Lung development involves 5 stages: embryonic, pseudoglandular, canalicular, saccular, and alveolar. The inner respiratory epithelium Epithelium The epithelium is a complex of specialized cellular organizations arranged into sheets and lining cavities and covering the surfaces of the body. The cells exhibit polarity, having an apical and a basal pole. Structures important for the epithelial integrity and function involve the basement membrane, the semipermeable sheet on which the cells rest, and interdigitations, as well as cellular junctions. Surface Epithelium: Histology arises from the endoderm Endoderm The inner of the three germ layers of an embryo. Gastrulation and Neurulation, and the cartilage Cartilage Cartilage is a type of connective tissue derived from embryonic mesenchyme that is responsible for structural support, resilience, and the smoothness of physical actions. Perichondrium (connective tissue membrane surrounding cartilage) compensates for the absence of vasculature in cartilage by providing nutrition and support. Cartilage: Histology, bronchial muscles, connective tissue Connective tissue Connective tissues originate from embryonic mesenchyme and are present throughout the body except inside the brain and spinal cord. The main function of connective tissues is to provide structural support to organs. Connective tissues consist of cells and an extracellular matrix. Connective Tissue: Histology, and vasculature all arise from the mesoderm Mesoderm The middle germ layer of an embryo derived from three paired mesenchymal aggregates along the neural tube. Gastrulation and Neurulation. Starting in the embryonic stage at 4 weeks of development, the lung bud branches off the ventral side of the foregut Foregut Development of the Abdominal Organs, forming the esophagus Esophagus The esophagus is a muscular tube-shaped organ of around 25 centimeters in length that connects the pharynx to the stomach. The organ extends from approximately the 6th cervical vertebra to the 11th thoracic vertebra and can be divided grossly into 3 parts: the cervical part, the thoracic part, and the abdominal part. Esophagus: Anatomy posteriorly and the trachea Trachea The trachea is a tubular structure that forms part of the lower respiratory tract. The trachea is continuous superiorly with the larynx and inferiorly becomes the bronchial tree within the lungs. The trachea consists of a support frame of semicircular, or C-shaped, rings made out of hyaline cartilage and reinforced by collagenous connective tissue. Trachea: Anatomy anteriorly. In the pseudomembranous stage, the trachea Trachea The trachea is a tubular structure that forms part of the lower respiratory tract. The trachea is continuous superiorly with the larynx and inferiorly becomes the bronchial tree within the lungs. The trachea consists of a support frame of semicircular, or C-shaped, rings made out of hyaline cartilage and reinforced by collagenous connective tissue. Trachea: Anatomy undergoes multiple generations of branching, and in the canalicular stage, primitive alveolar structures and capillaries Capillaries Capillaries are the primary structures in the circulatory system that allow the exchange of gas, nutrients, and other materials between the blood and the extracellular fluid (ECF). Capillaries are the smallest of the blood vessels. Because a capillary diameter is so small, only 1 RBC may pass through at a time. Capillaries: Histology develop. Next, in the saccular stage, gas exchange Gas exchange Human cells are primarily reliant on aerobic metabolism. The respiratory system is involved in pulmonary ventilation and external respiration, while the circulatory system is responsible for transport and internal respiration. Pulmonary ventilation (breathing) represents movement of air into and out of the lungs. External respiration, or gas exchange, is represented by the O2 and CO2 exchange between the lungs and the blood. Gas Exchange becomes possible as the capillaries Capillaries Capillaries are the primary structures in the circulatory system that allow the exchange of gas, nutrients, and other materials between the blood and the extracellular fluid (ECF). Capillaries are the smallest of the blood vessels. Because a capillary diameter is so small, only 1 RBC may pass through at a time. Capillaries: Histology more closely associate with maturing alveoli Alveoli Small polyhedral outpouchings along the walls of the alveolar sacs, alveolar ducts and terminal bronchioles through the walls of which gas exchange between alveolar air and pulmonary capillary blood takes place. Acute Respiratory Distress Syndrome (ARDS), and type II pneumocytes Type II pneumocytes Lungs: Anatomy have started secreting surfactant Surfactant Substances and drugs that lower the surface tension of the mucoid layer lining the pulmonary alveoli. Acute Respiratory Distress Syndrome (ARDS). In the alveolar stage, the alveoli Alveoli Small polyhedral outpouchings along the walls of the alveolar sacs, alveolar ducts and terminal bronchioles through the walls of which gas exchange between alveolar air and pulmonary capillary blood takes place. Acute Respiratory Distress Syndrome (ARDS) continue to grow in number and size and continue to mature until a child is 8 years old.
Last updated: Apr 11, 2023
Lung development occurs in 5 stages:
In utero, the lungs Lungs Lungs are the main organs of the respiratory system. Lungs are paired viscera located in the thoracic cavity and are composed of spongy tissue. The primary function of the lungs is to oxygenate blood and eliminate CO2. Lungs: Anatomy are:
Immediately following delivery:
Mnemonic:
To quickly recall the chronology of the stages of lung development, remember “Every Pulmonologist Can See Alveoli”:
Development of the bronchial tree Bronchial tree The collective term “bronchial tree” refers to the bronchi and all of their subsequent branches. The bronchi are the airways of the lower respiratory tract. At the level of the 3rd or 4th thoracic vertebra, the trachea bifurcates into the left and right main bronchi. Both of these bronchi continue to divide into secondary or lobar bronchi that bifurcate further and further. Bronchial Tree: Anatomy begins in the embryonic stage, with budding Budding Mycology of the embryonic gut tube to form the larynx Larynx The larynx, also commonly called the voice box, is a cylindrical space located in the neck at the level of the C3-C6 vertebrae. The major structures forming the framework of the larynx are the thyroid cartilage, cricoid cartilage, and epiglottis. The larynx serves to produce sound (phonation), conducts air to the trachea, and prevents large molecules from reaching the lungs. Larynx: Anatomy, trachea Trachea The trachea is a tubular structure that forms part of the lower respiratory tract. The trachea is continuous superiorly with the larynx and inferiorly becomes the bronchial tree within the lungs. The trachea consists of a support frame of semicircular, or C-shaped, rings made out of hyaline cartilage and reinforced by collagenous connective tissue. Trachea: Anatomy, and lungs Lungs Lungs are the main organs of the respiratory system. Lungs are paired viscera located in the thoracic cavity and are composed of spongy tissue. The primary function of the lungs is to oxygenate blood and eliminate CO2. Lungs: Anatomy by the end of the stage.
| Tissue layer | Structures formed in the bronchial tree Bronchial tree The collective term “bronchial tree” refers to the bronchi and all of their subsequent branches. The bronchi are the airways of the lower respiratory tract. At the level of the 3rd or 4th thoracic vertebra, the trachea bifurcates into the left and right main bronchi. Both of these bronchi continue to divide into secondary or lobar bronchi that bifurcate further and further. Bronchial Tree: Anatomy | |
|---|---|---|
| Endoderm Endoderm The inner of the three germ layers of an embryo. Gastrulation and Neurulation |
|
|
| Mesoderm Mesoderm The middle germ layer of an embryo derived from three paired mesenchymal aggregates along the neural tube. Gastrulation and Neurulation | Splanchnopleuric layer |
|
| Somatopleuric layer | Parietal pleura Parietal pleura Pleuritis | |
| Ectoderm Ectoderm The outer of the three germ layers of an embryo. Gastrulation and Neurulation | None | |
The bronchial tree Bronchial tree The collective term “bronchial tree” refers to the bronchi and all of their subsequent branches. The bronchi are the airways of the lower respiratory tract. At the level of the 3rd or 4th thoracic vertebra, the trachea bifurcates into the left and right main bronchi. Both of these bronchi continue to divide into secondary or lobar bronchi that bifurcate further and further. Bronchial Tree: Anatomy develops off of the foregut Foregut Development of the Abdominal Organs of the embryonic gut tube.
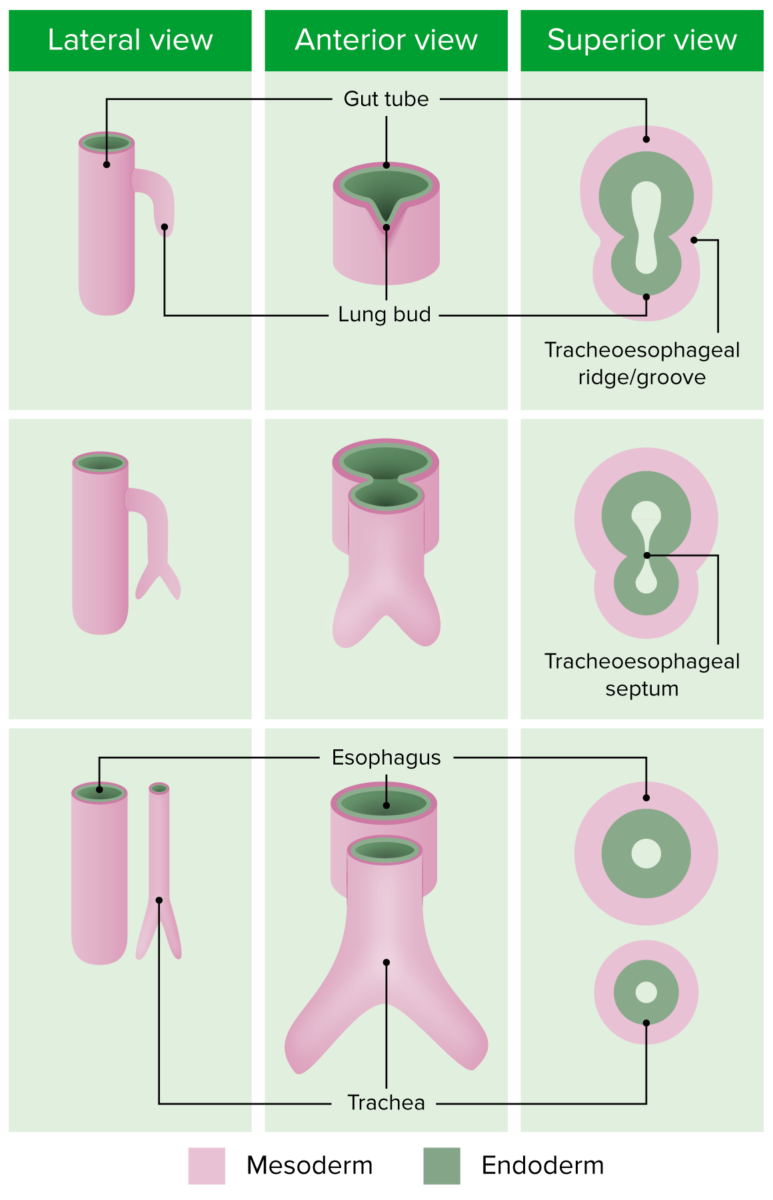
Embryonic development of the bronchial tree
Image by Lecturio.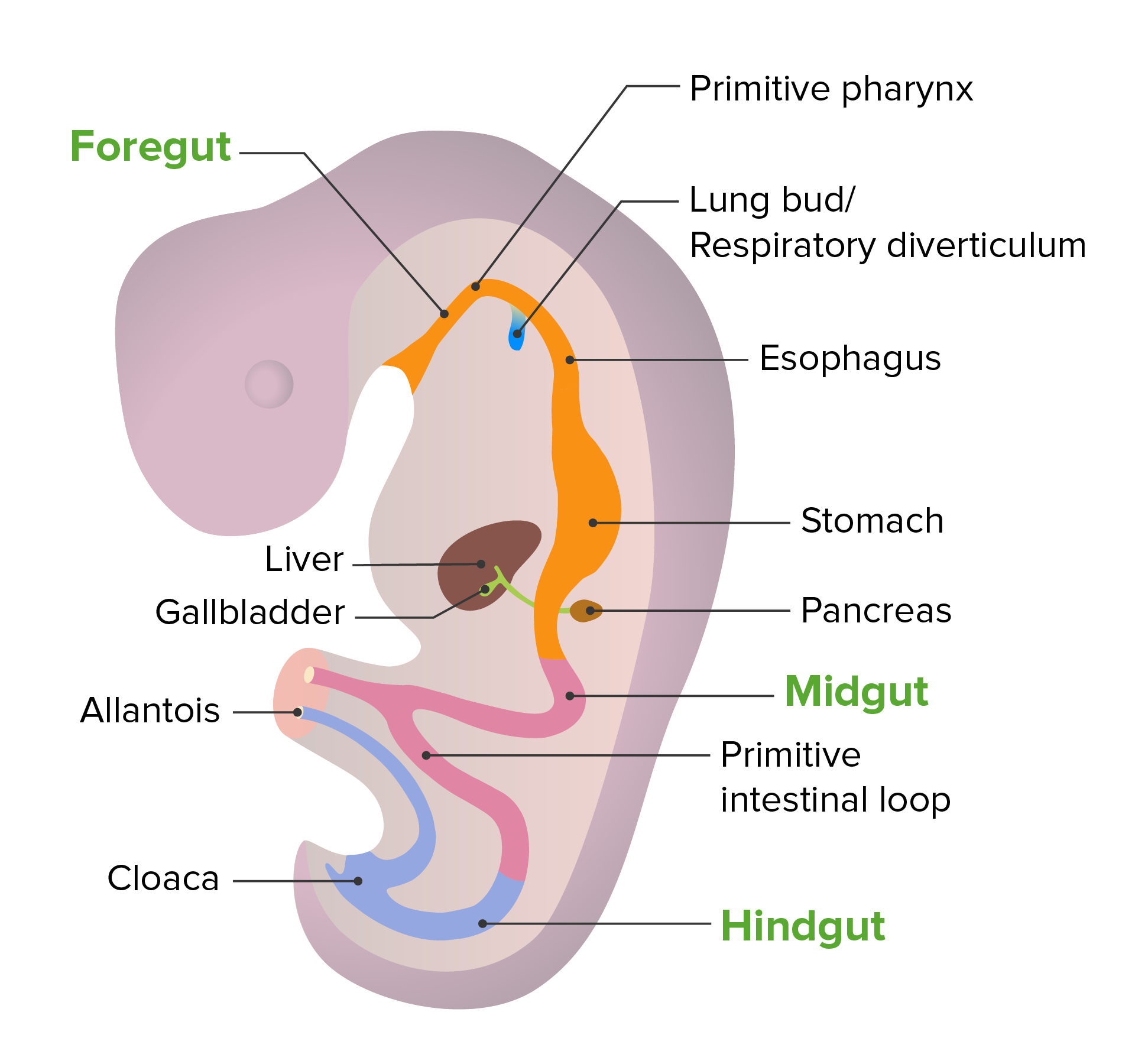
Embryonic development of the gut tube
Image by Lecturio. License: CC BY-NC-SA 4.0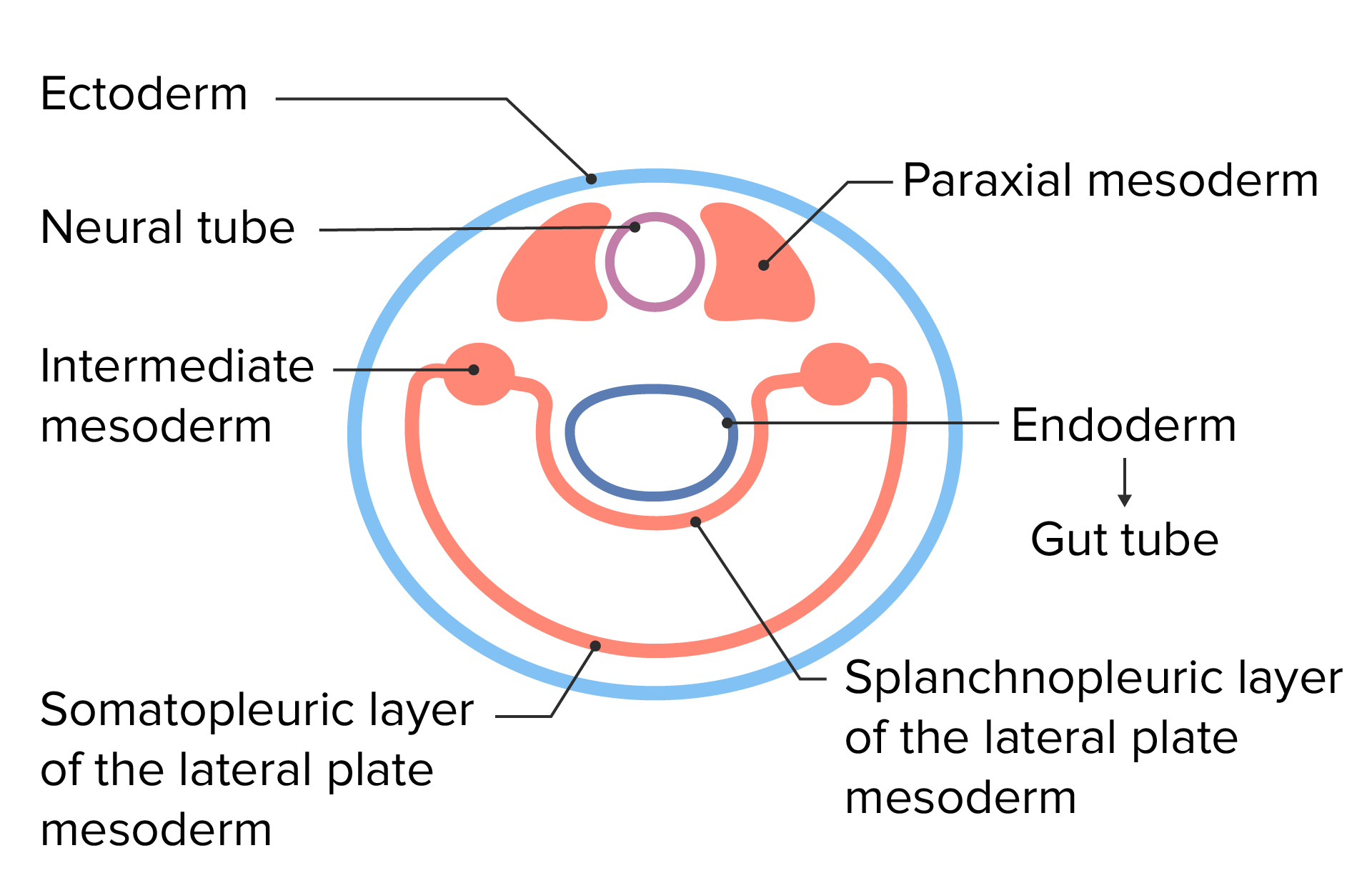
Embryonic cell layers following lateral folding of the trilaminar disc
Image by Lecturio.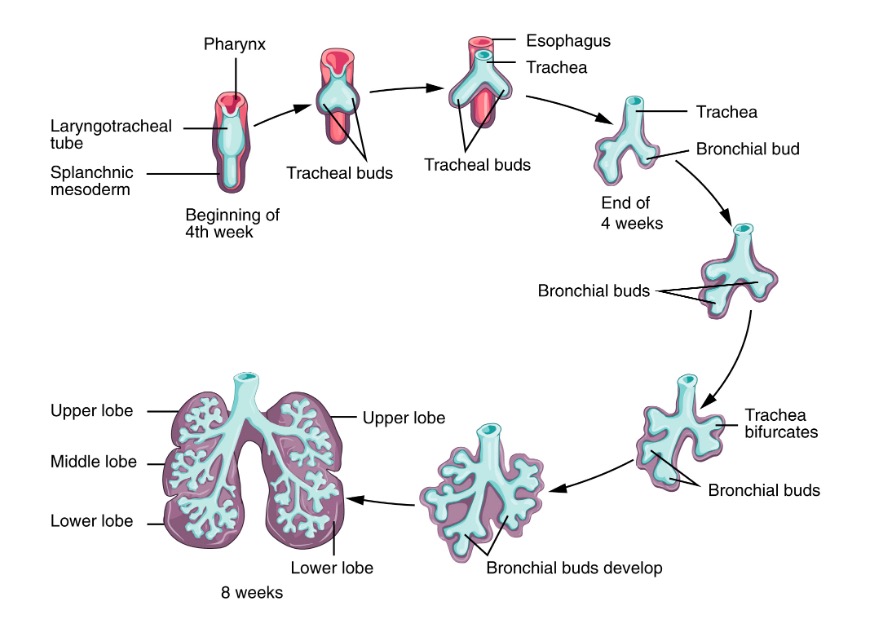
Early stage of lung development:
During the early stages of lung development (embryonic and pseudoglandular), the lung bud gradually transforms into the trachea and then bifurcates, forming the two mainstem bronchi. These bronchi will further divide, giving rise to lobar and segmental bronchi.
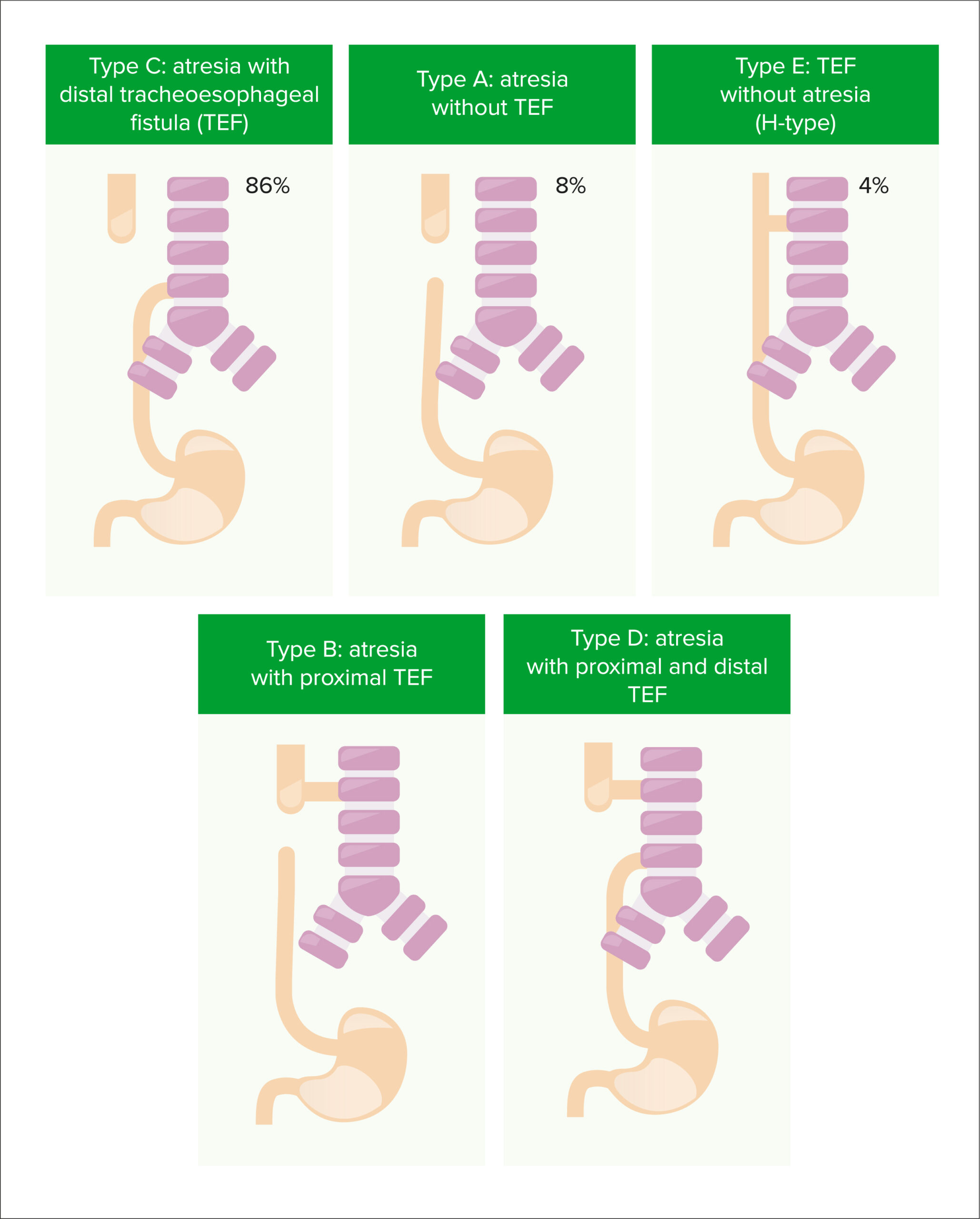
Types of tracheoesophageal fistula and esophageal atresia
Image by Lecturio.This stage generates most of the bronchial tree Bronchial tree The collective term “bronchial tree” refers to the bronchi and all of their subsequent branches. The bronchi are the airways of the lower respiratory tract. At the level of the 3rd or 4th thoracic vertebra, the trachea bifurcates into the left and right main bronchi. Both of these bronchi continue to divide into secondary or lobar bronchi that bifurcate further and further. Bronchial Tree: Anatomy. It gets its name because histologically, the bronchi Bronchi The larger air passages of the lungs arising from the terminal bifurcation of the trachea. They include the largest two primary bronchi which branch out into secondary bronchi, and tertiary bronchi which extend into bronchioles and pulmonary alveoli. Bronchial Tree: Anatomy (which are lined with cuboidal cells at this stage) resemble glands as they branch into the surrounding mesoderm Mesoderm The middle germ layer of an embryo derived from three paired mesenchymal aggregates along the neural tube. Gastrulation and Neurulation.
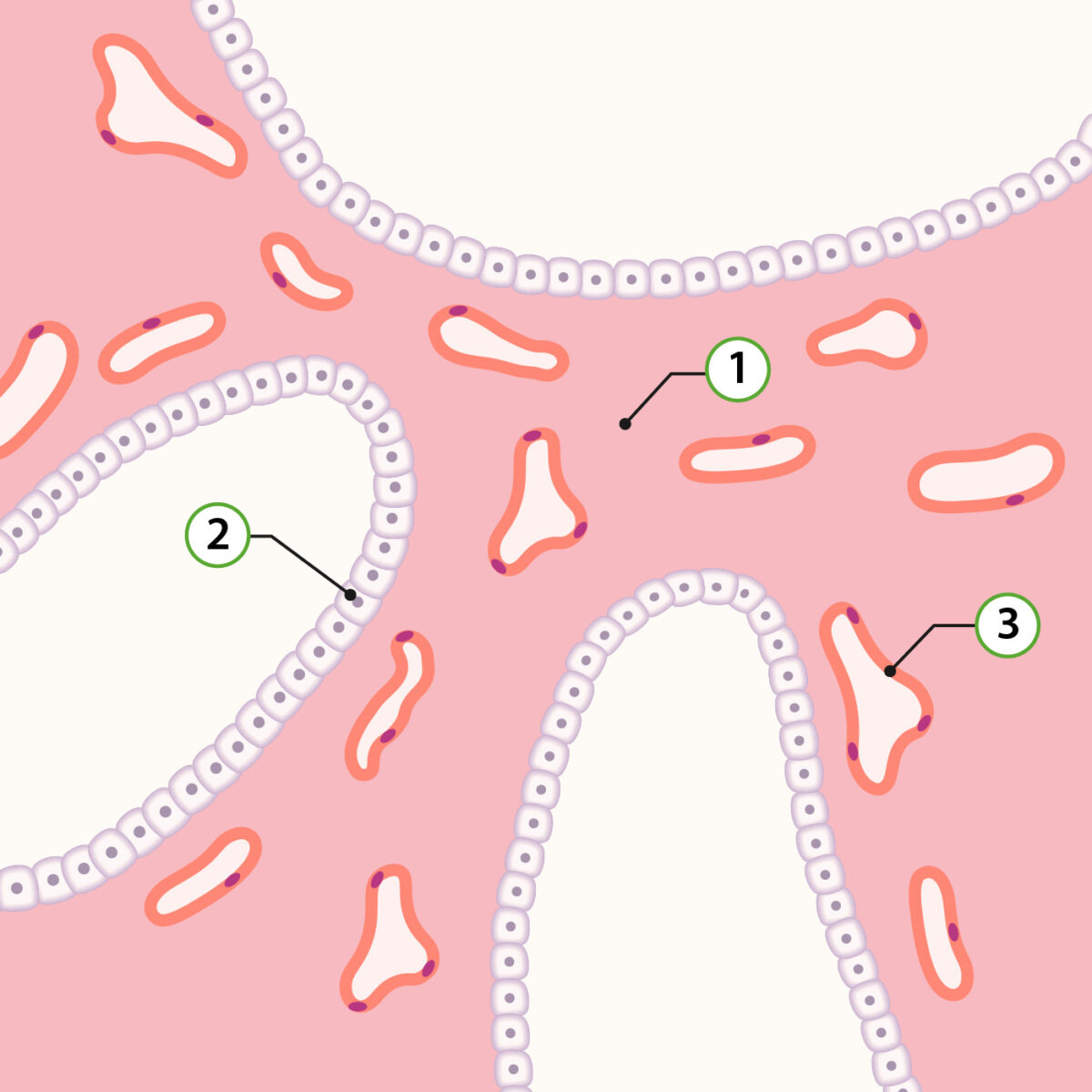
Representation of pulmonary histology in the pseudoglandular stage:
1: Lung mesenchyme
2: Type II pneumocyte precursors
3: Capillaries
During this stage, the respiratory units (also referred to as canaliculi) develop at the end of the terminal bronchioles Bronchioles The small airways branching off the tertiary bronchi. Terminal bronchioles lead into several orders of respiratory bronchioles which in turn lead into alveolar ducts and then into pulmonary alveoli. Bronchial Tree: Anatomy.
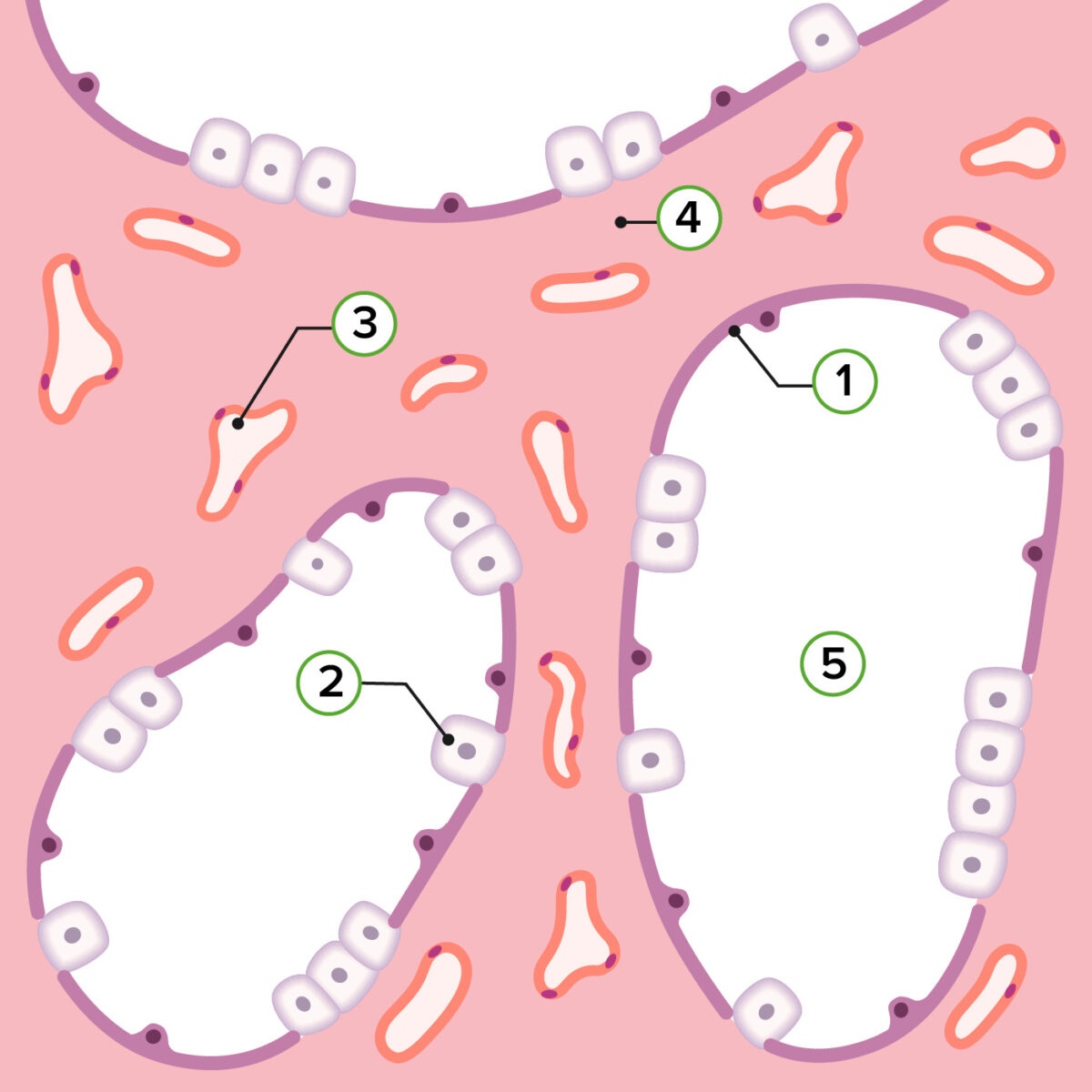
Representation of pulmonary histology in the canalicular stage:
1: Type I pneumocyte
2: Type II pneumocyte
3: Capillaries
4: Lung mesenchyme
5: Alveolar duct
Canalicular stage of lung development:
By the end of 6 months, type II pneumocytes begin secreting surfactant. This oily cell product spreads across airways, decreasing surface tension and allowing for smooth lung expansion.
During this stage, the alveoli Alveoli Small polyhedral outpouchings along the walls of the alveolar sacs, alveolar ducts and terminal bronchioles through the walls of which gas exchange between alveolar air and pulmonary capillary blood takes place. Acute Respiratory Distress Syndrome (ARDS) begin to mature, as type II pneumocytes Type II pneumocytes Lungs: Anatomy (cuboidal cells) flatten into type I pneumocytes Type I pneumocytes Lungs: Anatomy capable of gas exchange Gas exchange Human cells are primarily reliant on aerobic metabolism. The respiratory system is involved in pulmonary ventilation and external respiration, while the circulatory system is responsible for transport and internal respiration. Pulmonary ventilation (breathing) represents movement of air into and out of the lungs. External respiration, or gas exchange, is represented by the O2 and CO2 exchange between the lungs and the blood. Gas Exchange, creating the gas-exchange surface area. Surfactant Surfactant Substances and drugs that lower the surface tension of the mucoid layer lining the pulmonary alveoli. Acute Respiratory Distress Syndrome (ARDS) production increases significantly.
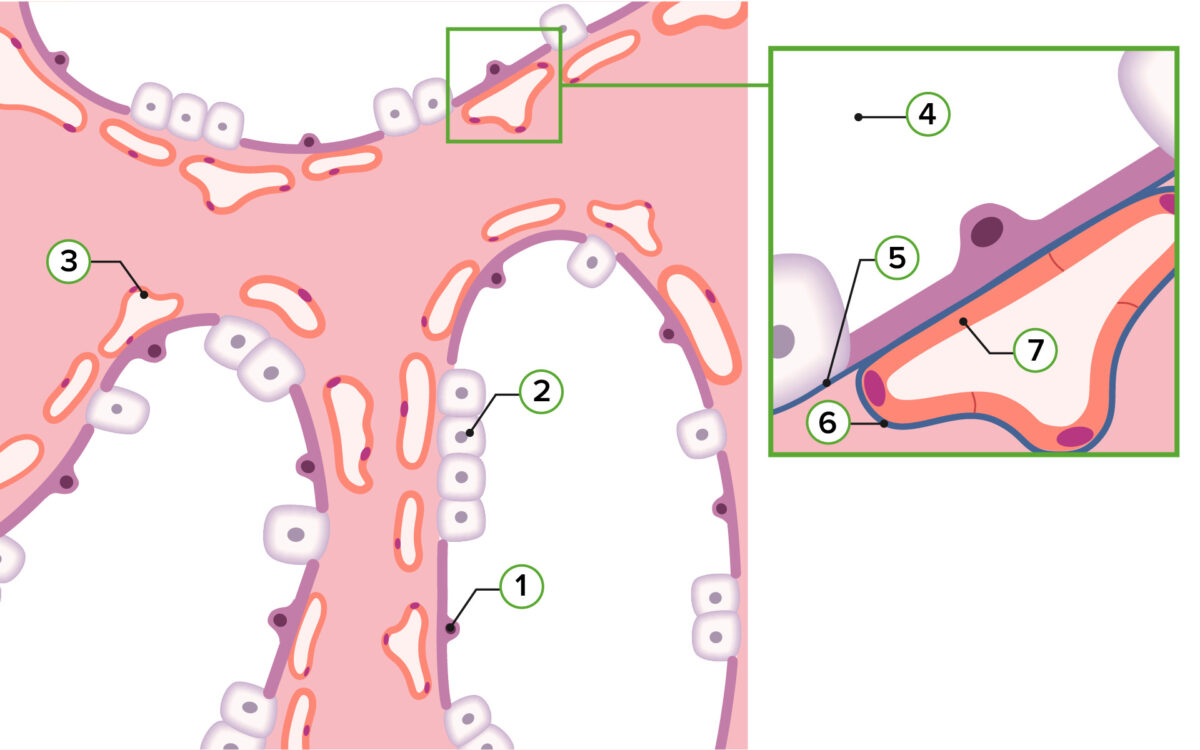
Representation of pulmonary histology in the saccular stage:
1: Type I pneumocyte
2: Type II pneumocyte
3: Capillaries
4: Saccular space
5: Basal membrane of the air passage
6: Basal membrane of the capillaries
7: Endothelium of the capillaries
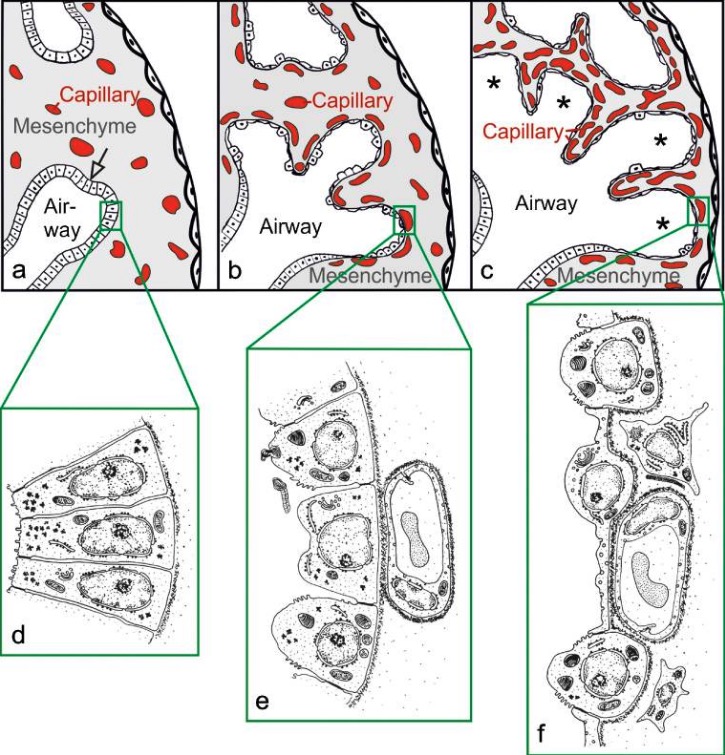
Morphologic development of the lung parenchyma during the pseudoglandular (a, d), canalicular (b, e) and saccular (c, f) stages:
The epithelial tubules branch repeatedly during the pseudoglandular stage and penetrate into the surrounding mesenchyme (a, arrow, branching point).
The mesenchyme contains a loose capillary network (a). The epithelium itself is tall and columnar (d). The canalicular stage (b) is characterized by (1) differentiation of the epithelial cells into type I and type II epithelial cells (e, f), (2) widening of the future airways (b), (3) multiplication of the capillaries and their first close contacts to the epithelium (b), and (4) formation of first future air–blood barriers (e→f).
During the saccular stage (c), thick immature inter-airspace septa are formed by further condensation of the mesenchyme. The immature septa contain a double-layered capillary network, 1 layer on either side of the septum. The terminal ends of the bronchial tree represent wide spaces and are called saccules (asterisks).
During the alveolar stage, the respiratory units continue to grow in number and maturity.
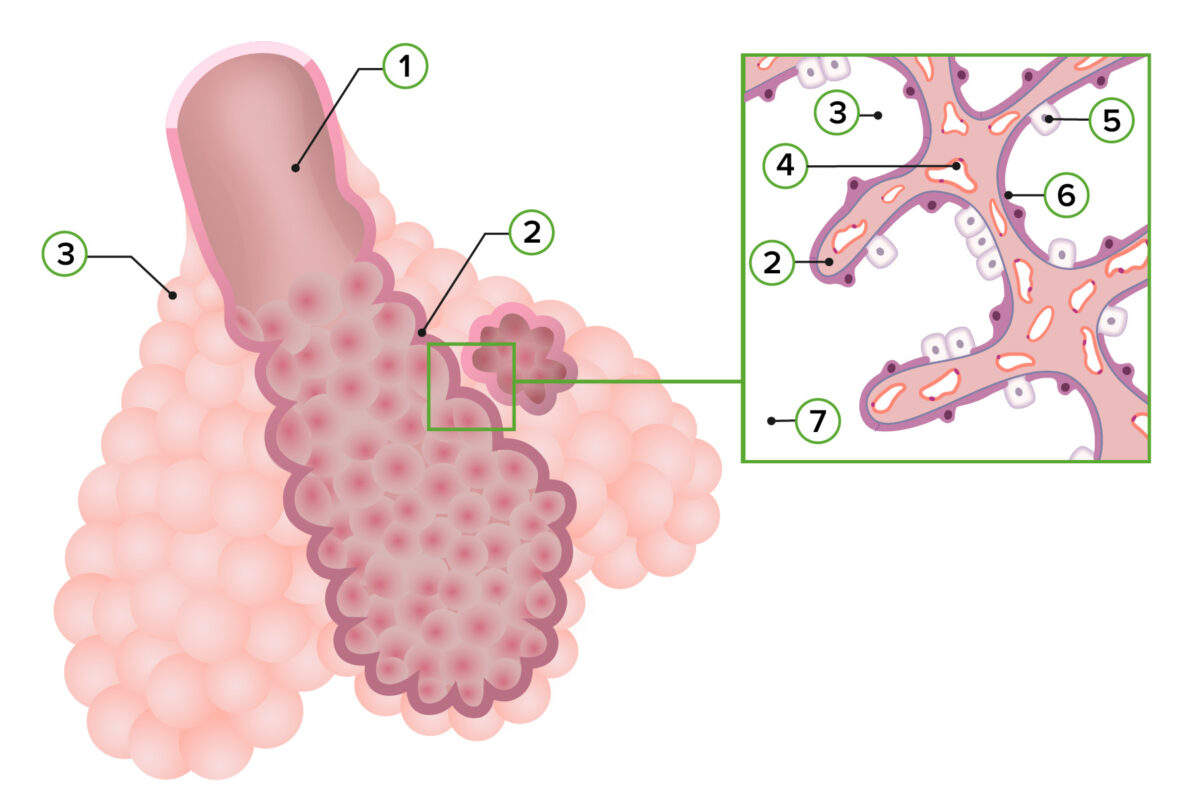
Representation of pulmonary histology in the alveolar stage:
1: Respiratory bronchiole
2: Primary septum
3: Alveolar sac
4: Capillaries
5: Type II pneumocyte
6: Type I pneumocyte
7: Alveolar duct
| Developmental stage | Description | Clinical relevance |
|---|---|---|
| Embryonic period (weeks 4–7) |
|
|
| Pseudoglandular period (weeks 5–16) |
|
|
| Canalicular period (weeks 16–26) |
|
|
| Saccular period (weeks 26–birth) |
|
|
| Alveolar period (week 32–8 years) |
|
|