There are 2 types of sex chromosomes Sex chromosomes The homologous chromosomes that are dissimilar in the heterogametic sex. There are the X chromosome, the Y chromosome, and the w, z chromosomes (in animals in which the female is the heterogametic sex (the silkworm moth bombyx mori, for example)). In such cases the w chromosome is the female-determining and the male is zz. Basic Terms of Genetics in humans: X and Y. Chromosomal sex Sex The totality of characteristics of reproductive structure, functions, phenotype, and genotype, differentiating the male from the female organism. Gender Dysphoria is male when a Y chromosome Y chromosome The male sex chromosome, being the differential sex chromosome carried by half the male gametes and none of the female gametes in humans and in some other male-heterogametic species in which the homologue of the X chromosome has been retained. Basic Terms of Genetics is present (e.g., 46,XY or 47, XXY XXY Klinefelter syndrome is a chromosomal aneuploidy characterized by the presence of 1 or more extra X chromosomes in a male karyotype, most commonly leading to karyotype 47,XXY. Klinefelter syndrome is associated with decreased levels of testosterone and is the most common cause of congenital hypogonadism. Klinefelter Syndrome) and female when the Y chromosome Y chromosome The male sex chromosome, being the differential sex chromosome carried by half the male gametes and none of the female gametes in humans and in some other male-heterogametic species in which the homologue of the X chromosome has been retained. Basic Terms of Genetics is absent (e.g., 46,XX or 45,X0). Male phenotypes develop when a specific gene Gene A category of nucleic acid sequences that function as units of heredity and which code for the basic instructions for the development, reproduction, and maintenance of organisms. Basic Terms of Genetics, called the SRY gene Gene A category of nucleic acid sequences that function as units of heredity and which code for the basic instructions for the development, reproduction, and maintenance of organisms. Basic Terms of Genetics (usually found on the Y chromosome Y chromosome The male sex chromosome, being the differential sex chromosome carried by half the male gametes and none of the female gametes in humans and in some other male-heterogametic species in which the homologue of the X chromosome has been retained. Basic Terms of Genetics), is present, stimulating differentiation of the gonads Gonads The gamete-producing glands, ovary or testis. Hormones: Overview and Types into testes Testes Gonadal Hormones. The testes Testes Gonadal Hormones then produce testosterone Testosterone A potent androgenic steroid and major product secreted by the leydig cells of the testis. Its production is stimulated by luteinizing hormone from the pituitary gland. In turn, testosterone exerts feedback control of the pituitary LH and FSH secretion. Depending on the tissues, testosterone can be further converted to dihydrotestosterone or estradiol. Androgens and Antiandrogens (triggering development of the penis Penis The penis is the male organ of copulation and micturition. The organ is composed of a root, body, and glans. The root is attached to the pubic bone by the crura penis. The body consists of the 2 parallel corpora cavernosa and the corpus spongiosum. The glans is ensheathed by the prepuce or foreskin. Penis: Anatomy and scrotum Scrotum A cutaneous pouch of skin containing the testicles and spermatic cords. Testicles: Anatomy externally and the ejaculatory system internally) and antimüllerian hormone ( AMH AMH A glycoprotein that causes regression of mullerian ducts. It is produced by sertoli cells of the testes. In the absence of this hormone, the mullerian ducts develop into structures of the female reproductive tract. In males, defects of this hormone result in persistent mullerian duct, a form of male pseudohermaphroditism. Primary Amenorrhea), which causes regression Regression Corneal Abrasions, Erosion, and Ulcers of the müllerian ducts. Without the SRY gene Gene A category of nucleic acid sequences that function as units of heredity and which code for the basic instructions for the development, reproduction, and maintenance of organisms. Basic Terms of Genetics, ovaries Ovaries Ovaries are the paired gonads of the female reproductive system that contain haploid gametes known as oocytes. The ovaries are located intraperitoneally in the pelvis, just posterior to the broad ligament, and are connected to the pelvic sidewall and to the uterus by ligaments. These organs function to secrete hormones (estrogen and progesterone) and to produce the female germ cells (oocytes). Ovaries: Anatomy develop; without testosterone Testosterone A potent androgenic steroid and major product secreted by the leydig cells of the testis. Its production is stimulated by luteinizing hormone from the pituitary gland. In turn, testosterone exerts feedback control of the pituitary LH and FSH secretion. Depending on the tissues, testosterone can be further converted to dihydrotestosterone or estradiol. Androgens and Antiandrogens, external female genitalia develop; and without AMH AMH A glycoprotein that causes regression of mullerian ducts. It is produced by sertoli cells of the testes. In the absence of this hormone, the mullerian ducts develop into structures of the female reproductive tract. In males, defects of this hormone result in persistent mullerian duct, a form of male pseudohermaphroditism. Primary Amenorrhea, the müllerian ducts persist and differentiate into the fallopian tubes Fallopian tubes The uterus, cervix, and fallopian tubes are part of the internal female reproductive system. The fallopian tubes receive an ovum after ovulation and help move it and/or a fertilized embryo toward the uterus via ciliated cells lining the tubes and peristaltic movements of its smooth muscle. Uterus, Cervix, and Fallopian Tubes: Anatomy, uterus Uterus The uterus, cervix, and fallopian tubes are part of the internal female reproductive system. The uterus has a thick wall made of smooth muscle (the myometrium) and an inner mucosal layer (the endometrium). The most inferior portion of the uterus is the cervix, which connects the uterine cavity to the vagina. Uterus, Cervix, and Fallopian Tubes: Anatomy, and upper vagina Vagina The vagina is the female genital canal, extending from the vulva externally to the cervix uteri internally. The structures have sexual, reproductive, and urinary functions and a rich blood supply, mainly arising from the internal iliac artery. Vagina, Vulva, and Pelvic Floor: Anatomy.
Last updated: Dec 22, 2022
The following definitions are relevant to the understanding of normal sex Sex The totality of characteristics of reproductive structure, functions, phenotype, and genotype, differentiating the male from the female organism. Gender Dysphoria development:
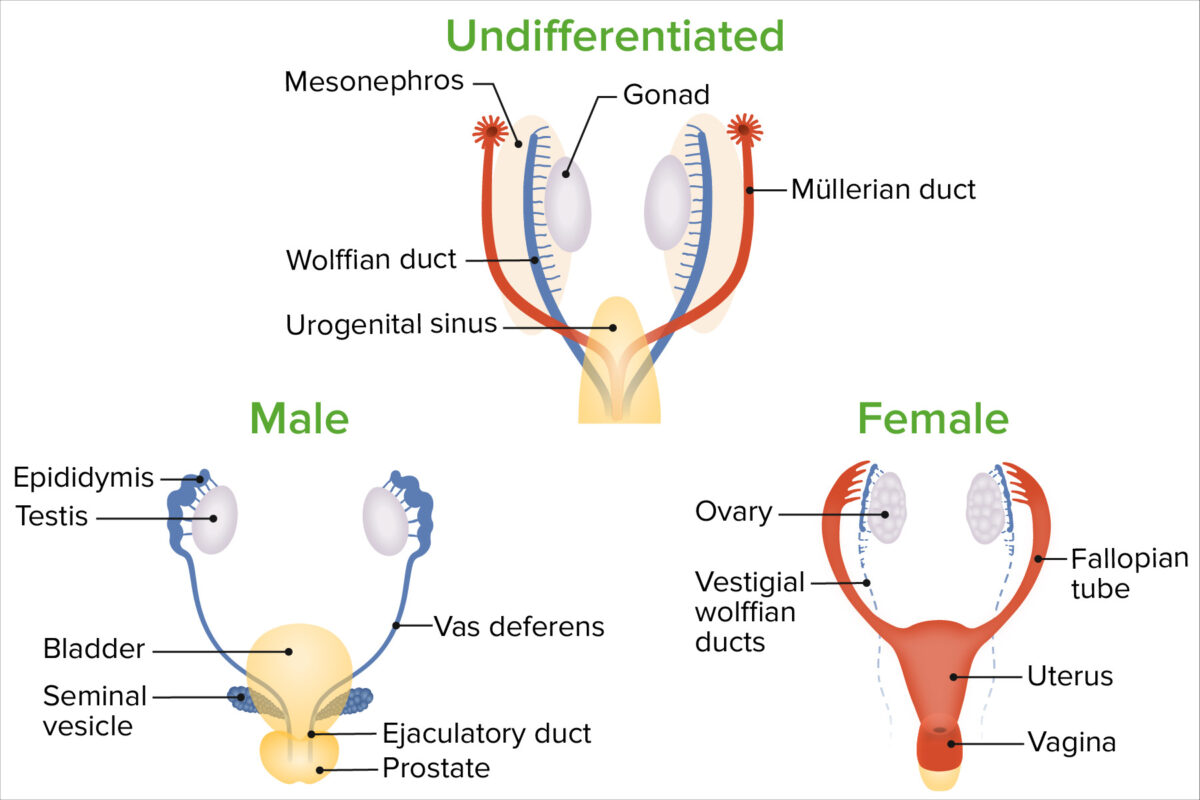
Embryologic sex differentiation between males and females
Image by Lecturio.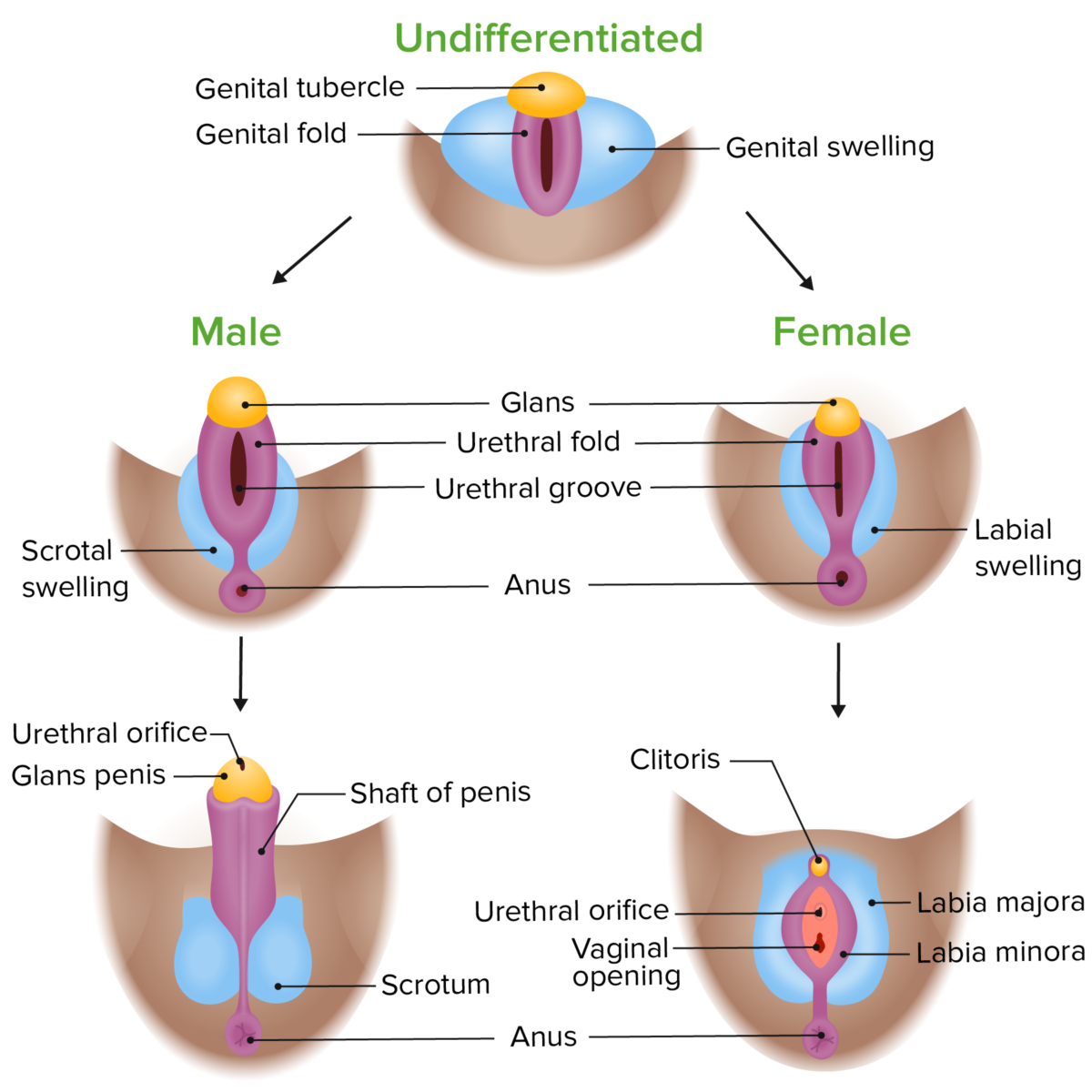
Phenotypic differentiation of the external genitalia in male and female embryos
Image by Lecturio. License: CC BY-NC-SA 4.0Secondary sexual characteristics Secondary Sexual Characteristics Precocious Puberty develop based on the hormonal milieu at puberty Puberty Puberty is a complex series of physical, psychosocial, and cognitive transitions usually experienced by adolescents (11-19 years of age). Puberty is marked by a growth in stature and the development of secondary sexual characteristics, achievement of fertility, and changes in most body systems. Puberty.
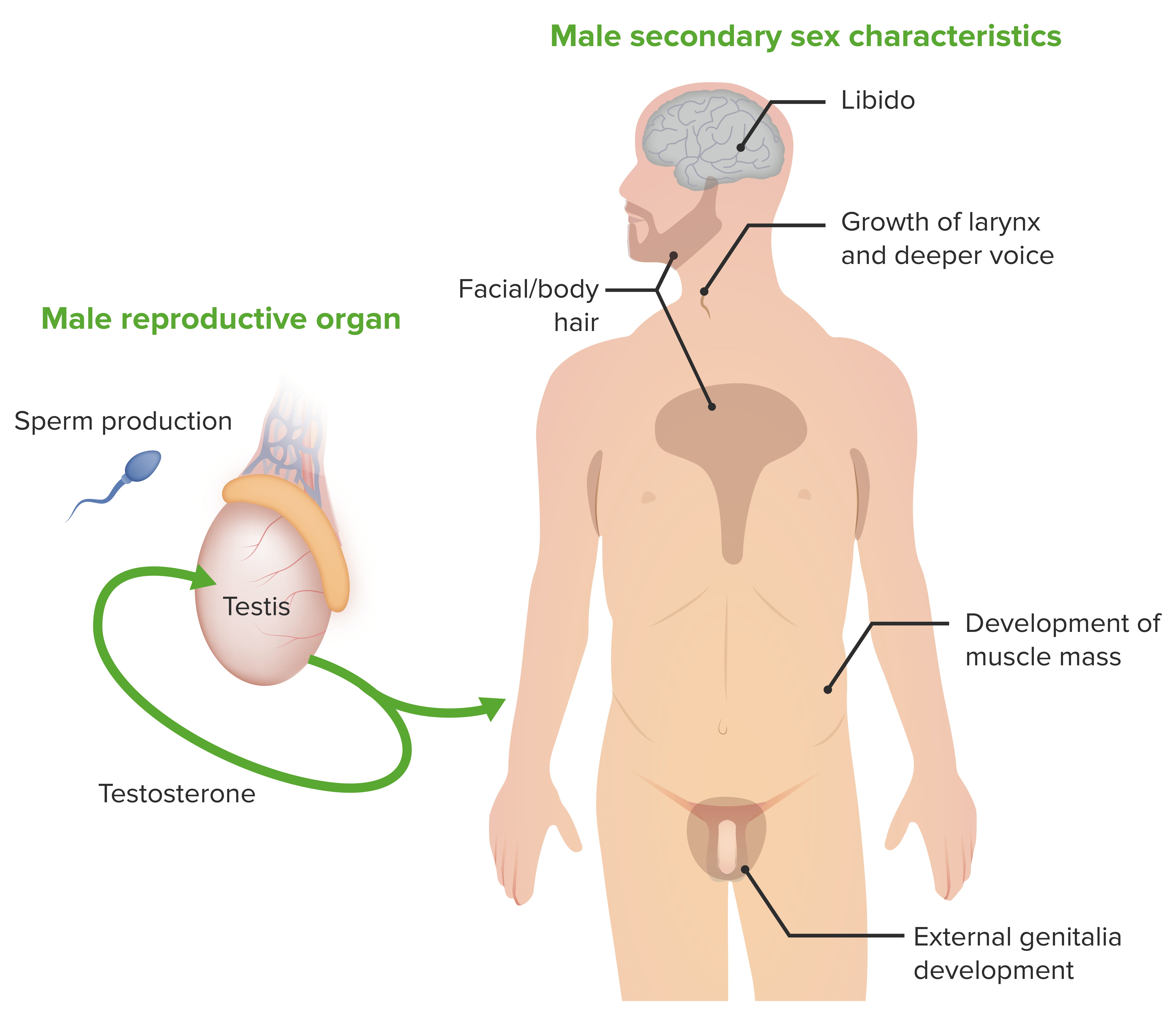
Illustration of the male secondary sex characteristics, the male reproductive organ, and testosterone, the hormone responsible for the characteristics
Image by Lecturio.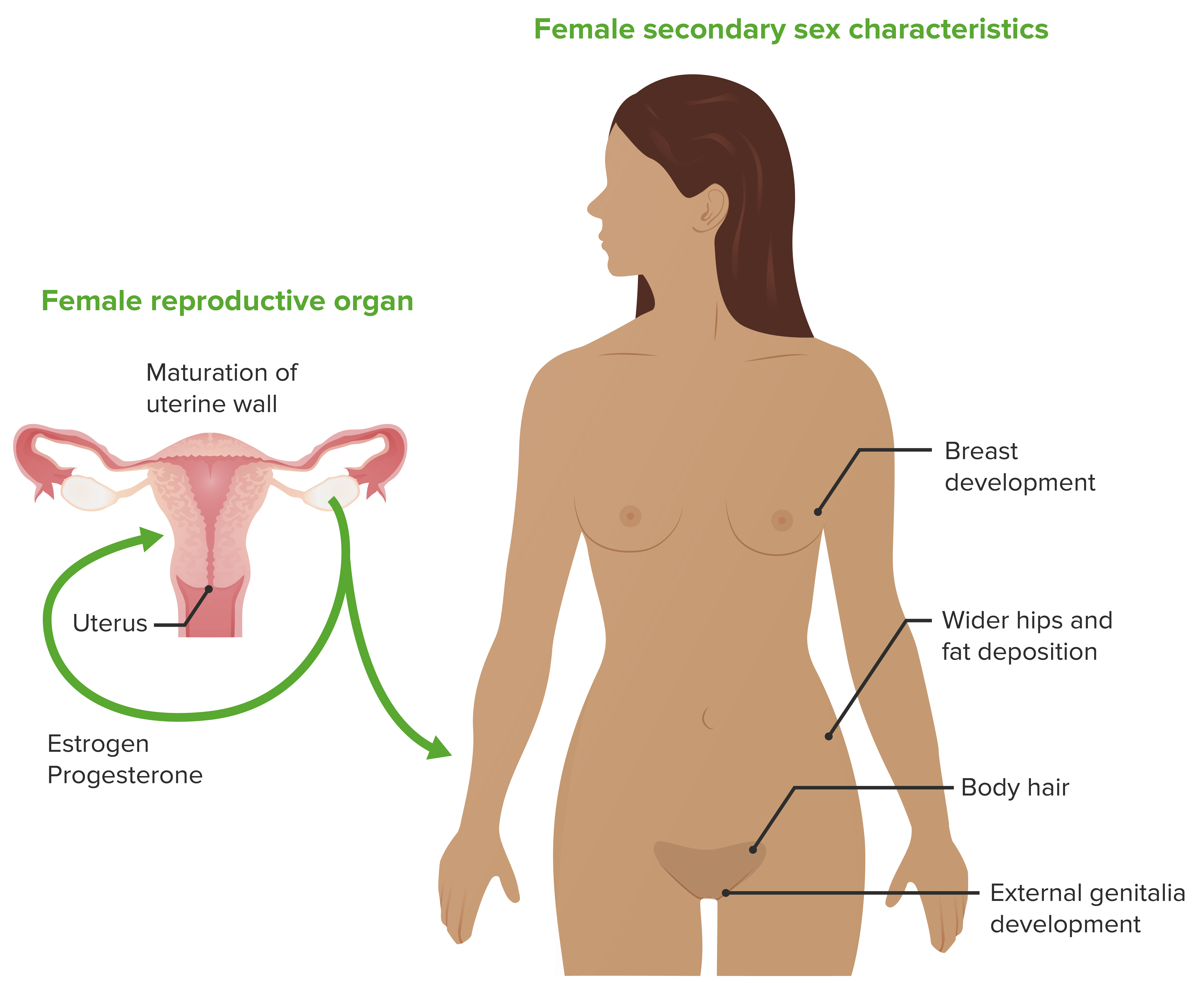
Illustration of the female secondary sex characteristics, the female reproductive organ, and estrogen and progesterone, the hormones responsible for the characteristics
Image by Lecturio.There are 2 sex chromosomes Sex chromosomes The homologous chromosomes that are dissimilar in the heterogametic sex. There are the X chromosome, the Y chromosome, and the w, z chromosomes (in animals in which the female is the heterogametic sex (the silkworm moth bombyx mori, for example)). In such cases the w chromosome is the female-determining and the male is zz. Basic Terms of Genetics in humans: X and Y.
A male set of chromosomes:
The Y chromosome is highlighted.
Differentiation and development of the müllerian ducts and external genitalia will proceed when testicular androgens Androgens Androgens are naturally occurring steroid hormones responsible for development and maintenance of the male sex characteristics, including penile, scrotal, and clitoral growth, development of sexual hair, deepening of the voice, and musculoskeletal growth. Androgens and Antiandrogens and AMH AMH A glycoprotein that causes regression of mullerian ducts. It is produced by sertoli cells of the testes. In the absence of this hormone, the mullerian ducts develop into structures of the female reproductive tract. In males, defects of this hormone result in persistent mullerian duct, a form of male pseudohermaphroditism. Primary Amenorrhea are absent.
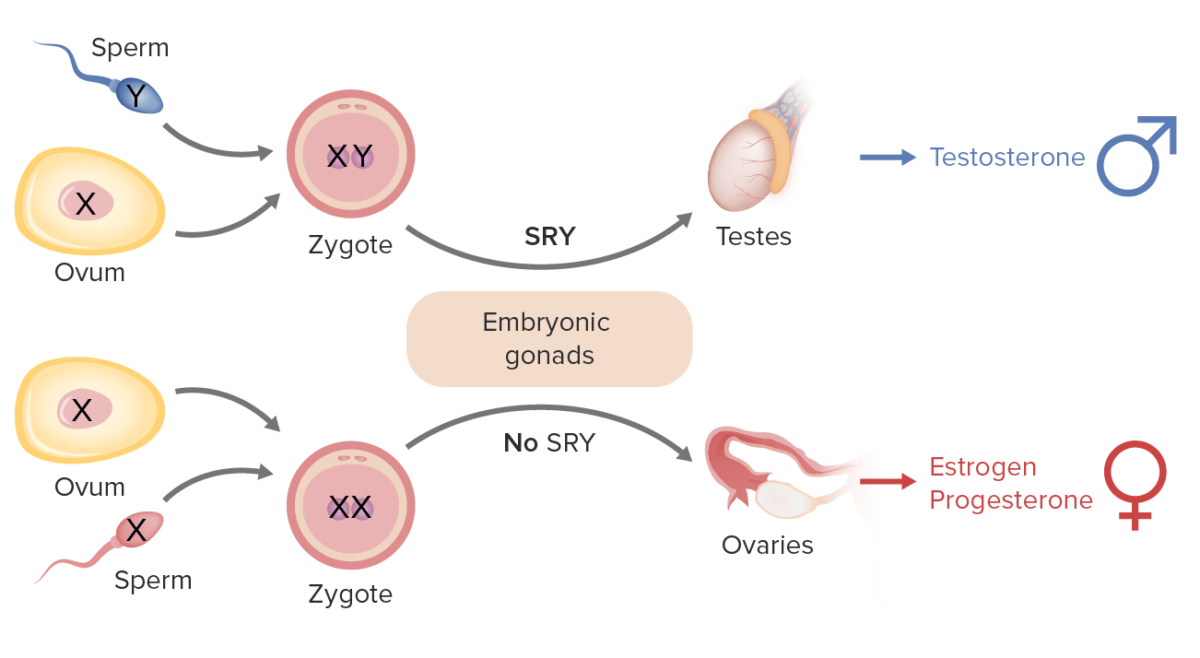
Sex determination in humans:
The Y chromosome includes the SRY gene (Sex-determining Region Y) that codes for testis-determining factor (TDF), causing embryonic gonads to form into testes (male gonads) and lead to male development.
In the absence of the TDF protein (i.e., no Y chromosome), the embryonic gonads develop into ovaries (female gonads).
Females possess 2 copies of the X chromosome (XX);
males possess 1 X and 1 Y (shorter) chromosome (XY).