Epidemiological studies epidemiological studies Epidemiological studies are used to establish associations between risk factors and health-related outcomes. These studies can take the form of observational or interventional studies. Epidemiological Studies are designed to evaluate a hypothesized relationship between an exposure and an outcome; however, the existence and/or magnitude of these relationships may be erroneously affected by the design and execution of the study itself or by conscious or unconscious Unconscious Those forces and content of the mind which are not ordinarily available to conscious awareness or to immediate recall. Psychotherapy errors perpetrated by the investigators or the subjects. These systematic errors are called biases. If not avoided or accounted for, biases can completely invalidate the results of an otherwise well-thought-out study.
Last updated: Dec 15, 2025
Bias is an error Error Refers to any act of commission (doing something wrong) or omission (failing to do something right) that exposes patients to potentially hazardous situations. Disclosure of Information in the design, conduct, or analysis of a study that results in a deviation of the test statistic from its true value. This results in an incorrect estimate of an association between an exposure and the target population such that the test statistic generated deviates from its true value.
| Masking | Hides an existing relationship between the independent and a dependent variable Dependent variable Types of Variables |
|---|---|
| False relationships | Creates spurious relationships between the independent and a dependent variable Dependent variable Types of Variables |
| Overestimation | Exaggerates the size of an existing relationship between the independent and a dependent variable Dependent variable Types of Variables |
| Underestimation | Diminishes the size of an existing relationship between the independent and a dependent variable Dependent variable Types of Variables |
Selection Selection Lymphocyte activation by a specific antigen thus triggering clonal expansion of lymphocytes already capable of mounting an immune response to the antigen. B cells: Types and Functions bias is the error Error Refers to any act of commission (doing something wrong) or omission (failing to do something right) that exposes patients to potentially hazardous situations. Disclosure of Information introduced when the study population does not represent the target population due to some selection Selection Lymphocyte activation by a specific antigen thus triggering clonal expansion of lymphocytes already capable of mounting an immune response to the antigen. B cells: Types and Functions preference.
Information bias results from systematic errors in the measurement of some exposure, outcome, or variable Variable Variables represent information about something that can change. The design of the measurement scales, or of the methods for obtaining information, will determine the data gathered and the characteristics of that data. As a result, a variable can be qualitative or quantitative, and may be further classified into subgroups. Types of Variables. The major types of information bias are misclassification bias, recall bias, interviewer bias, response bias, reporting bias, observer bias, ascertainment bias, and confirmation bias.
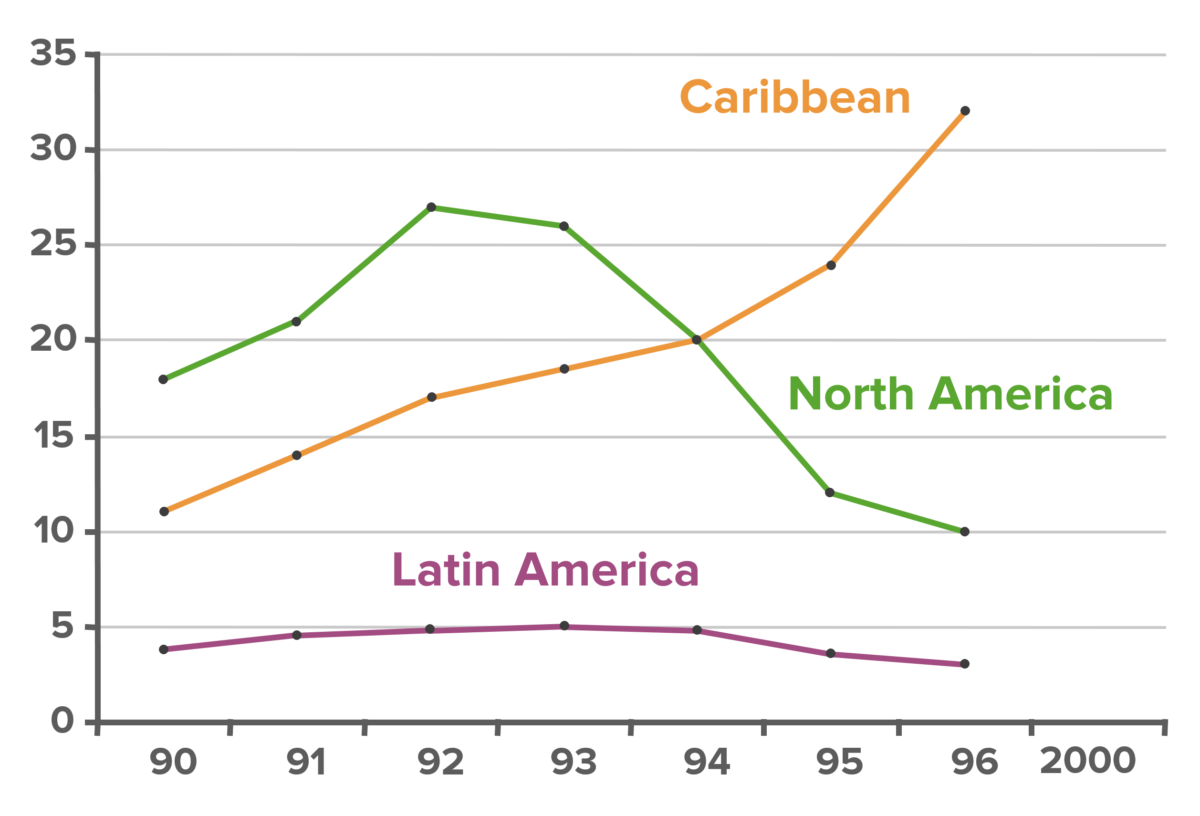
Example of detection bias. Shown above is a graph of the number of new AIDS cases per year per 100,000 population over the time period from 1990–2000. Although the chart seems to suggest that cases of AIDS in the Caribbean are on the rise (orange line), in reality, the steady increase in new cases is due to the increase in screening number and frequency. This is an example of detection bias, a subtype of information bias.
Image by Lecturio.Definition
A confound is an additional variable Variable Variables represent information about something that can change. The design of the measurement scales, or of the methods for obtaining information, will determine the data gathered and the characteristics of that data. As a result, a variable can be qualitative or quantitative, and may be further classified into subgroups. Types of Variables other than the independent variable Independent variable Types of Variables that has an effect on the dependent variable Dependent variable Types of Variables, causing an erroneous relationship to be inferred between them.
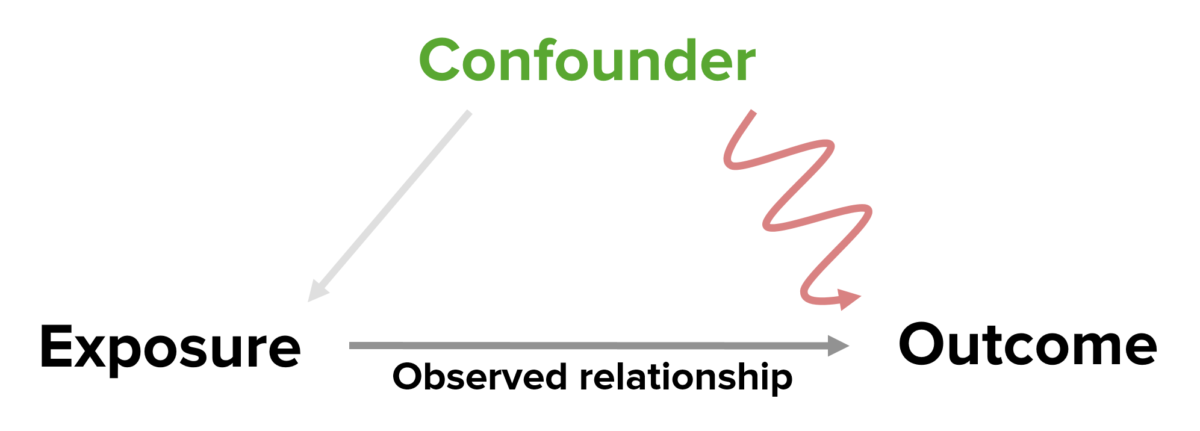
A diagram detailing how a confounding variable is related to the exposure and the outcome. The confound is related to the exposure and may contribute to or cause the outcome; if not accounted for, it can contribute to causing or affecting the magnitude of the observed (true) relationship.
Image by Lecturio.| Confounders | Effect modifiers | |
|---|---|---|
| Masks associations | Yes | No |
| Creates the illusion Illusion The misinterpretation of a real external, sensory experience. Schizophrenia of association | Yes | No |
| Modifies the nature of the relationship | No | Yes |