Playlist
Show Playlist
Hide Playlist
Complement Deficiencies – Primary Immunodeficiency
-
Slides Immunodeficiency.pdf
-
Reference List Immune System.pdf
-
Download Lecture Overview
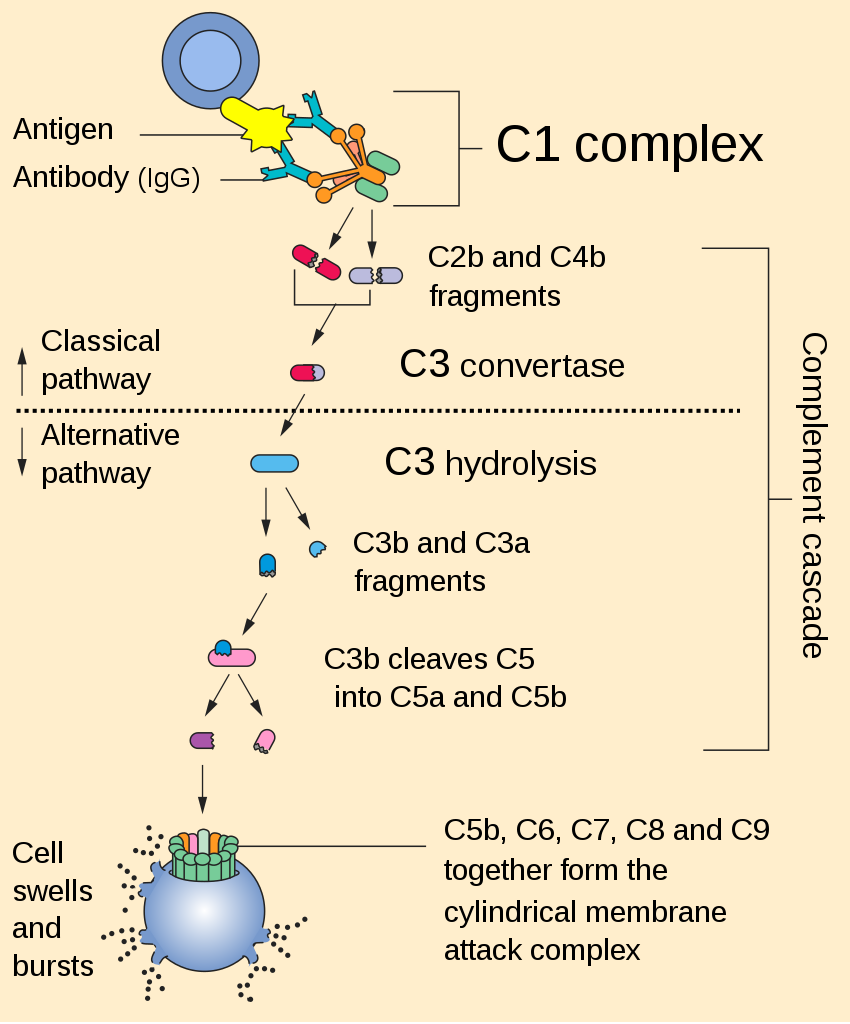
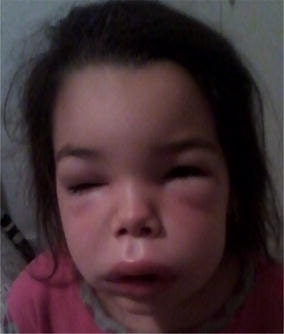
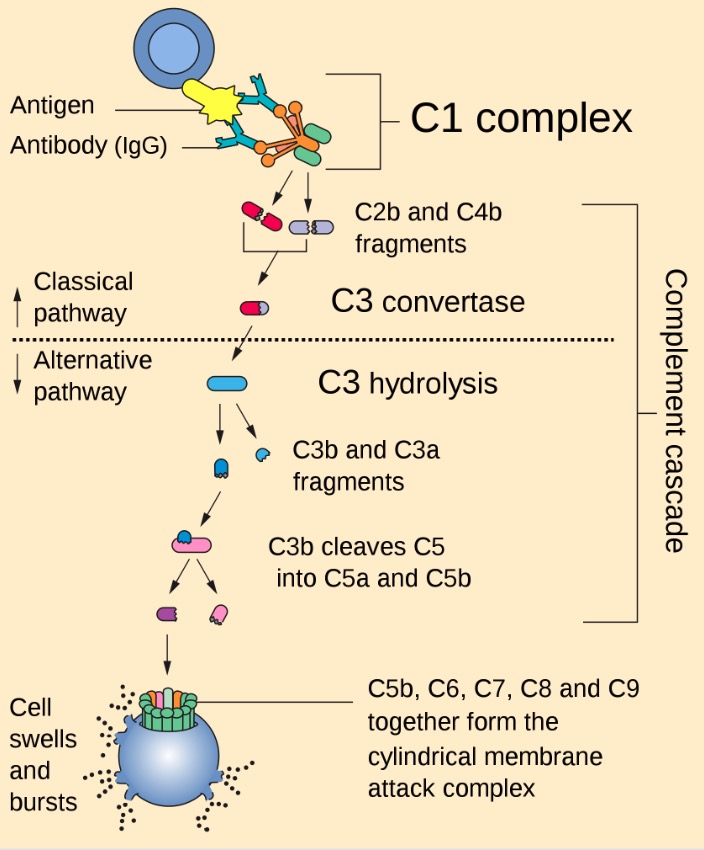
00:01 Looking at complement deficiencies, any one of the many different complement components can be affected. 00:09 For example, if there is a defective gene encoding the complement components C1q, C1r, C1s, C2, C3, C4 or Factor I, there will be impaired clearance of immune complexes. 00:25 And this can result in the condition, systemic lupus erythematosus which is an autoimmune disease. 00:33 And typical infections seen when there is deficiency of these particular complement components include a number of pyogenic bacteria. 00:44 Deficiencies in the later complement components, C6-- C5, C6, C7, C8, C9, Factor D, Properdin; this will lead to a range of other types of infections, particularly disseminated Neisseria infections. 01:02 MASP-2 deficiency, one sees an increased instance of Streptococcus pneumoniae. 01:09 And in mannose binding lectin deficiency, which has quite a high prevalence for a primary immunodeficiency, in fact it’s one of the most prevalent primary immunodeficiencies; there is usually no consequence. 01:22 So most people with mannose binding lectin deficiency don’t even know they’ve got it. 01:27 And the same is true of another relatively common primary immunodeficiency, which is selective-IgA deficiency. 01:33 Again, usually other parts of the immune response compensate for the lack, and one doesn’t see a greatly increased instance of particular infections. 01:43 One interesting complement regulatory component deficiency is paroxysmal nocturnal hemoglobinuria (PNH). 01:53 In this disease, there is a mutation in the PIG-A gene. 01:58 This encodes an α-1,6-N-acetyl-glucosaminyltransferase, which results in an inability to synthesize the glycosyl phosphatidylinositol, or GPI anchors. 02:13 And a lack of these GPI anchors mean that complement control proteins, CD55 and CD59 are absent from the surface of erythrocytes. 02:26 And this means that these erythrocytes are susceptible to complement-mediated lysis. 02:33 So normally, CD55 which requires a GPI anchor to hold it on to the cell surface of the erythrocyte, this molecule CD55 inhibits the C3 convertase, which is necessary for complement activation. 02:49 Likewise, CD59 which also requires a GPI anchor to hold it on the surface of the erythrocyte, this functions to prevent insertion of the membrane attack complex. 03:00 So in the absence of these GPI anchors, neither of these molecules is present on the surface of the red blood cell, and the membrane attack complex can cause lysis of the red blood cell resulting in PNH. 03:18 Hereditary angioedema is due to a C1 inhibitor deficiency. 03:25 C1 inhibitor is required to regulate the uncontrolled activation of the coagulation system. 03:34 So in its absence, there is excessive clotting. 03:38 It’s also required to regulate the complement system. 03:43 And in its absence there is excessive production of activated complement components resulting in inflammation and cell lysis. 03:52 And it also is involved in regulating the kinin system. 03:57 So there is excessive production of bradykinin and endothelial cell activation as a result.
About the Lecture
The lecture Complement Deficiencies – Primary Immunodeficiency by Peter Delves, PhD is from the course Immunodeficiency and Immune Deficiency Diseases. It contains the following chapters:
- Complement Deficiencies
- Paroxysmal Nocturnal Hemoglobinuria
- Hereditary Angioedema
Included Quiz Questions
In terminal complement (C5-C9) deficiencies, recurrent systemic infections with which of the following pathogens is most likely?
- Neisseria meningitidis
- Streptococcus pneumoniae
- Staphylococcus aureus
- Escherichia coli
- Candida albicans
A deficiency of which of the following complement components is LEAST associated with rheumatologic diseases such as systemic lupus erythematosus?
- C5
- C3
- C1
- C2
- C4
In patients with paroxysmal nocturnal hemoglobinuria, impaired conformation of the glycosylphosphatidylinositol-anchor results in the deficient expression of which of the following complement regulatory proteins? (CD: cluster of differentiation)
- CD55 and CD59
- CD1 and CD2
- CD3 and CD4
- CD5-9
- CD35 and CD46
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
1 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
I really like the professor, just wish the lecture was a bit more in depth