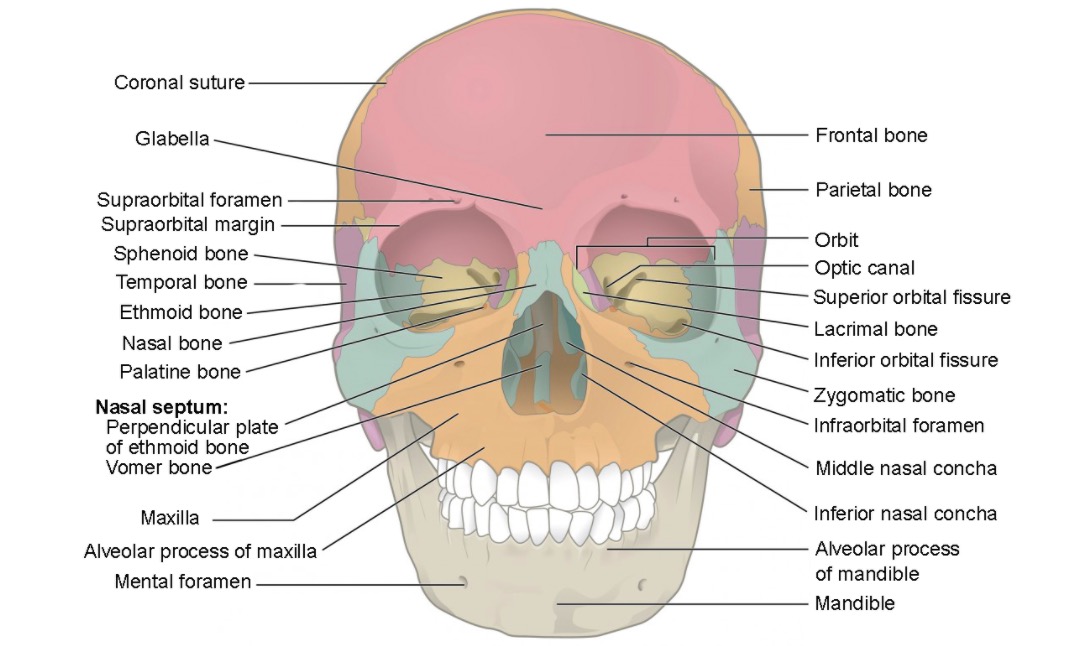
Anterior view of the skull featuring the bones of the viscerocranium:
Zygomatic (blue), lacrimal (green), nasal (blue/midline), inferior nasal conchae (blue inside the nasal cavity), palatine (dark orange), maxillary (light orange), vomer (blue/midline), mandible (beige), and ethmoid (gray within the nasal cavity)
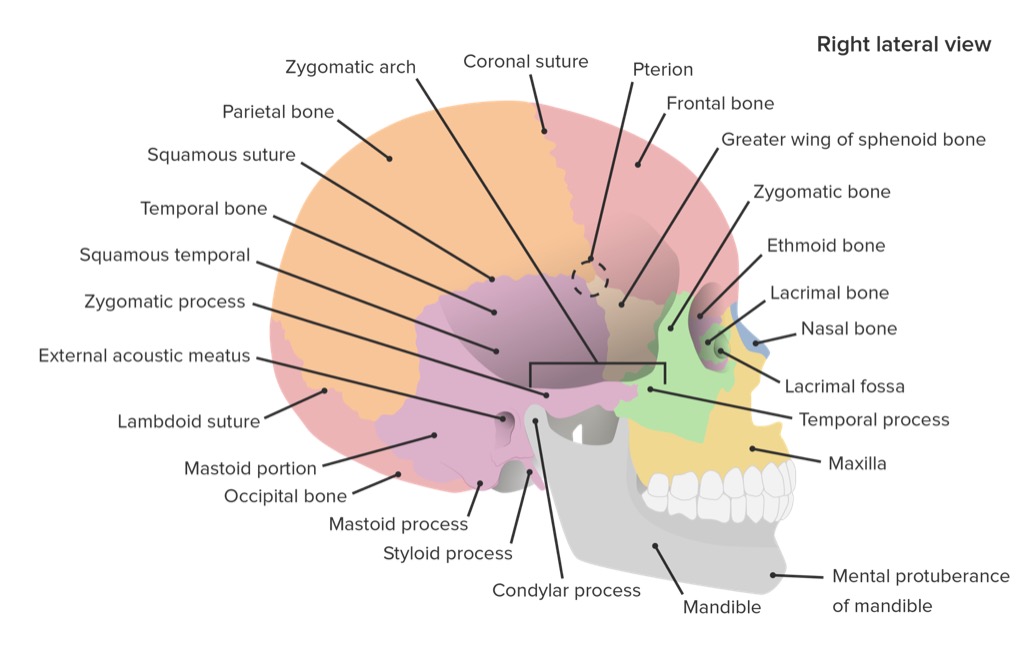
Lateral view of the skull featuring the bones and sutures
Image by Lecturio.The neurocranium is the complex of bones encasing the brain Brain The part of central nervous system that is contained within the skull (cranium). Arising from the neural tube, the embryonic brain is comprised of three major parts including prosencephalon (the forebrain); mesencephalon (the midbrain); and rhombencephalon (the hindbrain). The developed brain consists of cerebrum; cerebellum; and other structures in the brain stem. Nervous System: Anatomy, Structure, and Classification. The calvarium is the upper portion of the neurocranium, and consists of the unpaired frontal bone Bone Bone is a compact type of hardened connective tissue composed of bone cells, membranes, an extracellular mineralized matrix, and central bone marrow. The 2 primary types of bone are compact and spongy. Bones: Structure and Types, unpaired occipital bone Bone Bone is a compact type of hardened connective tissue composed of bone cells, membranes, an extracellular mineralized matrix, and central bone marrow. The 2 primary types of bone are compact and spongy. Bones: Structure and Types, bilateral parietal bones, and bilateral temporal bones. The bones are initially separated (the fontanelles Fontanelles Physical Examination of the Newborn at birth) and fuse over childhood. The bones are then connected by specific sutures: coronal Coronal Computed Tomography (CT), squamous, lambdoid, and sagittal Sagittal Computed Tomography (CT).
Bones of the neurocranium:
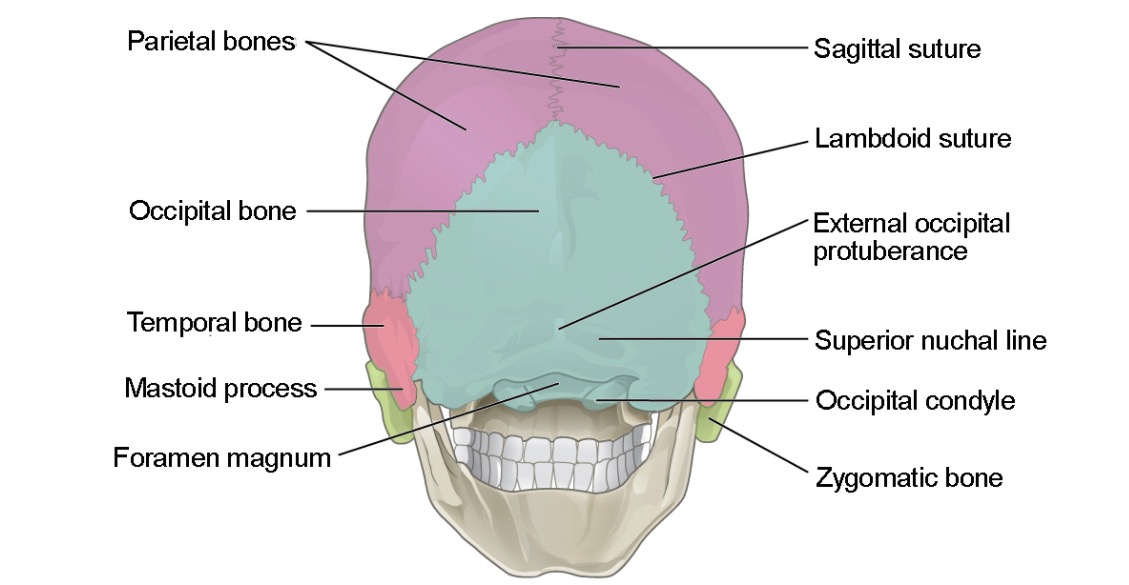
Posterior (slightly inferior) view of the skull featuring the bones of the calvarium:
the frontal bone (not pictured), occipital bone (blue), and parietal bone (purple)

Lateral view of the skull featuring the bones and sutures of the skull
Image by Lecturio.Main sutures are immobile joints between adjacent bones of the skull:

Lateral view of the skull featuring the coronal, lambdoid, and squamous sutures
Image: “Skull sutures” by OpenStax College. License: CC BY 3.0Major fontanelles Fontanelles Physical Examination of the Newborn are areas of connective tissue Connective tissue Connective tissues originate from embryonic mesenchyme and are present throughout the body except inside the brain and spinal cord. The main function of connective tissues is to provide structural support to organs. Connective tissues consist of cells and an extracellular matrix. Connective Tissue: Histology within the gaps/between the bone Bone Bone is a compact type of hardened connective tissue composed of bone cells, membranes, an extracellular mineralized matrix, and central bone marrow. The 2 primary types of bone are compact and spongy. Bones: Structure and Types plates, which are open during infancy:
| Fontanelle | Shape | Location | Closure |
|---|---|---|---|
| Posterior, occipital, or lambdoid | Triangle | Junction of the sagittal Sagittal Computed Tomography (CT) and lambdoid sutures | 3 months |
| Anterior, bregmatic, or frontal | Diamond | Junction of the sagittal Sagittal Computed Tomography (CT) and coronal Coronal Computed Tomography (CT) sutures | 18–24 months |
| Sphenoidal or anterolateral | Irregular and bilateral | Between the frontal, parietal, and sphenoid bones | 6 months |
| Mastoid or posterolateral | Irregular and bilateral | Between the parietal, temporal, and occipital bones | 24 months |
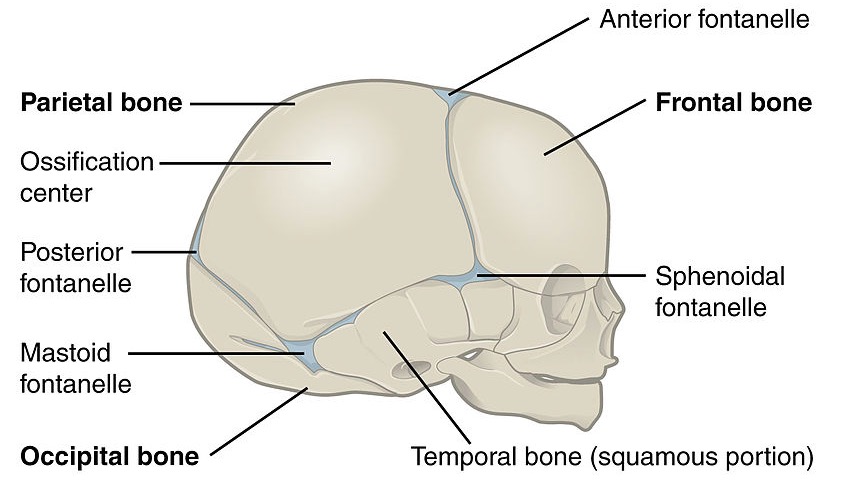
Lateral view of a newborn skull featuring the posterior, anterior, sphenoidal, and mastoid fontanelles
Image: “Lateral view of a newborn’s skull” by OpenStax College. License: CC BY 3.0The skull base is a highly complex region of the skull with many important anatomical landmarks and foramen for cranial nerves Cranial nerves There are 12 pairs of cranial nerves (CNs), which run from the brain to various parts of the head, neck, and trunk. The CNs can be sensory or motor or both. The CNs are named and numbered in Roman numerals according to their location, from the front to the back of the brain. The 12 Cranial Nerves: Overview and Functions and vasculature to travel in and out of the skull. In-depth knowledge of the location of the foramen and the structures traversing the foramen are critically important.
| Region | Boundaries | Bones | Structures penetrating the foramina | Content |
|---|---|---|---|---|
| Anterior cranial fossa |
|
Frontal (orbital plates) | Foramen cecum Cecum The blind sac or outpouching area of the large intestine that is below the entrance of the small intestine. It has a worm-like extension, the vermiform appendix. Colon, Cecum, and Appendix: Anatomy: nasal emissary veins Veins Veins are tubular collections of cells, which transport deoxygenated blood and waste from the capillary beds back to the heart. Veins are classified into 3 types: small veins/venules, medium veins, and large veins. Each type contains 3 primary layers: tunica intima, tunica media, and tunica adventitia. Veins: Histology leading to the superior sagittal Sagittal Computed Tomography (CT) sinus |
|
| Ethmoid (cribriform plate and crista galli) |
|
|||
| Middle cranial fossa |
|
Sphenoid (lesser and greater wings) |
|
|
| Sphenoid (greater wings) |
|
|||
| Temporal |
|
|||
| Posterior cranial fossa | Anterior: dorsum sellae of the sphenoid/superior border of the petrous part of the temporal bones | Temporal |
|
|
| Occipital |
|
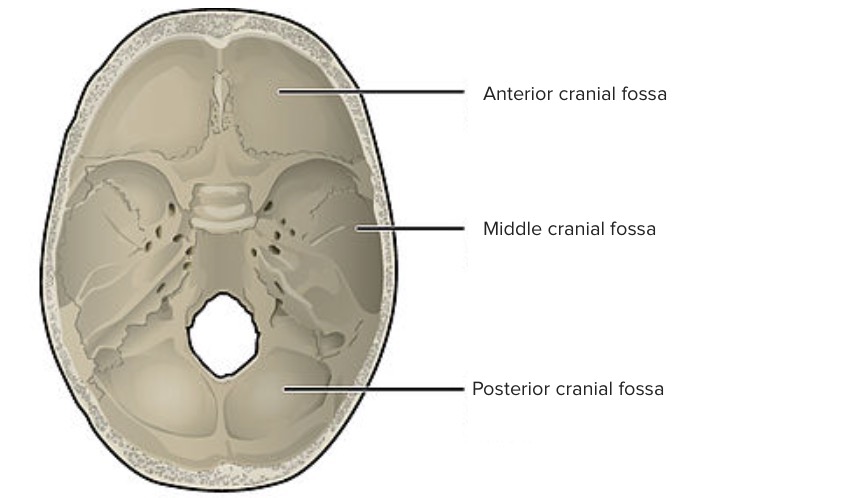
Superior view of the base of the skull:
The internal surface of the cranial base is divided into anterior, middle, and posterior cranial fossa. The fossae contain the frontal and temporal lobes of the cerebral hemispheres, the cerebellum, the pons, and the medulla oblongata.
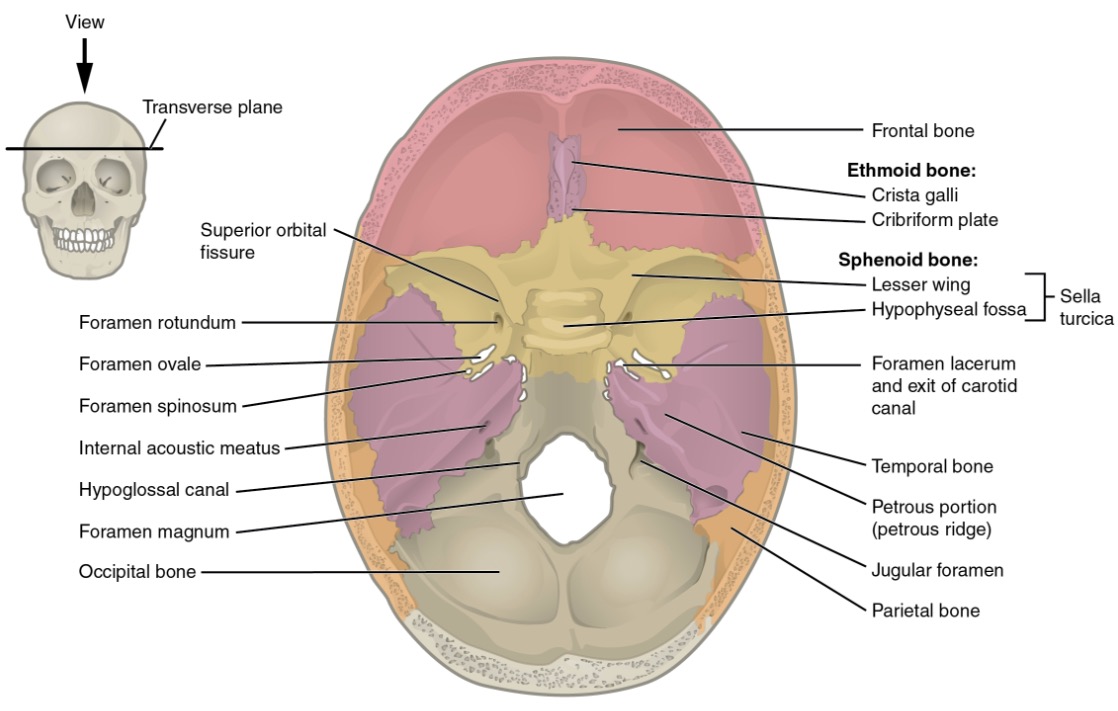
Superior view of the base of the skull featuring the borders of the cranial fossa
Image: “Superior view of skull base” by OpenStax College. License: CC BY 3.0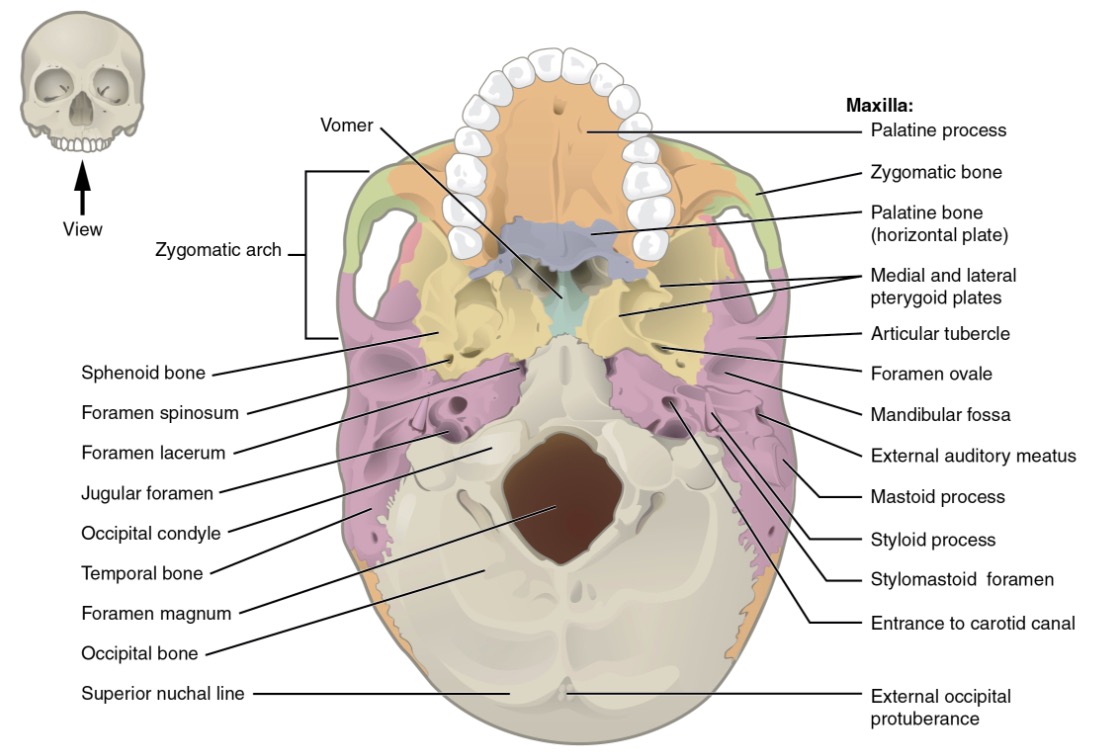
Inferior view of the skull featuring the various foramina
Image: “Inferior view of skull base” by OpenStax College. License: CC BY 3.0| Temporal fossa* | Infratemporal fossa | Pterygopalatine fossa | |
|---|---|---|---|
| Location | Above the zygomatic arch of the temporal bone Temporal bone Either of a pair of compound bones forming the lateral (left and right) surfaces and base of the skull which contains the organs of hearing. It is a large bone formed by the fusion of parts: the squamous (the flattened anterior-superior part), the tympanic (the curved anterior-inferior part), the mastoid (the irregular posterior portion), and the petrous (the part at the base of the skull). Jaw and Temporomandibular Joint: Anatomy |
|
|
| Boundaries |
|
|
|
| Content |
|
|
|
| Foramina (and the connecting structure) | Inferior orbital fissure Fissure A crack or split that extends into the dermis Generalized and Localized Rashes: orbit |
|
|
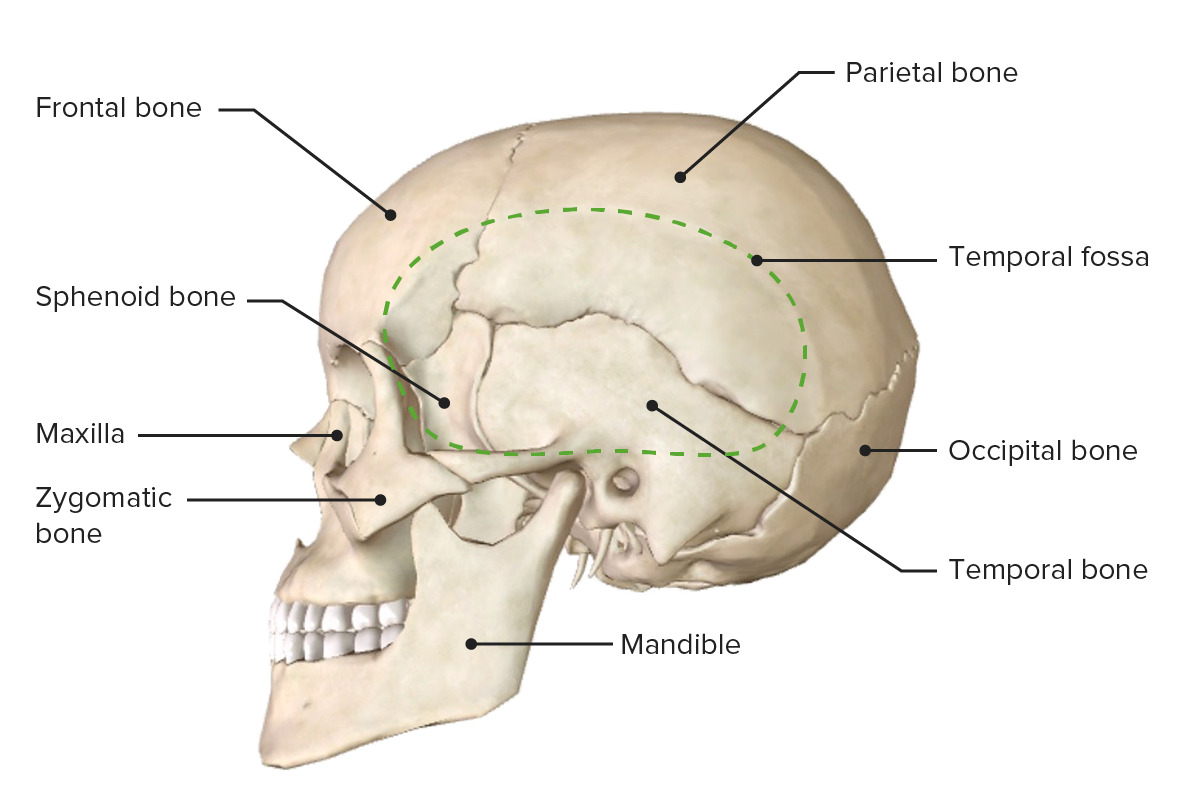
Lateral view of the skull featuring the temporal fossa
Image by BioDigital, edited by Lecturio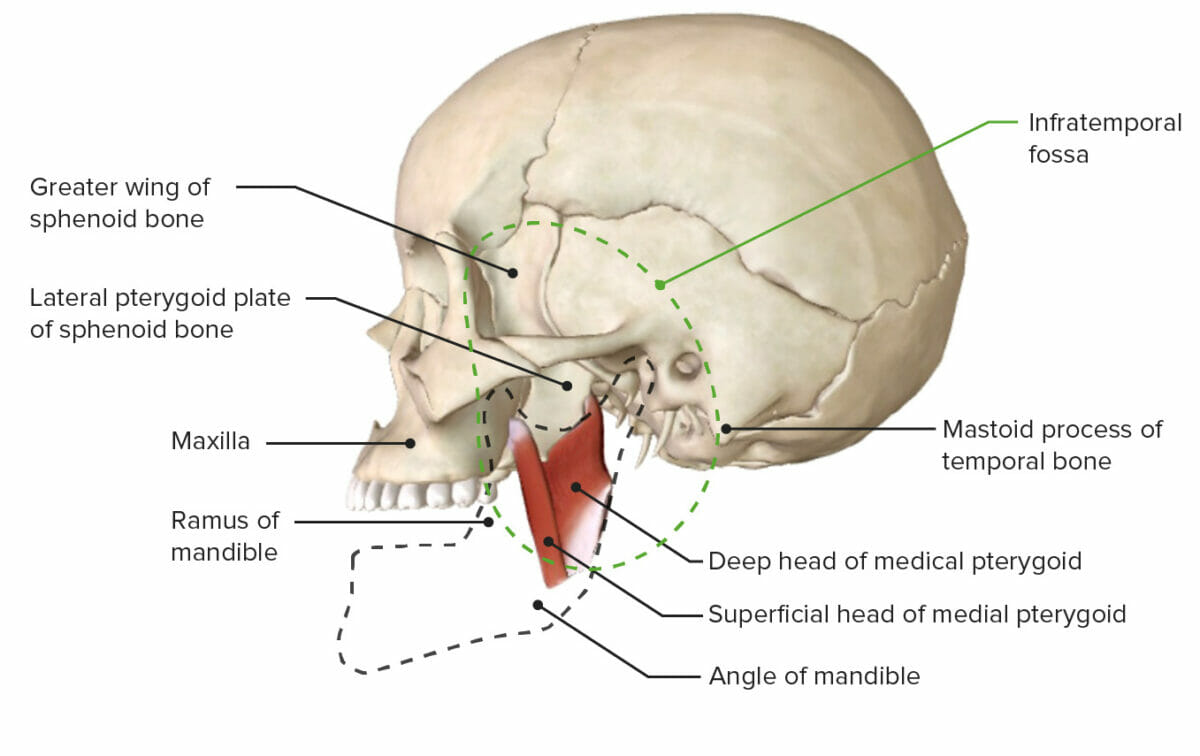
Lateral view of the base of the skull featuring the infratemporal fossa
Image by BioDigital, edited by Lecturio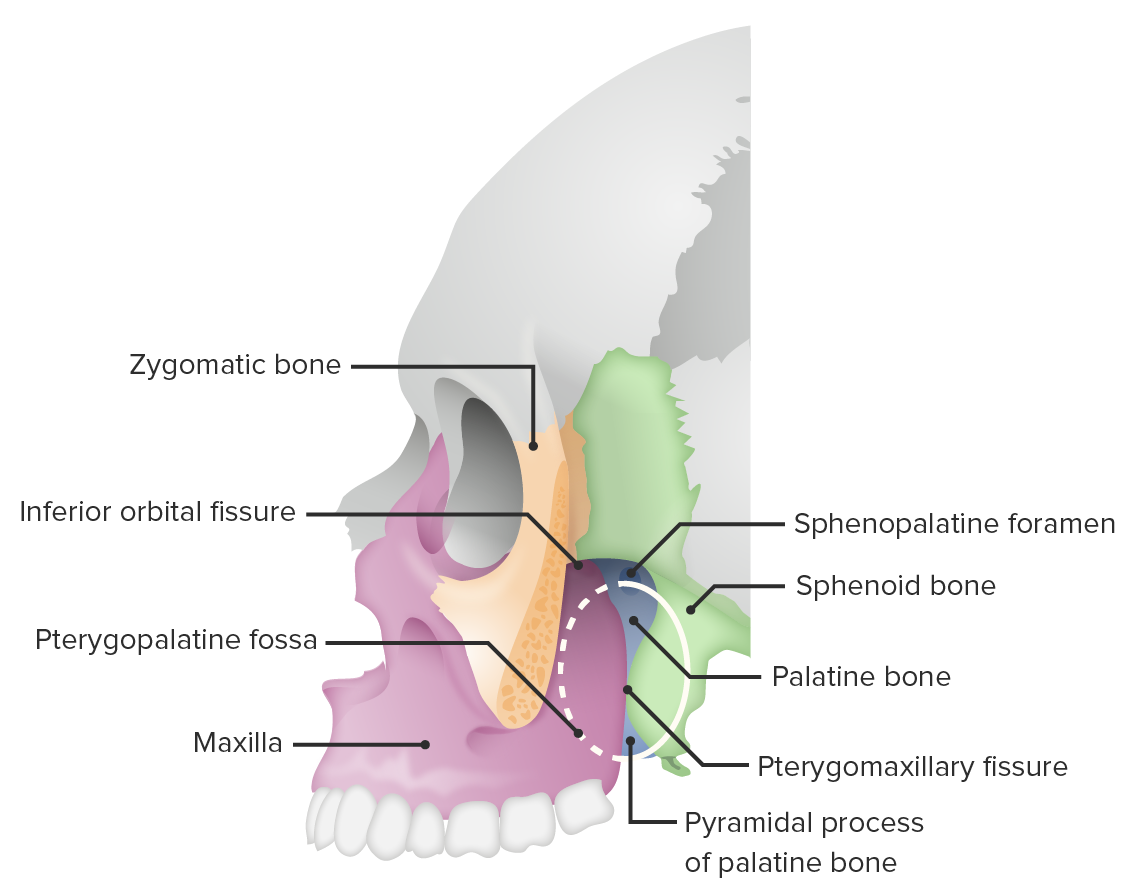
Lateral view of the skull (zygomatic bone cut) featuring the pterygopalatine fossa
Image by BioDigital, edited by Lecturio