Playlist
Show Playlist
Hide Playlist
Myeloproliferative Disease – Non-malignant conditions and Multiple Myeloma
-
Slides Other haemopoietic malignancies and aplastic anaemia.pdf
-
Reference List Hematology.pdf
-
Download Lecture Overview
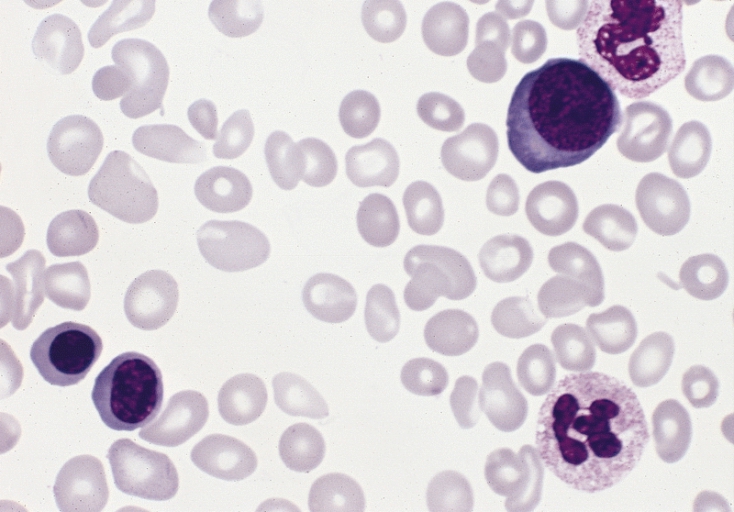
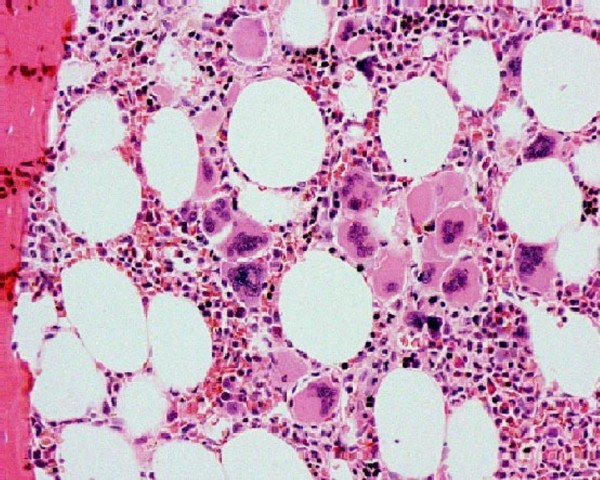
00:01 Now, let's move on to the second major category of additional malignant disease of the hemopoietic system, myeloproliferative disease. 00:13 These are a group of conditions arising from marrow stem cells, and the name suggests and tells us that these are highly proliferative disorders, where we see proliferation of one or indeed more of the hemopoietic components in the bone marrow. 00:30 So, rather than myelodysplasia having low and dysfunctional cells within the blood, here, we often see increased cells within the peripheral blood. 00:40 There are three major types, polycythemia vera or PV, associated with an increase in the red cell count, essential thrombocythemia, ET, with a high platelet count, and primary myelofibrosis, where there is increased fibrotic activity within the bone marrow. 01:02 Bone marrow scarring, really, with anemia, and often very large spleen. 01:08 And that brings me to the slide on the right where you can see a very enlarged spleen in this patient. 01:16 And splenomegaly is a very common feature of all of these disorders and gave us one clinical clue that these disorders should indeed be grouped together. 01:28 But genetic analysis over the last three years has been very exciting, and now indicates that we got it right by putting these diseases together, as they often have a similar genetic basis. Let's look at this story in a little more detail. 01:46 JAK2 is a protein that's involved in signal transduction within hemopoietic cells, and it mediates the response of myeloid cells to growth factors and cytokines stimulation. 02:00 Now, the JAK2 gene is mutated in almost all cases of polycythemia vera, and in many cases of essential thrombocythemia and primary myelofibrosis, and that mutation leads to high activity of the JAK2 gene. 02:19 The tumor cell believes itself to be chronically stimulated by growth factors or cytokines. 02:27 Calreticulin mutations are usually found in patients with ET, essential thrombocythemia, that don't have a JAK2 mutation. 02:38 And finally, MPL or mipple, which is the receptor for thrombopoietin, is also mutated in a minority, five to 10%, of patients with ET or myelofibrosis. 02:52 And I think you can see that if we put these three genes together, JAK2, calreticulin, or MPL, we can now explain 99% of cases of polycythemia vera, and up to 90% of cases of essential thrombocythemia and myelofibrosis. 03:13 So, a very nice story for common genetic basis, many of these disorders. 03:19 Now, as we'll talk about later, this is also useful clinically, because drugs that inhibit JAK2 activity are now finding a very important place in the treatment of these disorders. 03:33 Let's consider them individually. Polycythemia vera, polycythemia is an increase in the hemoglobin concentration above the upper limit for a person's age and gender. 03:47 And this increased hemoglobin leads to hyperviscosity. 03:52 Headaches. Pruritis itching, that's a really common feature, particularly after showers. 04:00 The patient has a plethoric appearance. Just look at the top right-hand slide. 04:06 Patient is very red. They may also have an enlarged spleen, as you saw in the previous slides. 04:13 Now, the treatment of polycythemia is to reduce the hemoglobin and the danger that arises from that. 04:21 Ideally, we want to keep the hematocrit ratio of the red blood cells within the blood volume to less than 0.45, and we want to keep the platelet count down as well to stop the risk of blood clots. 04:36 The easiest way to do this is to just take blood from the patient, venesection. 04:43 We can also use drugs such as hydroxycarbamide, which dampens down DNA synthesis within the bone marrow. 04:53 And finally, JAK2 inhibitors, as you will not be surprised to hear, are now being introduced in this disorder to try and address the fundamental problem that's present within the malignant cells. 05:05 If you're wondering what the slide on the bottom right indicates, that shows somebody with gout, and that can be seen in patients with myeloproliferative disease, because they are producing and destroying too many hemopoietic cells all the time, and the excess DNA destruction can raise the level of uric acid and precipitate those episodes. 05:30 The second major disorder within myeloproliferation is essential thrombocythemia. 05:35 Too many platelets. The main clinical problem here is a blood clot, thrombosis, but actually, bleeding as well, which is paradoxical that a patient with a very high platelet count is at risk of bleeding. 05:52 On the right-hand side, you'll see a blood film. 05:56 And you'll see, typically, lots of red cells, there's one neutrophil there, and too many of those small platelets throughout that blood film. 06:07 If you look carefully, one or two very large platelet forms that we often see in this disorder. 06:14 Now, here, we don't necessarily need to rush into treatment to get the platelet count down to a normal level, but we need to assess the risk within individuals of developing thrombotic disease. 06:29 That will depend on the patient's age, whether they have a history of heart disease, whether they are taking drugs such as estrogens, which might raise their thrombotic risk. 06:42 And after assessment of their risk group, we can use drugs such as Hydroxyurea, which dampen down blood cell production, Aspirin, which reduces the function of these excess platelets, or perhaps interferon, which is a natural product within our bodies, and it's actually quite useful at treating myeloproliferative disorders. 07:06 Finally, the third major subtype of myeloproliferation, primary myelofibrosis. 07:14 And in this condition, there is proliferation of a cell within the bone marrow, which leads to fibrotic scarring of hemopoiesis. 07:25 And what this means is that blood production struggles within the bone marrow and tries to find another place for hemopoiesis to be established. 07:35 And that's often in the spleen and the liver. 07:37 Patient can develop a massive spleen which enlarges even down into the left iliac -- right iliac fossa. 07:45 The clinical problems here are of anemia and this very large spleen. 07:51 The typical blood film in primary myelofibrosis comes under that word leucoerythroblastic. 08:00 What does that mean? Just think of that word, leuco, white, erythro, red, blastic, you see the primitive precursors of both white cells and red cells. 08:12 And you can see in that film there, the blood film, some of these features. 08:18 And the bottom cell is a nucleated red cell. 08:20 And you will also see teardrop red cells. 08:23 Just look at some of those red cells in the middle of the picture, and you'll see the teardrop features that you see in this condition. 08:31 How do we treat this? Well, we can get blood transfusions to support the patient, but this is a disorder in which JAK2 inhibitors are really finding a very important role in therapy. 08:44 Stem cell transplantation can be curative Now, let's move on to a different type of malignant disease and a very important one, multiple myeloma. 08:56 This is a malignant disease of plasma cells. 09:00 Now, plasma cells live within our bone marrow. 09:03 There's very, very few of them in the blood and they produce antibodies. 09:07 And the tumor retains these properties. 09:10 It's present within the bone marrow, hence the name, myelo, bone marrow. 09:17 And again, the cells produce antibodies. 09:20 You get a range of clinical problems in myeloma, and I've listed some of them on the left. 09:28 Increased calcium because of bone destruction. 09:31 Renal damage because of the antibodies, which may precipitate out in the kidney. 09:38 Anemia, because the plasma cells crowd out the normal hemopoiesis. 09:44 And bone disease, because of the expansion of the malignant cells within the bone, damaging normal ossification and bone formation. 09:55 Let's just look at some of the pictures on the right to illustrate this. 10:00 The top-right blood film, I don't know if you can see anything obviously different in that, but if you look carefully, you see a phenomenon called rouleaux. 10:09 That's when red cells stack one on top of the other. 10:12 And you can see some of the red cells lining up. 10:16 That's due to the paraprotein, the high levels of antibodies in the blood. 10:21 And in the bottom, you see some x-rays. 10:25 And what I hope you can see is the bone damage from the myeloma. 10:29 Look at the skull x-ray and you'll see that dark patch in the middle. 10:33 That's an area of multiple myeloma. 10:36 Whereas if you look on the right at the long bones, you will see small areas of dark damage within the long bones, and that's an area of myeloma as well. 10:49 Now, you can see why patients are so prone to fractures of the bone in this disease. 10:57 Now, the diagnosis of multiple myeloma depends on finding a monoclonal antibody in the blood or urine. 11:06 This monoclonal antibody or immunoglobin is called a paraprotein. 11:10 On the top right, you'll see an electrophoresis that is used to detect this sort of paraprotein. 11:18 On the right, you can see a paraprotein right at the bottom of the screen in number three. 11:24 Now, very often, because the bone marrow is producing one paraprotein, all the normal antibodies are reduced. 11:32 And that, of course, leads patient very prone to infection. 11:37 It's a characteristic feature of multiple myeloma. 11:41 Of course, if we do a bone marrow biopsy, we see the tumor cells and we see an excess of plasma cells. 11:48 Look at the bottom right and you'll see these plasma cells. 11:53 Very large cytoplasm and those dark nucleus. 11:58 Some seems a little bit like a fried egg. 12:02 You can see on the left, that big cell with a white -- gray cytoplasm, and the dark purple nucleus. Those are all plasma cells. 12:11 If we do radiology imaging, either by x-rays or perhaps even a CT or MRI scan, we may see damaged bones, and that would indicate to us that we need to treat this disease quickly. 12:25 However, let me just finish off this slide with an important point. 12:29 Low levels of these monoclonal immunoglobulins or paraproteins are often seen in people as they get older. 12:39 And if you find a low level of paraprotein without any evidence of clinical damage, then that's just called monoclonal gammopathy of undetermined significance. 12:52 It's not the most pretty name or the shortest name, but what it means, it's stated clearly in the name, we have an abnormal paraprotein, but we don't know how significant it is. You do not need to treat that. 13:06 However, around one in every hundred of those patients will go on to develop myeloma every year that you follow them up. 13:16 When myeloma does develop and we have clinical problems that we must treat, how do we go about treating it? This remains a very difficult condition to cure, but treatment has improved a lot in recent years, and the median survival now can be up to seven to 10 years. 13:36 Now, younger patients will have chemotherapy, and I'll talk to you about the sorts of drugs we use in a minute, followed by an autologous stem cell transplant. 13:47 What do I mean by that? Well, in that procedure, you take blood from the patient, which contains stem cells, and you can freeze it down. 13:57 Then, that allows you to give the patient a high dose of chemotherapy. 14:01 And then when their blood is trying to recover, you can put in the stem cells that you previously frozen to support the regrowth of the blood compartment. 14:13 So, it allows a very intense chemotherapy to be given, and that being shown to improve survival. 14:20 Older patients can't really tolerate that approach, and so they tend to have chemotherapy alone. 14:27 Sometimes after the chemotherapy, you may wish to give maintenance treatment with a drug such as lenalidomide, and we'll talk about that in a second. 14:36 Let's talk about some of the drugs that are used in myeloma. 14:40 There's a wide range of very interesting agents. 14:43 The top there, I've put a drug called Velcade or bortezomib. 14:47 It's a drug that inhibits the proteasome. 14:52 That's part of our normal cell that breaks down proteins within the cell. 14:57 Very effective agent. 15:00 Secondly, I've grouped together lenalidomide, thalidomide, and pomalidomide. 15:05 Thalidomide, I'm sure you've heard of. 15:08 These drugs have a similar mechanism of action, and myeloma is one of the most effective areas for their use. 15:16 And they're used in many, many drug regimens. 15:19 Dexamethasone, steroids, often used in myeloma. 15:24 And Melphalan is a classic alkylating agent that's still used in some patients with myeloma and is often used for preparing people for an autologous stem cell transplant. 15:40 For patients with multiple myeloma in their third or greater relapse, combination with the anti-CD38 monoclonal antibody daratumumab is used in the US in conjunction with pomalidomide and dexamethasone For patients with further refractory disease, therapies that target the B cell maturation antigen (BCMA) protein on plasma cells can be used. 16:10 The BCMA can be targeted using chimeric antigen receptor cells called CAR-T cells, which can be highly effective for advanced disease.
About the Lecture
The lecture Myeloproliferative Disease – Non-malignant conditions and Multiple Myeloma by Paul Moss, PhD, OBE, FMed, FRCPath is from the course Hematologic Disorders. It contains the following chapters:
- Myeloproliferative Disease
- Polycythemia Vera
- Essential Thrombocythemia
- Primary Myelofibrosis
- Multiple Myeloma
Included Quiz Questions
Which of the following is NOT considered a myeloproliferative disorder?
- Lymphoma
- Polycythemia vera
- Essential thrombocythemia
- Primary myelofibrosis
Which of the following genes is most commonly found to be mutated in myeloproliferative disorders?
- JAK2
- CALR
- MPL
- NF1
- MEN1
What role does the JAK2 protein play within hemopoietic cells?
- Signal transduction
- Signal initiation
- Signal transcription
- Signal elimination
- Signal re-entry
In patients with essential thrombocytosis who do not have a JAK2 mutation, what gene is most commonly mutated?
- Calreticulin
- BRCA
- NF1
- MEN1
- CFTR
Which of the following is NOT a common sign or symptom of polycythemia vera?
- Fever
- Headaches
- Enlarged spleen
- Pruritus
- Plethoric faces
Which of the following is NOT part of the treatment of polycythemia vera?
- Sirolimus
- Venesection
- JAK2 inhibitors
- Hydroxycarbamide
Which of the following is part of the treatment protocol for essential thrombocytosis?
- Interferon
- Cyclophosphamide
- Methotrexate
- Vincristine
- Vinblastine
What is the pathology causing illness in primary myelofibrosis?
- Fibrosis of the bone marrow
- Necrosis of the bone marrow
- Production of red blood cells in the liver
- Enlargement of the spleen
- Decreased red blood cell lifespan
Which of the following finding is NOT suggestive of the diagnosis of multiple myeloma?
- Hypocalcemia
- Decreased renal function
- Clonal proliferation of plasma cells
- Anemia
- Lytic bone lesions
Which of the following findings, commonly seen in the peripheral blood film of patients with multiple myeloma, is due to increased antibody levels in the blood?
- Rouleux formation
- Anemia
- Proliferation of plasma cells
- Tear drop cells
- Leukoerythroblastic blood picture
Which of the following techniques can be used to detect paraproteins in the blood of a patient with multiple myeloma?
- Serum electrophoresis
- ELISA
- Western blot
- Peripheral blood smear
- Nephlometry
Which of the following agents is NOT usually used to treat patients with multiple myeloma?
- Docetaxel
- Melphalan
- Lenalidomide
- Bortezomib
- Dexamethasone
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |