Playlist
Show Playlist
Hide Playlist
Renal reabsorption of HCO3 – Acid Base Balance
-
Slides 08 MetabolicAcidosisAlkalosis AcidBaseBalance GeneralPhysiology.pdf
-
Download Lecture Overview
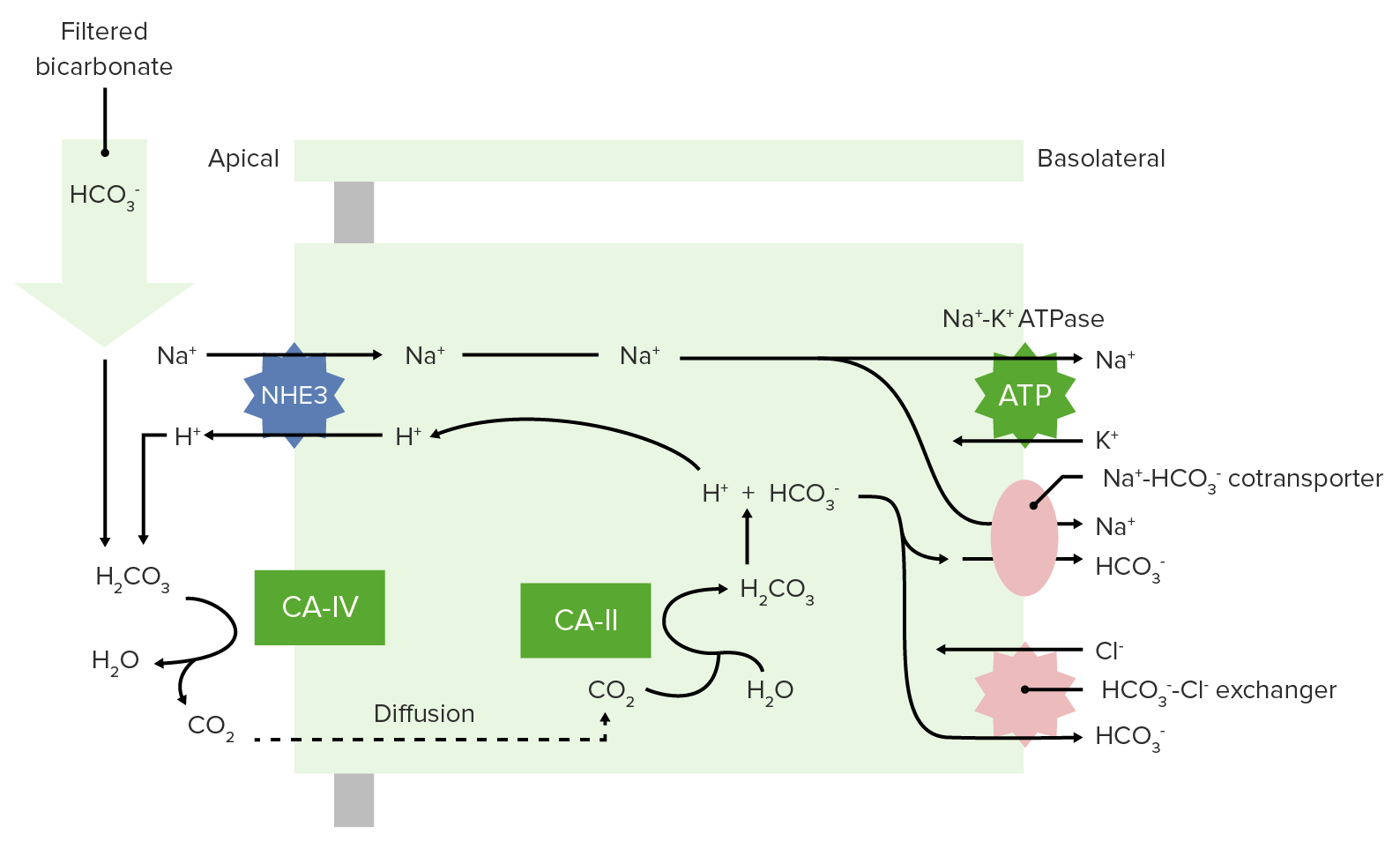
00:01 Bicarb reabsorption occurs in the nephron. 00:05 About 80 percent of it is pulled off in the proximal tubule, about 10 percent in the thick ascending limb, about 6 percent in the distal convoluted tubule and about 4 percent in the collecting duct. 00:19 And I like to highlight here that you really absorb all of your bicarb. 00:23 you will not urinate any of it out in normal conditions. 00:27 In the proximal tubule, but let’s now take this into a little bit more detail. 00:33 How are you reabsorbing the bicarb? It’s actually more complex than you think because bicarb is a charged molecule. 00:44 If we don’t have a specific transporter, that’s going to be able to move it across the apical membrane. 00:52 So, we have a little and genius way of doing this. 00:55 We utilize a transporter called the NHE3, sodium hydrogen ionic exchanger. To kick out a hydrogen ion. 01:08 It kicks out this hydrogen ion combines to form bicarbonic acid. 01:14 Carbonic anhydrase then converts it into water and carbon dioxide. 01:22 So, now you have water, which can be in the renal tubule and carbon dioxide, which now can freely move across the membrane. 01:32 It moves into the cytosol, you have a different carbonic anhydrase, which is the enzyme that helps convert this back to bicarbonate acid. 01:42 So, you have two carbonic anhydrases. 01:45 Carbonic anhydrase 4, which is on the apical membrane. Carbonic anhydrase 2, which is in the cytosol. 01:53 This complex process all it did was allow you to get a molecule across the apical membrane. 02:01 Now, once you have formed carbonic acid it once again can disassociate into a hydrogen ion which then can be used to be kicked back out through the sodium hydrogen exchanger. 02:14 and the bicarb can be reabsorbed across the basolateral membrane. 02:19 So, that’s how you reabsorb 80 percent of your bicarb through this mechanism. 02:25 Across the basolateral membrane, which is the opposite side of the apical membrane, where the sodium hydrogen ion exchanger occurs. 02:33 There is two different ways we move bicarbonate out of the cell. 02:38 The first way to do that is by a co-transporter that involves sodium. 02:47 The sodium co-transporter then allows for both bicarb and sodium to travel across the basolateral membrane following the sodium gradient to move out bicarb. 03:01 The second mechanism that you would have to move bicarb out of the basolateral membrane is through an exchanger. 03:09 This exchanger uses chloride, and chloride moves into the cell and bicarb moves out of the cell. 03:17 This is an important process to think about because you need to now get the bicarb, which was in the cytosol out across the basolateral membrane. 03:27 So, either co-transported with bicarb and sodium or exchanged for chloride.
About the Lecture
The lecture Renal reabsorption of HCO3 – Acid Base Balance by Thad Wilson, PhD is from the course Acid-Base Balance.
Included Quiz Questions
Which segment of the nephron reabsorbs the greatest quantity of bicarbonate filtered from the blood?
- Proximal convolute tubule
- Thick ascending limb
- Distal convoluted tubule
- Cortical collecting duct
Which isoform of carbonic anhydrase is located on the apical membrane in the proximal convoluted tubule?
- Carbonic anhydrase IV
- Carbonic anhydrase I
- Carbonic anhydrase II
- Carbonic anhydrase III
Which of the below transporters is located on the apical membrane of the proximal tubule cell?
- NHE3
- Na-HCO3 cotransporter
- Na-K ATPase
- Anion exchanger (Cl-HCO3)
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |