Playlist
Show Playlist
Hide Playlist
Structure and Acidity – Carboxylic Acids and Their Derivatives
-
Slides 08 Chemistry Advanced Le Gresley.pdf
-
Download Lecture Overview
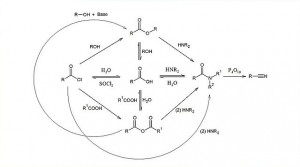
00:01 Inductive effect. 00:02 If you go back to what we were talking about in terms of bond polarisation, this has an effect of in terms of the delta positive or delta negative, dipolar or charge separation we observe in a particular bond. The acidity of a carboxylic acid can be increased if electron withdrawing groups are attached to it or decreased, if electron donating groups are attached to it. 00:25 So, let’s have a quick look at the pKa for some of these commonly occurring carboxylic acids. Pay particular attention to not just the nature of the substituent, but also its location on the alkanoic acid chain itself. Let us take, for example, the pKa in the top set on the board where in this scenario we are looking at a 1, 2, 3, 4 butanoic acid, okay, derivative, count your carbons. 00:53 So, in the case of the unadorned butanoic acid derivative, we have a pKa there of 4.8. 01:00 If we add an electron withdrawing chlorine group to the alpha position, we decrease the pH… pKa to 2, rendering this carboxylic acid, this 2 chlorobutanoic acid far more acidic than the starting unadorned carboxylic acid. If, on the other hand, we take that chloro group and we move it to the beta position or the 3 position on this butanoic acid chain, we see that it has a substantially less pronounced effect on the acidity of this carboxylic acid. 01:36 And finally, if we move the electron withdrawing group all the way back to the 4 position on the butanoic acid, we see that it has almost a marginal effect on the acidity of this carboxylic acid. 01:47 So, it’s not just the nature of the substituent, but also its position which is important when you are trying to modify the acidity of a carboxylic acid. Let us take, for example, however, the difference between the groups. I have used halogens, just for the case of ease, but there are a number of other electron withdrawing or electron donating groups. 02:08 Right. Okay. So, let’s have a look at methanoic acid which is shown in the bottom left of the board. This has a pKa of 3.75. If we compare that to acetic acid, shown there with a pKa of 4, we can see that it’s the actual electron donating effect of the CH3 group on the ethanoic acid which actually decreases the stability of the carboxylate. In the earlier equilibrium we showed the formation of the hydronium ion and the carboxylate, thus methanoic acid is actually more acidic, is a stronger acid than ethanoic acid. 02:46 Again, we talked earlier a little bit about the positioning of the different electron withdrawing groups, but I just want to touch upon the different types of electron withdrawing group as shown here in the bottom right of the board. We have substituted in the 2 or the beta position… alpha position - fluorine, chlorine and bromine. As you can see fluorine, which is substantially more electronegative than either chlorine or bromine, results in a stronger carboxylic acid in terms of its acidity. Chlorine is the next one along in terms of acidity and finally, the bromine substituted carboxylic acid is the weakest acid out of the group. 03:27 So, hopefully, you have been paying attention. So, now, what that means is you should be able to put the following structures in the correct order of decreasing acidity starting with the strongest acid and moving down to the weakest acid. I will give you a couple of minutes to attempt that, after which time, I will provide the answers. 03:53 Right. Okay. So, preparation of carboxylic acids. 03:59 As you’ll see a lot of the organic chemistry that we have covered is largely intertwined and all of the functional groups are related to one another or are interconvertible in between each other in one way, shape or form, but it is actually important to actually separate these out into core functional groups for the sake of clarity amongst other things. 04:18 But, the reality is that if we take an alcohol, primary alcohol or an aldehyde, if we oxidise it up with potassium permanganate, shown at the top, or potassium… sorry, potassium dichromate, shown at the top, or potassium permanganate, shown at the bottom, we can generate a carboxylic acid. In fact, in exhaustive oxidation, a carboxylic acid is usually what is generated any way. So, they are all relatively easy to form. 04:44 Carboxylic acid derivatives. 04:47 This is where the real synthetic interest lies. Carboxylic acids, most acidic, are not particularly in of themselves very reactive, as we all find out. The general formula for a carboxylic acid derivative is given here and comprises a number of different possible compounds. Note, where we have an R, which can be aliphatic or an aryl group such as a benzene ring attached directly to a carbonyl, double bound to the oxygen and then with a group, denoted Z. 05:18 We touched upon this in the previous lecture, where Z can either be an alkoxy group, an amine group or it can be a carboxyl group or it can be a halide group such as chloride or bromide. Note also, what I said before in the previous lecture slide about the fact that this polarisability or polarisation of that carbonyl double bond. So, you have a delta positive charge on the carbon, a delta negative charge on the oxygen is one of the things that actually adds to the reactivity of these carboxylic acid derivatives as we will see.
About the Lecture
The lecture Structure and Acidity – Carboxylic Acids and Their Derivatives by Adam Le Gresley, PhD is from the course Organic Chemistry.
Included Quiz Questions
Complete the following statement. The acidity of carboxylic acids is caused by …
- … the attachment of a hydroxyl group to the carbonyl carbon and the tendency of the carboxylic acid group to readily ionize into proton and carboxylate ion.
- … the attachment of an alkyl group to the carbonyl carbon.
- … the attachment of an aryl group to the carbonyl carbon.
- … the absence of direct attachment of any hydrogen atom to the carbonyl carbon.
- … certain unknown factors.
Complete the following statement. Ionization of carboxylic acids produces …
- … carboxylate and hydronium ions.
- … carboxylate ions only.
- … hydronium ions only.
- … protons and carboxylate ions.
- … protons only.
Which of the following is NOT true about ionizations of carboxylic acids?
- pKa value merely represents a number with no significant information.
- The acid dissociation constant of a carboxylic acid is a ratio of the equilibrium concentrations of ionization products like conjugate base and hydrogen ions to the equilibrium concentration of the carboxylic acid.
- Ethanoic acid, ethanoate ions and hydronium ions are said to be in equilibrium when there is no change in their concentrations with the passage of time.
- Ka = ([A-] × [H+]) ÷ [HA] equation represents the acid dissociation constant of a carboxylic acid.
- pKa value represents the acidic strength of an acid.
Which of the following is true about electronegative substituents?
- They enhance the acidity of carboxylic acids when they are placed near the carbonyl group.
- They decrease the acidity of carboxylic acids when they are placed near the carbonyl group.
- They enhance the acidity of carboxylic acids, when they are placed away from the carbonyl group.
- They decrease the acidity of carboxylic acids when they are placed away from the carbonyl group.
- They do not affect the acidity of carboxylic acids.
Which of the following is true about 2-fluoroheptanoic acid?
- It is more acidic in nature than 6-fluoroheptanoic acid.
- It has a less acidic nature than 6-fluoroheptanoic acid.
- It has the same acidic nature as 6-fluoroheptanoic acid.
- It is less acidic in nature than 7-bromoheptanoic acid.
- It has a less acidic nature than 6-chloroheptanoic acid.
What determines the acidic character of a carboxylic acid?
- The inductive effect and position of substituent regarding the carbonyl group
- Some unknown factors
- The carbon atom present at the alpha position to that of the carbonyl group
- The carbon atom present at the beta position to that of the carbonyl group
- The carbon atom present at the delta position to that of the carbonyl group
Which of the following is an INCORRECT statement?
- Resonance phenomenon does not play any role in the stability of a conjugate base and hence does not alter the acidic strength of a carboxylic acid.
- With an increase in the carbon chain length of carboxylic acids, the acidic nature of the carboxyl group decreases.
- Any alkyl group substitution near to the carbonyl group decreases the acidic strength of carboxylic acids due to its electron donation effect.
- The substitution of fluorine at the alpha carbon enhances the acidic strength of propanoic acid to a greater extent than bromine substitution at the same position.
- Propanoic acid is more acidic than propanol because the negative charge gets distributed over two oxygen atoms in the propanoate ion, whereas in the propoxide ion, the negative charge stays on the single oxygen atom.
Complete the following statement. Carboxylic acid production can be carried out …
- … from the corresponding aldehyde or methanol or primary alcohols via exhaustive oxidation in the presence of KMnO4 or K2Cr2O7.
- … from the corresponding aldehyde or alcohols via exhaustive reduction in the presence of haloacids.
- … from the corresponding alkanes via exhaustive reduction in the presence of metal catalysts.
- … from CO2 and H2O in the presence of sunlight.
- … from the corresponding alkenes via hydrogenation process in the presence of Pd.
Why are acyl halides more reactive than the parent carboxylic acids, whereas amides are the least reactive?
- In acyl halides, the halogens impart a negative inductive effect, whereas amides release electrons towards the carbonyl group.
- In acyl halides, the halogens impart a positive inductive effect, whereas amides pull electrons away from the carbonyl group.
- In acyl halides, halogens lead to non-polarization of carbonyl group, whereas amides intensify polarization on the carbonyl group.
- In acyl halide, a stronger conjugate base is formed, whereas in amides a relatively weaker conjugate base is formed.
- In acyl halide, the halogen atom prevents delocalization of negative charge, whereas in amides there is a better delocalization of negative charge.
Which of the following shows the correct order of relative reactivity for carboxylic acid derivatives?
- RCOCl > RCOOCOR > RCOSR > RCOOH > RCONHR
- RCOOCOR > RCOSR > RCOCl > RCOOH > RCONHR
- RCONHR > RCOOH > RCOSR > RCOOCOR > RCOCl
- RCONHR > RCOCl > RCOSR > RCOOH > RCOOCOR
- RCOCl > RCONHR > RCOOH > RCOSR > RCOOCOR
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |