Start your online physiology class
with Thad Wilson, PhD, from Kentucky University
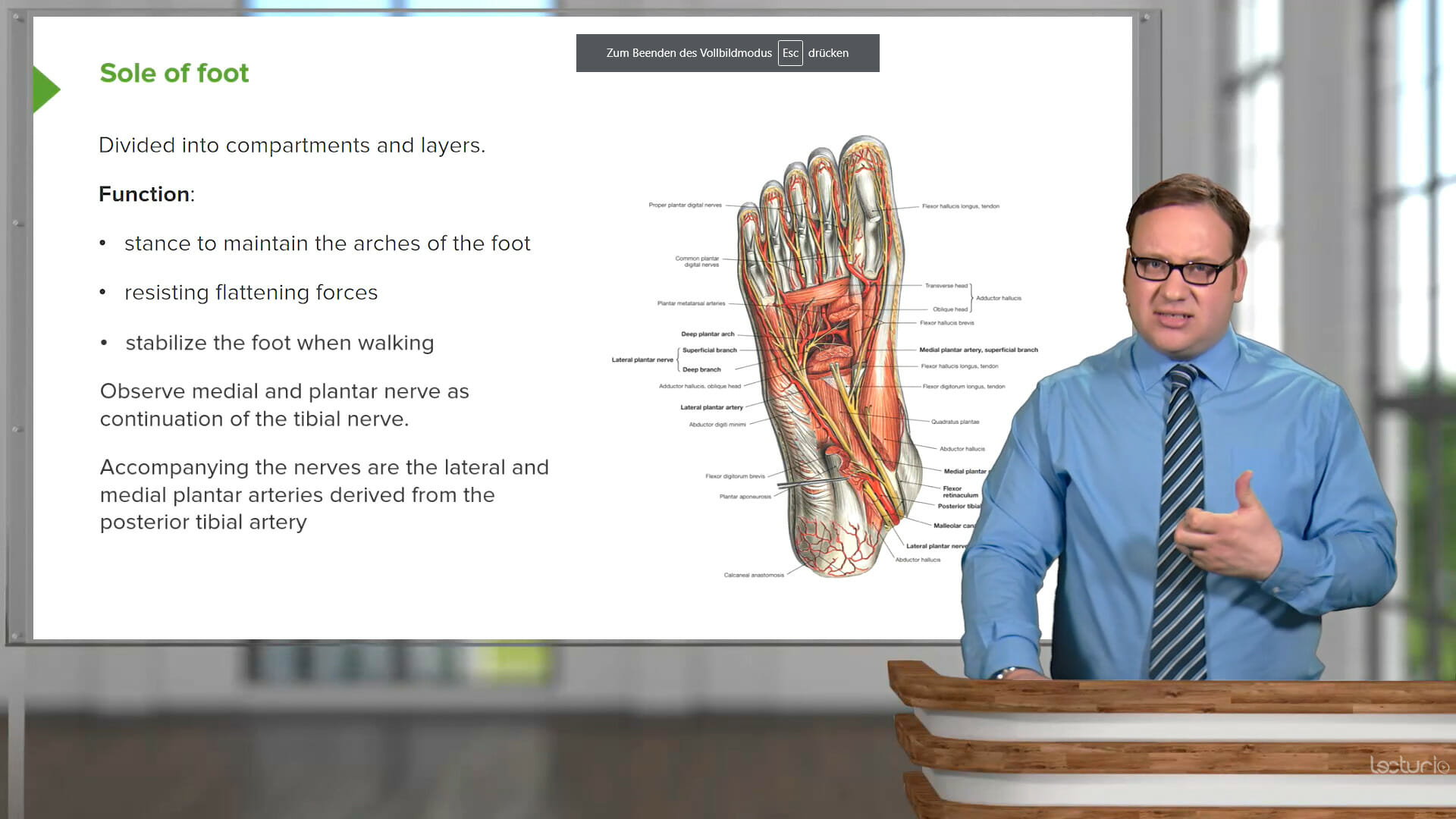
Physiology is the foundation of more complex concepts of pathology, in particular how disease processes are initiated when physiological processes are affected. However, the complexity of physiological processes across different body systems can be overwhelming.
This course covers the physiological processes of every organ system, including neurophysiology, musculoskeletal, cardiac and vascular, respiratory, GI and urinary, endocrine, and reproductive. Thad Wilson, PhD, from Kentucky University will help you grasp both the basic principles of physiology including the concept of homeostasis and the connections to how different disruptions in physiological processes lead to pathologies.
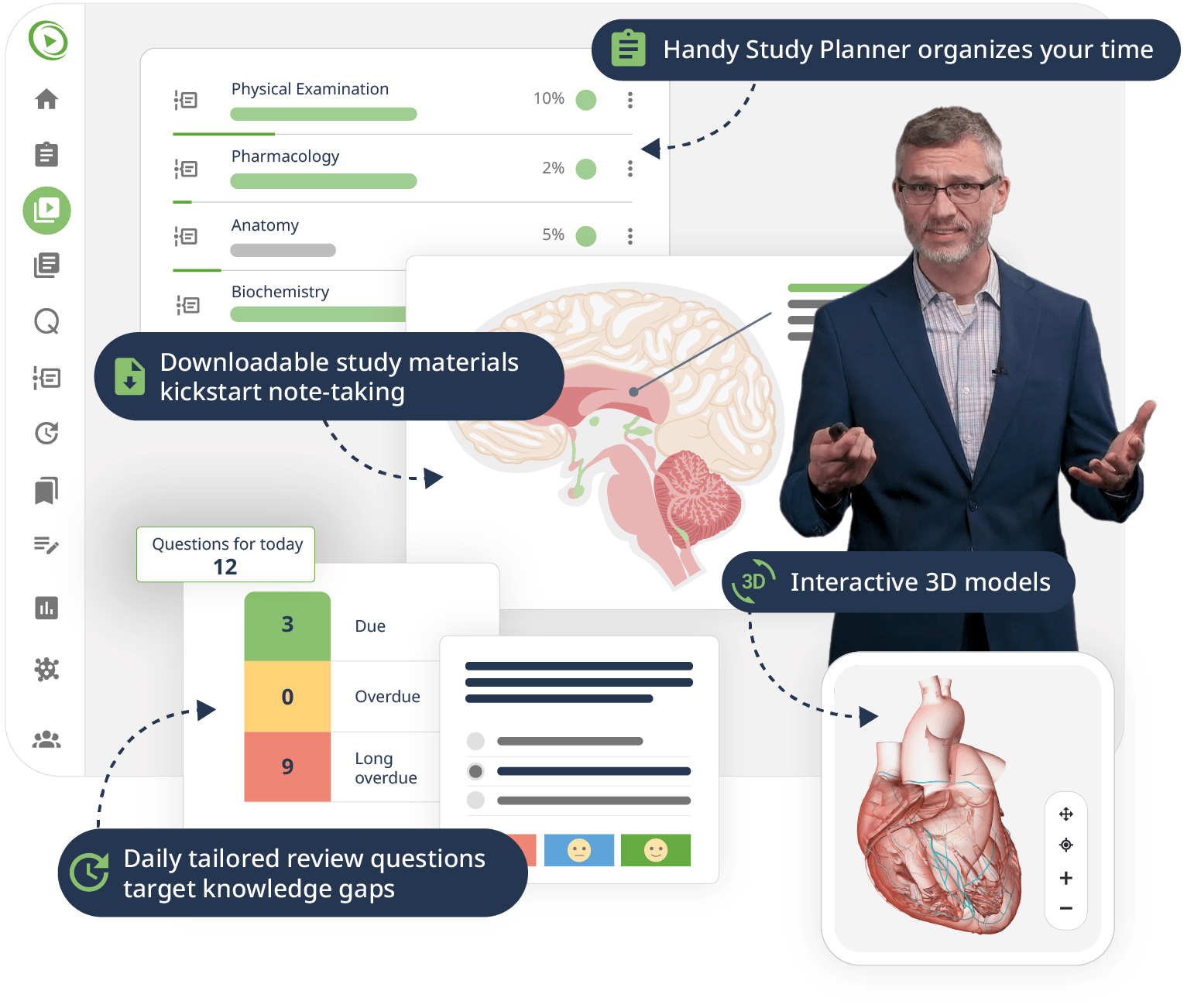
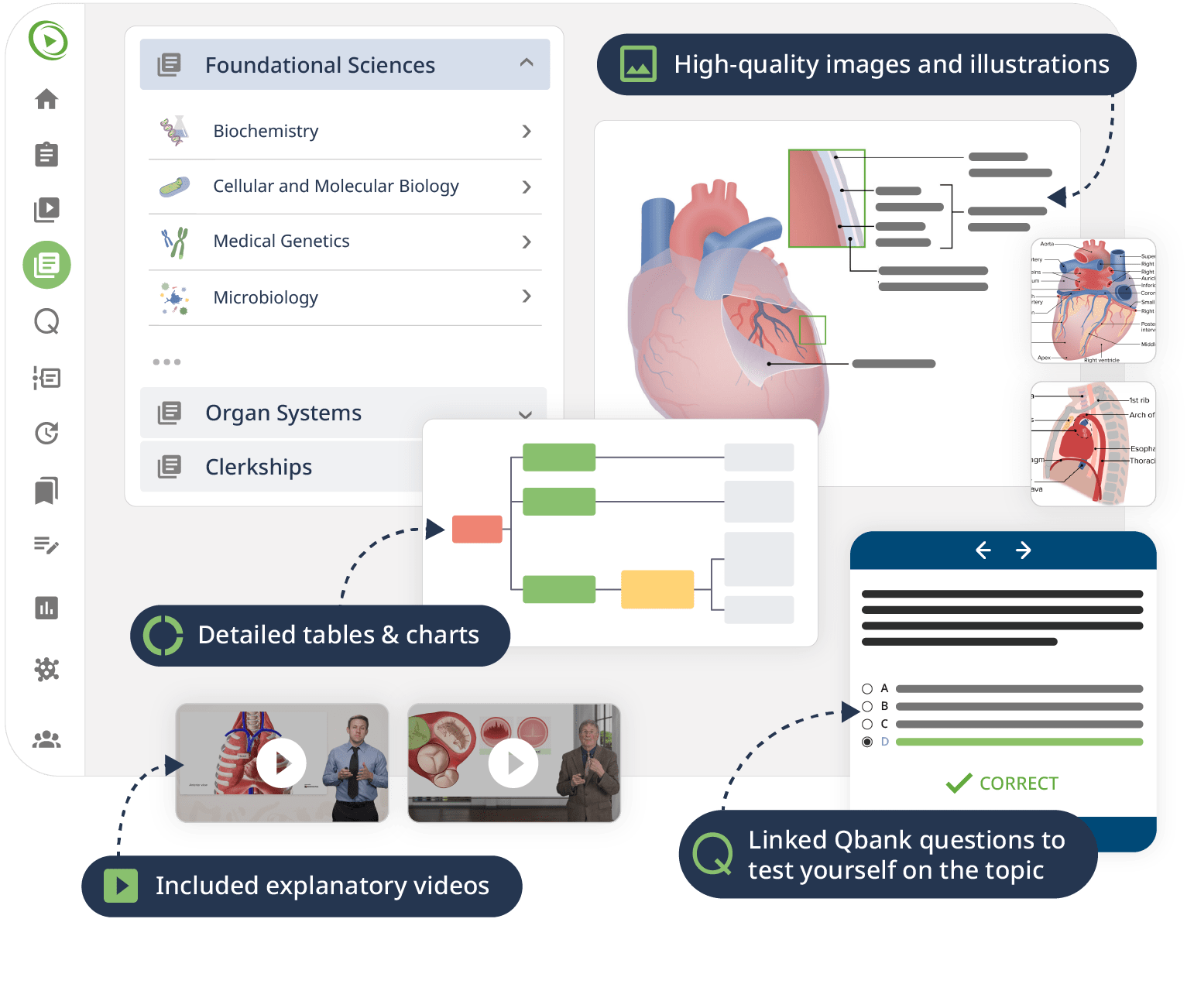
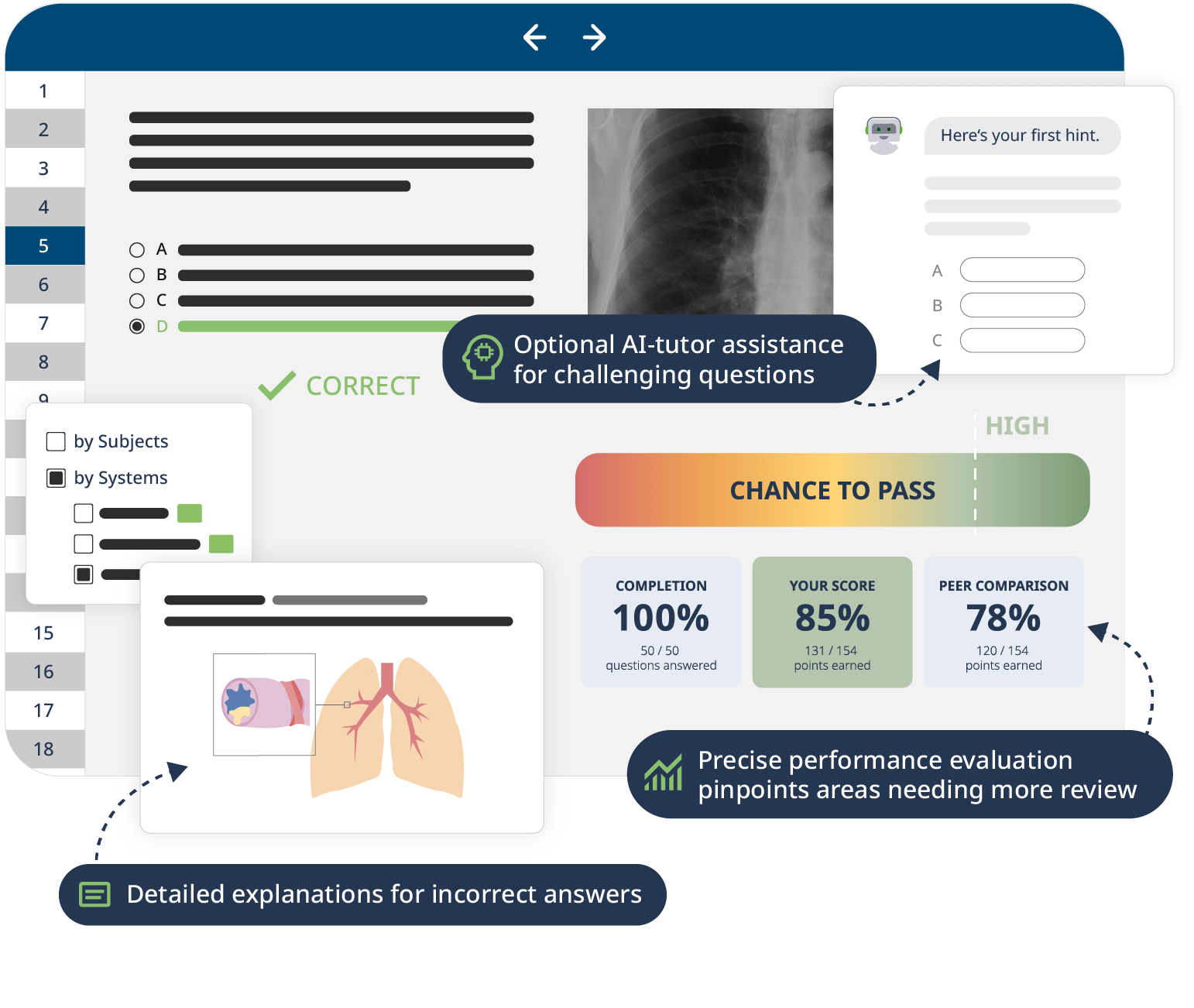
The combination of Video Lessons with interactive quiz questions, downloadable study materials, and a USMLE-style Qbank makes it easy to understand and retain the topics. By the end of this course, you will have a thorough understanding of human physiology, ready to apply this knowledge in your medical studies, exams, and future practice.