Playlist
Show Playlist
Hide Playlist
Chronic Leukemia – Leukemia
-
Slides Leukaemia.pdf
-
Reference List Hematology.pdf
-
Download Lecture Overview
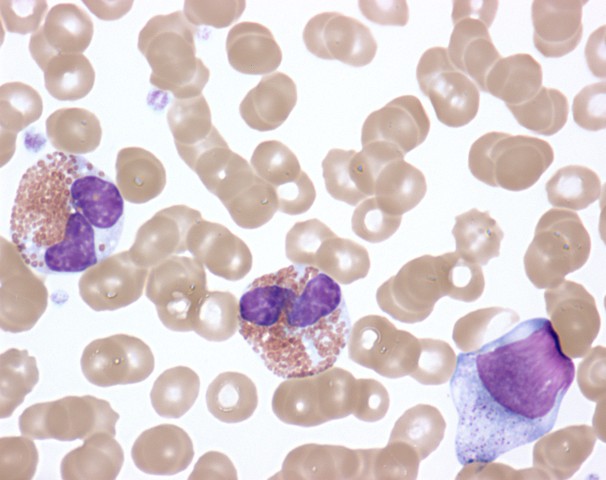
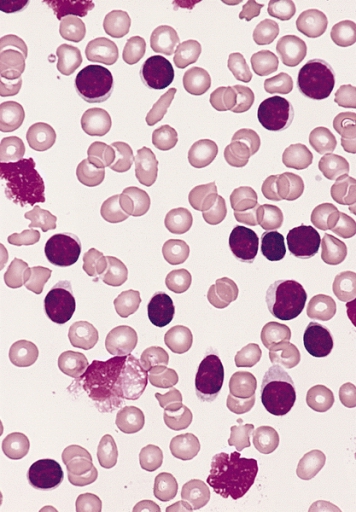
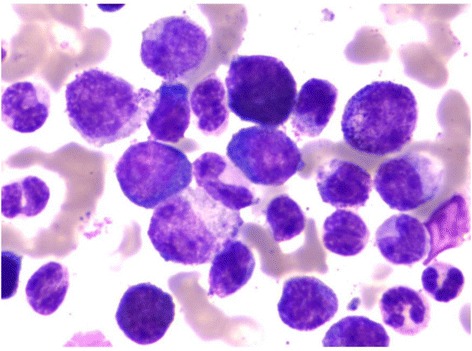
00:01 Now let us turn to the second major subset of leukaemias. We move from acute leukaemia into chronic leukaemia and I will start with chronic lymphoid leukaemia. This is the most common leukaemia in many societies. Interestingly not all societies because there seems to be a genetic predisposition to CLL. That is more common in those of Caucasian and Asian origin. CLL develops from the accumulation of a clone of B cells, which you can see on the blood film there in the bone marrow in the blood and in the lymph nodes. On that blood film, you will see many lymphocytes. You will see many purple smears and indeed very cells are lymphocytes, which have undergone destruction during the preparation of the blood film. Smear cells are a characteristic feature of CLL. 01:06 Now the B cells in CLL are very interesting. They have a number of unusual proteins on the surface, unusual for B cell. I have put two there, CD5 and CD23 and it is the presence of those proteins that we look for when are making a diagnosis of CLL in a patient who has too many lymphocytes within the blood. Now, CLL is quite slowly progressive. 01:41 It developed over many years and now from 90 percent of patients are diagnosed with CLL simply because they have blood taken for another reason. They may go for a health check or they may go as the doctor saying that I feel quite so well or dizzy or whatever it is, a blood test is done and we notice that there are too many lymphocytes. So it can be what we call an indolent disease slowly progressive and not needing any treatment. Indeed, treatment is generally only started when the patient needs it. We call this as a watch and wait policy. 02:22 So when do patients need treatment, if they develop anaemia, when the CLL crowd out the normal cells and the bone marrow and lead to anaemia or perhaps when the lymph nodes in the neck swell up and cause problems or the spleen, which can also be involved. 02:48 So it may seem unusual that we make the diagnosis of leukaemia, but don´t treat it. But certainly results from several years ago showed that if one treats too early, you can actually have a worse outcome than watching and waiting. 03:05 Genetic testing for management of CLL Analysis for the presence of mutations or deletion of the TP53 gene that encodes the p53 protein can guide the treatment plan p53 protien is needed for chemotherapy to kill the CLL tumor cell and so chemotherapy should not be used when it is mutated or missing. 03:35 Treatment of Chronic Lymphocytic Leukemia There are three main approaches for treating CLL by current time The first drugs that inhibit the Bruton tyrosine kinase protein. These, known as BTK inhibitors The second class of drugs are those that reduces the level of bcl-2 protein inside lymphocytes and therefore cause cellular apoptosis of the leukemia cells the main drug in this class is called Venetoclax finally in some patients a combination of a CD20 binding antibody together with some form of chemotherapy and this is called Immuno-chemotherapy Finally a fourth subset of leukaemia, chronic myeloid leukaemia. This is a relatively rare disorder but is seen in people of all ages and is remarkable because we know so much about its etiology and it lead to the introduction of a new treatment that is changed our approach to cancer. In many cases, it is caused by a translocation. This was the first translocation to be identified in cancer called the Philadelphia chromosome between chromosomes 9 and 22. Just look at that cartoon on the right and you will see those pink chromosomes. On the left of those pink diagrams, you will see the normal chromosome 9 and below it the normal chromosome 22. But if those break at ABL and BCR, they can stop that genetic material and lead to those pink chromosomes on the right, 9q+, 22q-. Now, what is fascinating about this translocation is it leads to the formation of a new protein, a fusion protein that is shown in the top right? BCR and ABL come together to form a new fusion protein only present within the tumor cells. As you will see on the right-hand side of the diagram, this protein encodes an enzyme, which has tyrosine kinase activity and this tyrosine kinase activity is overactive within the tumor cells and through a range of mechanism that leads to proliferation of this myeloid cell and generation of the leukaemia and as you will see, it drives the bone marrow to access proliferation of the myeloid compartment. The clinical features of CML reflect that myeloid proliferation. Let us look down the left-hand side first of all splenomegaly often you will see a very large spleen under the left-hand side of the ribs and patients with CML. Fevers, sweats and indeed gout because the patient is making so many cells and also many of these cells are dying and gout interestingly is precipitated by the formation of uric acid from DNA of cells that have been broken down. Let us look now on the right-hand side. You will see on the right the particular blood film of the patient with chronic myeloid leukaemia. Lots of neutrophils, but also earlier and more primitive cells that we should not see in the blood, promyelocytes and myelocytes and in those two tubes, we see the startling feature of CML that we see in some patients, very high numbers of leukaemic cells. Let me explain. On the left-hand side of those two tubes, you will see normal blood. It has been placed in the centrifuge and the bottom of the red cells or the top is the plasma and in between those two, there would normally be a very thin layer of white cells sometimes known as the buffy coat whereas in the right-hand tube, you can see that enormous white area that is the huge buffy coat of the malignant white cells within the blood of this patient. So if you ever wanted to see an example of how leukaemia really does mean white blood that is a typical picture. 08:35 Now, the treatment of chronic myeloid leukaemia is one of the fascinating areas of medicine. 08:44 Imatinib is a drug that was designed specifically treat chronic myeloid leukaemia and it can specifically block the BCR-ABL protein. You can already see how exciting that is because that fusion protein is only in the tumor cells. It is exactly the sort of drug that we have been looking for when it comes to specifically affecting the tumor cells as opposed to chemotherapy. 09:14 It is highly effective and simple oral medication can control the disease in most cases. 09:23 Now it is true that the disease can get resistant in some cases or the patient might perhaps be intolerant of Imatinib, but there are now several new drugs, which also inhibit BCR-ABL in different ways and indeed some evidence that they may be more effective than Imatinib as a first line of therapy. So there are many trials now seeing if these different drugs are effective at different stages and to bring about a more rapid and complete control of CML. But the treatment of this disease has been transformed. 10:07 There are some newer tyrosine kinase inhibitors that are used for treatment of CML Imatinib was inded ther original BCR-ABL tyrosine kinase inhibitor But nilotinib, dasatinib and bosutinib are econd generation TKI drugs that can also be used for first line therapy and ponatinib is the third generation TKI drug that can treat CML that has developed a T315I mutations
About the Lecture
The lecture Chronic Leukemia – Leukemia by Paul Moss, PhD, OBE, FMed, FRCPath is from the course Hematologic Disorders.
Included Quiz Questions
Which of the following is the MOST common presentation of chronic lymphoid leukemia?
- Elevated lymphocites noted on labs ordered for other reasons
- The patient develops indigestion and examination shows a greatly enlarged spleen
- Life threatening infection
- Severe bleeding
- Patient notices swollen glands in the neck
What is the treatment for chronic lymphocytic leukemia?
- A combination of chemotherapy and antibodies directed to B cells
- Radiotherapy
- Surgical removal
- Bone marrow transplant
Which of the following CD markers are positive in chronic lymphocytic leukemia?
- CD 5+ and CD 23+
- CD 3+ and CD 15+
- CD 10+ and CD 20+
- CD 5+ and CD 21+
- CD 8+ and CD 21+
Which B-cell receptor signalling pathway is blocked in order to inhibit the proliferation signals produced by tumor cells?
- BTK
- WT1
- p53
- PTEN
- K-ras
Which of the following is NOT associated with chronic myeloid leukemia?
- Calcium pyrophosphate
- Gout
- Massive splenomegaly
- Fever
- Sweats
Which of the following is a characteristic feature of the blood smear in chronic lymphocytic leukemia?
- Smudge cells
- Increased prolymphocytes
- Cells with basophilic stippling
- Cells with nuclear inclusions
- Increased myeloblasts
What causes the characteristic appearance of smudge cells?
- Preparation artifact causing rupture of the fragile cytoplasm of cells
- Swelling of the cells causes rupture of the fragile cytoplasm of cells
- Pressure from the surrounding cells causes rupture of the fragile cytoplasm of cells
- Accumulation of the mutations causes rupture of the fragile cytoplasm of cells
- Self destruction by apoptosis leads to rupture of the fragile cytoplasm of cells
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
2 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
Nice overview of the different treatments. Presentation is helpful in learning and remembering.
Dr. Moss is a wonderful lecturer! He cadence and speed is perfect to allow students the ability to grasp information. I learned so much.