Playlist
Show Playlist
Hide Playlist
Reactivity – Carboxylic Acids and Their Derivatives
-
Slides 08 Chemistry Advanced Le Gresley.pdf
-
Download Lecture Overview
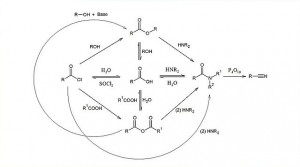
00:01 So, let’s have a brief look at the reactivity of carboxylic acid derivatives as a general group. This is the order of reactivity starting with the least reactive on the right hand side and going to the most reactive on the left hand side. And this, to be honest with you, is a crucial element when it relates to the biology. As you know, proteins have a primary structure which consists of amino acids joined by a series of amide bonds to each other. And so, therefore, this is rather a good job that they are the least reactive, otherwise you would be getting a relatively easy, simple hydrolysis of these amide bonds of your proteins. 00:38 As it happens, specific enzymes are usually required to break these down; enzymes known as peptidases or amidases. If we move from right to left, we can see that the degree of reactivity goes along in terms of nucleophilic addition elimination reactions as carboxylic acid, followed by carboxylic ester, followed by carboxylic acid, followed by anhydride until finally we get to the most reactive, an acid chloride. In fact, acetyl chloride is so reactive that it is otherwise known as a lachrymator. 01:12 What happens is that fumes of acetyl chloride in a laboratory, if they are exposed to the mucous membranes like, for example, in the eyes, can react with the water and generate quantities of hydrochloric acid and also of the acetic acid and this is irritant and causes you to cry, hence the term lachrymatory, that’s how reactive they are. 01:35 These are the types shown in that rectangle that you tend to use where you can, in order to carry out an actylation reaction, as they are, generally speaking, the easiest to work with and afford you the largest yields. The reactivity, as you can appreciate, depends on the electrophilicity of the carbon-oxygen double bond. And so, what I’d like to do before we go on is draw your attention to the electronegativity of the groups attached to the carbonyls. 02:03 In the case of the chlorine, we know that it is electronegative because it is in group 7. And so, this… what this does is render the delta positive on the carbon even more pronounced and even more delta positive than on, for example, the carboxylic acid. So, aside from just affording us a good leaving group as the chloride, it also actually makes that carbonyl carbon more electrophilic than in the case of either the carboxylic ester and certainly, in the case of the amide. 02:30 So, let’s have a look. How do we make these things, at the end of the day, since obviously, a carboxylic acid in itself is not particularly useful from a synthetic perspective very often, how do we actually make these things? So, I am going through, here, just a few reactions as to how you can convert a carboxylic acid into an ester, an acid chloride, an acid anhydride and also, an amide. 02:54 So, in the first instance, conversion into an ester can be accomplished by dissolving the carboxylic acid up usually in an alcohol of choice. In this case, we have shown the OR group in green, this could be an ethoxy group and this could be a propoxy group, it’s largely irrelevant. And this is known as a Fischer Esterification. So, you add a catalytic amount of H+ in the form of maybe a mineral acid like hydrochloric acid and it converts the carboxylic acid into the ester. 03:25 Now, this is okay if it’s possible in your reaction to be able to heat the thing up under reflux, but if this is going to cause problems with other parts of the molecule then there are simpler ways and coupling reagents and something otherwise known as the Mitsunobu reaction, which if you are really interested about how esters are formed, you may wish to read up about. 03:44 Making acid chlorides is also can be a little bit on the tricky side, but this is facilitated in the same way that alcohols can be converted to haloalkanes by thionyl chloride or SOCl2. 03:58 Another way of converting these is via PCl5, this is phosphorous pentochloride. Again, care should be taken when working with either of these compounds as they can produce toxic fumes. 04:10 In the third scenario, an acid anhydride, this is where we have taken two acids, put them together and removed water. This can be facilitated in a number of ways, one of those is actually by reacting a carboxylic acid directly with an acid chloride generating hydrochloric acid and the associated acid anhydride. However, the way I have shown here is to use phosphorous pentoxide which is a very strong dehydrating agent which removes water from those two carboxylic acids forming the acid anhydride. 04:43 And finally, amide formation. Now, I have shown a very basic way of generating an amide from a carboxylic acid. You take an amine, which we will see in the next lecture, and then you heat it up, usually in the presence of an acid catalyst, and you dehydrate it. 05:01 The conditions for this are sometimes really, really unpleasant, in terms of the actual temperatures you have to achieve and the length of time you have to get it under reflux. 05:11 When you are talking about synthesising polypeptides from a biological perspective, there are far better ways of doing it than this, but this just demonstrates the basic principles by which OH-, in this case, can be lost from the carboxylic acid and an amine can be put in its place. If you are interested in looking at polypeptide synthesis and coupling agents, then I recommend that you look at dicyclohexylcarbodiimide, that is a basic one and although also, EDCI and [Unaware 00:27:11] as another.
About the Lecture
The lecture Reactivity – Carboxylic Acids and Their Derivatives by Adam Le Gresley, PhD is from the course Organic Chemistry.
Included Quiz Questions
Complete the following statement. The breakdown of peptide bonds present in the primary structure of proteins takes place …
- … in the presence of peptidase or amidase via a hydrolysis reaction.
- … in the presence of alcohols via a hydrolysis reaction.
- … in the presence of mineral acids via a hydrolysis reaction.
- … in the presence of a base via a hydrolysis reaction.
- … in the absence of water via an esterification reaction.
Complete the following statement. Acetyl chloride (CH₃COCl) and Chloroacetyl chloride (C₂H₂Cl₂O) act as lachrymator compounds as …
- … these react with water present in the mucous membranes to produce acidic derivatives irritable to eyes.
- … these get absorbed into the mucous membranes.
- … these react with oxygen to produce oxides.
- … these get reduced by hydrogen.
- … these react with DNA molecules and cause mutations.
Complete the following statement. The reactivity of carboxylic acid derivatives depends on the …
- … electrophilic nature of carbonyl group.
- … number of hydrogen atoms directly attached to a carbonyl group.
- … temperature of the reaction mixture.
- … moles of water molecules present in the reaction mixture.
- … presence of mercury as a contaminant.
Why is acetyl chloride more reactive than acetic acid?
- The chlorine atom directly attached to the carbonyl group makes a positive delta charge on the carbon atom more pronounced. It also acts as a good leaving group during the nucleophilic substitution reaction.
- The chlorine atom makes the negative delta charge on carbon atom more pronounced.
- The chlorine atom acts as a poor leaving group during a nucleophilic substitution reaction.
- The chlorine atom serves as a catalyst during nucleophilic substitutions.
- The chlorine atom protects the carbonyl group from nucleophilic attack.
Which of the following is a WRONG statement about Fischer esterification?
- Fisher esterification is a nucleophilic addition reaction.
- The alcohol used during the reaction acts as both a solvent and reactant.
- Acids like H₂SO₄ , HCl or tosic acid is used as a catalyst to facilitate the esterification process.
- Fischer esterification is an example of a nucleophilic substitution reaction.
- Fisher esterification involves the addition of alcohol followed by the elimination of a water molecule.
Which of the following does not describe the production of a carboxylic acid derivative from a parent carboxylic acid correctly?
- CH₃COOH + C₂H₅OH → C₂H₅COOCH₃
- C₃H₇COOH + SOCl₂ or PCl₅ → C₃H₇COCl
- C₂H₅COOH + P₂O₅ → C₂H₅COOCOC₂H₅
- CH₃COOH + C₂H₅OH + H₂SO₄ or HCl → CH₃COOC₂H₅
- C₄H₉COOH + (C₆H₅)2NH + H₂SO₄ or HCl → C₄H₉CON(C6H5)₂
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |