Playlist
Show Playlist
Hide Playlist
Hyponatremia: Clinical Approach
-
Slides Water Balance Hypo and Hypernatremia.pdf
-
Reference List Nephrology.pdf
-
Download Lecture Overview
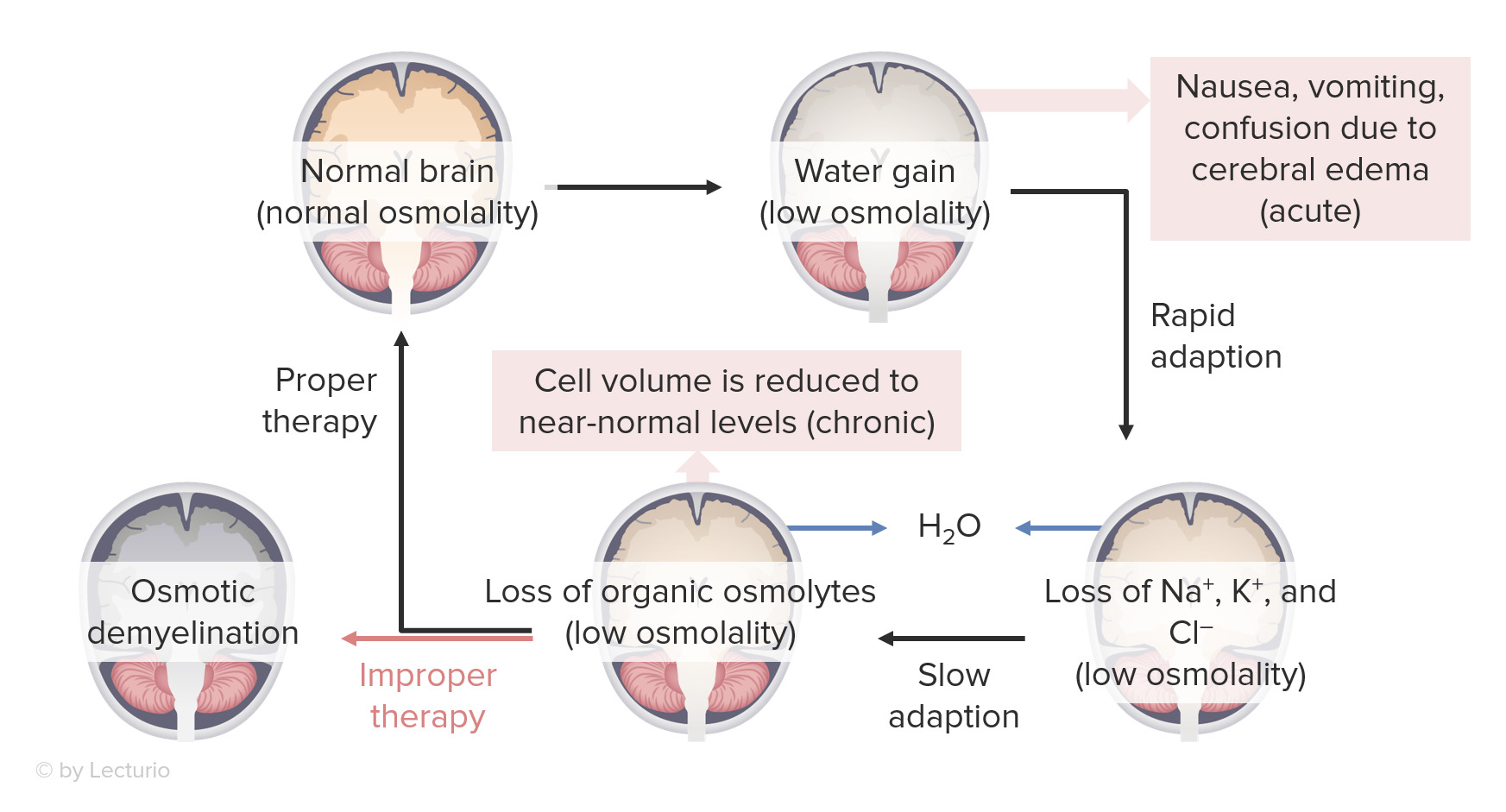
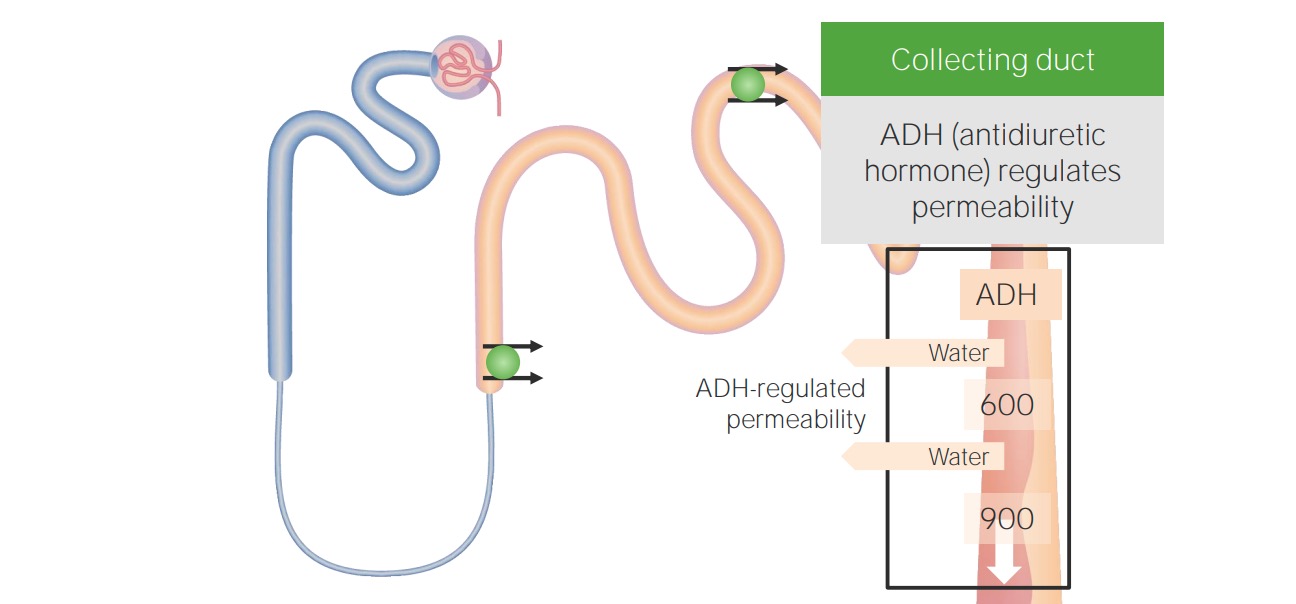
00:01 So, there's two important determinants in the workup of hyponatremia. 00:05 Number one, when our patients present with hyponatremia, we wanna determine their plasma tonicity. 00:12 Again, this determines the transcellular distribution of water. 00:15 The next thing we wanna do is we wanna determine our patient's volume status. 00:20 And remember what volume status correlates to. 00:23 That's the total body sodium. 00:26 So, the first thing we wanna do, remember, is to determine the plasma tonicity. 00:31 So, if we have a patient who's sitting before us, and they have hyponatremia, and we find that their plasma osmolality is greater than 290 mOsmol/kg, they have what we call hypertonic hyponatremia. 00:45 And that results due to the presence of another effective osmole that causes free water to move from the intracellular fluid compartment to the extracellular fluid compartment. 00:55 So, examples include mannitol, which is an osmotic diuretic, glycine, which is an irrigant that we use for procedures of the prostate or hysteroscopies, and finally, marked hyperglycemia. 01:08 So, for example, if I have a patient who comes in with significant hyperglycemia, remember, glucose is an effective osmole. 01:17 It cannot move freely to that intracellular compartment, therefore it exerts a tonic effect, and water will move now from my intracellular compartment to my extracellular fluid compartment. 01:29 I don't have a total body water increase, I'm simply translocating where water is moving from. 01:36 The treatment is to correct the underlying condition in the situation, so I wanna either remove the offending agent if I have a patient who perhaps has mannitol or glycine, or if a patient's hyperglycemic, I wanna correct the hyperglycemia with insulin. 01:54 A second category that we have to think about are patients who present hyponatremically, but yet, their plasma osmolality is within the normal range. 02:03 That means that that plasma osmolality is somewhere between 275-290 mOsmol/kg. 02:09 We refer to this as isotonic hyponatremia, and almost always, this is due to pseudohyponatremia, which is a laboratory artifact. 02:18 That means that that patient usually has a marked increase in serum lipids or proteins, and that's going to result in a reduction of the fraction of serum that's water, and then artificially low serum sodium. 02:31 Now, the plasma water sodium concentration and osmolality are actually unchanged, but the measured sodium concentration and the total plasma volume will be reduced, given that the specimen contains less plasma water. 02:44 This only occurs when patient -- when we're using flame photometry or indirect potentiometry. 02:50 When we use things like direct potentiometry or ion-specific electrodes, this avoids this misdiagnosis, so I think it's very important when you have a patient who presents before with pseudohyponatremia, you should call the lab to find out exactly what they're using. 03:05 Our third situation is a patient who is hyponatremic, we measured their plasma osmolality, and we see that they are truly hypoosmolar, meaning that that plasma osmolality is less than 275 mOsmol/kg, and that is what we refer to as hypotonic hyponatremia. 03:25 It's true physiologic hyponatremia that results from excess water, either due to ADH stimulation or impaired water excretion. 03:34 Now, there's two main classification systems for hypotonic hyponatremia. 03:40 We can either classify disorders according to ADH levels or to patient's volume status. 03:47 When we classify according to ADH levels, we can talk about those patients who have circulating ADH levels that are appropriately elevated, circulating ADH levels that are inappropriately elevated, or circulating ADH levels that are appropriately suppressed. 04:05 Alternatively, we can classify patients according to their volume status. 04:11 So, that would include patients who have hypovolemic hypotonic hyponatremia, euvolemic hypotonic hyponatremia, and hypervolemic hypotonic hyponatremia. 04:25 So, for the purposed of this lecture, I think it's easiest to really classify patients according to their volume status.
About the Lecture
The lecture Hyponatremia: Clinical Approach by Amy Sussman, MD is from the course Water Balance: Hypo- and Hypernatremia.
Included Quiz Questions
Which of the following statements is true regarding hypertonic hyponatremia?
- There is no actual increase in total body water.
- Water moves from the extracellular compartment to the intracellular compartment.
- It should be corrected with hypertonic saline.
- It can be caused by marked hypoglycemia.
Which of the following pairings is correct?
- Hypotonic hyponatremia, impaired water excretion
- Hypertonic hyponatremia, Acetazolamide
- Hypotonic hyponatremia, laboratory artifact
- Pseudohyponatremia, plasma osmolality = 300 mOsm
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |