Playlist
Show Playlist
Hide Playlist
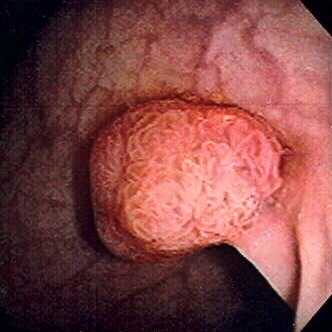
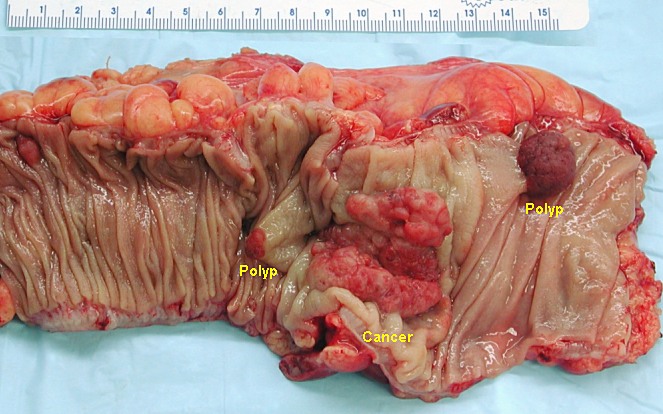
Polyp-Cancer Sequence
-
Slides Colon cancer General Surgery.pdf
-
Download Lecture Overview
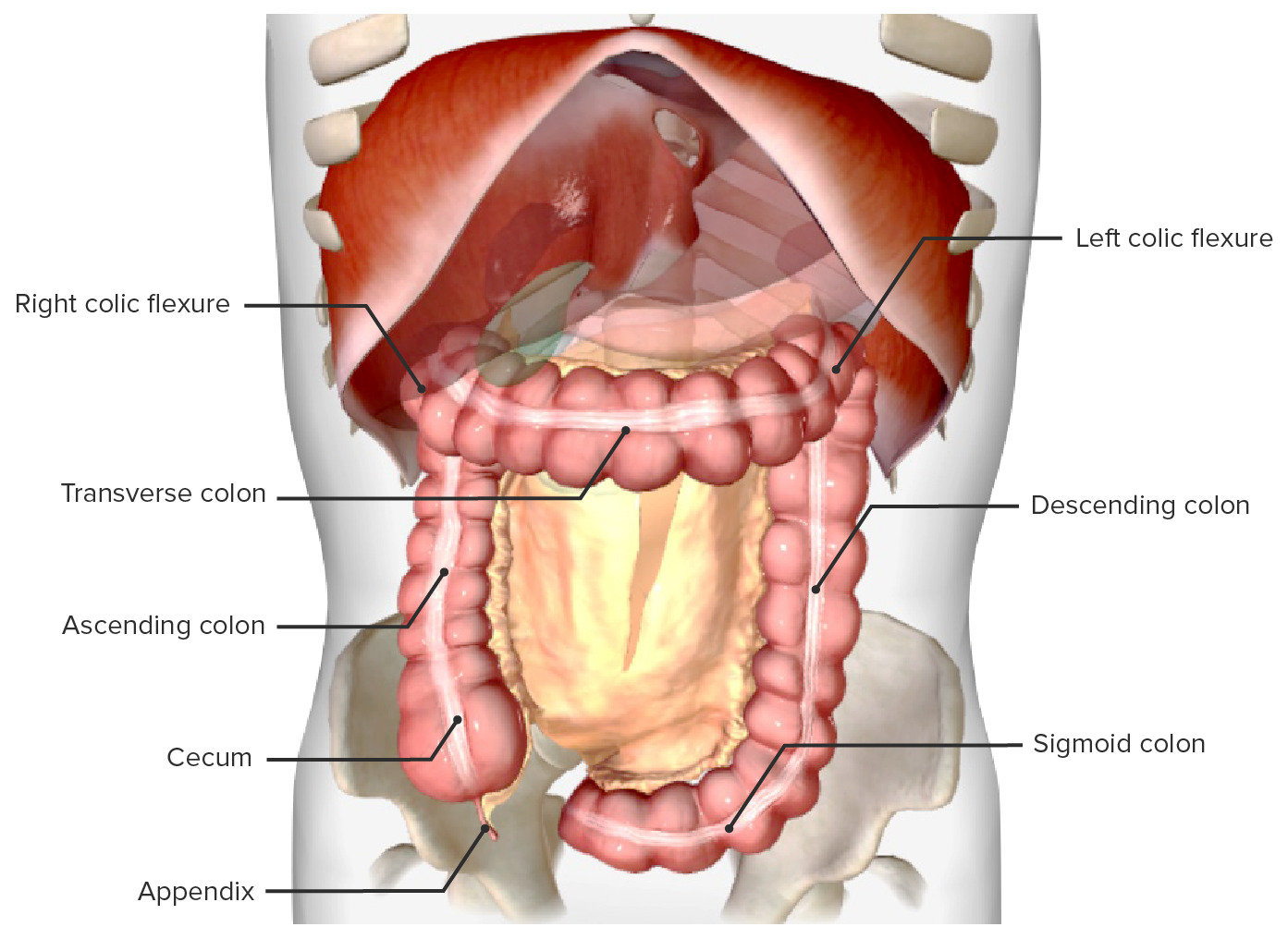
00:01 Now, let's talk about the predictable pathway from a polyp to a cancer. 00:05 This is called appropriately the polyp-cancer sequence. 00:11 Here's a slide that demonstrates a mutation from normal epithelium on the left side of the screen to a full long carcinoma. 00:19 Through this process, there are predictable sides of genetic mutation that allows hyperproliferation, forming a polyp, eventually degrading to a carcinoma. 00:32 Let's look at some of this in detail. 00:34 First mutation that occurs is an APC-tumor suppressor gene. 00:39 This allows tumor initiation. 00:41 Now, remember, tumor suppressor genes, which we have a lot of its main job is to suppress overgrowth. 00:51 K-Ras is a proto-oncogene. 00:52 K-Ras is a proto-oncogene mutation only causes hyperplastic syndrome. 00:56 But if it follows APC in this predictable line of mutations, it leads to adenoma. 01:01 Scientists are still working on why the K-Ras in this context leads to adenoma versus the mutation alone, that only leads to hyperplastic polyps. 01:10 This then progress to a deleted in colorectal cancer, which is yet another tumor suppressor gene. 01:16 And finally, the p53, which leads to apoptosis. 01:21 Mutations of p53, then allows a cancer phenotype. 01:27 Let's review for a second, another question. 01:30 What's the difference between Lynch Syndromes 1 and 2? I'll give you a second to think about this. 01:40 That's right. 01:40 Lynch syndrome 1, again, is a familial colon cancer without extracolonic manifestations versus Lynch syndrome 2, its associated with other cancers, particularly of the GI and reproductive tumors. 01:56 Amsterdam criteria is an important set of criteria for the diagnosis of Lynch syndrome or hereditary nonpolyposis. 02:04 This is the classic definition of three relatives with colon cancer over two generations, at least one of which was diagnosed at a fairly young age. 02:14 There have been subsequent iterations of this diagnosis. 02:17 But the bottom line is fairly similar in that we want to see multiple young people diagnosed with colon cancer in a family over multiple generations suggestive of number one, a genetic mutation and two, potentially an autosomal dominance. 02:32 Here's Amsterdam criteria II. 02:35 Amsterdam criteria II liberalize the definitions a little bit. 02:40 Here, the requirements that there should be at least three relatives with any Lynch syndrome associated cancer, be a colorectal, cancer of the endometrium, small bowel, ureter, or renal pelvis. 02:52 One should be a first-degree relative of the other two, at least two successive generations much like the original definition, and at least one should diagnosed before the age of 50. 03:02 Again, very familiar to you. 03:04 Remember that FAP should be excluded in colorectal cancer cases. 03:08 Tumors should be verified by pathologic examination. 03:15 The another criteria that called the Bethesda criteria is also used for Lynch syndromes. 03:21 In the Bethesda criteria, colorectal cancer diagnosed in a patient who is less than 50 years of age. 03:27 There has to be presence of synchronous or metachronous colorectal. 03:31 Synchronous or metachronous, meaning at the same time or at different times, or other nonpolyposis-associated tumors, regardless of age, that the colorectal cancer with a gene repair mutation and the histology diagnosing a patient who is less than 60 years of age. 03:51 The colorectal cancer diagnosing a patient with one or more first degree relatives in a nonpolyposis related tumor. 03:59 And lastly, that the colon cancer is diagnosed in a patient with two or more first or second degree relatives. 04:05 Basically different spins on the same criteria of multiple relatives, multiple generations, and diagnosis early on. 04:15 What are some physical findings in colorectal cancer? Unfortunately, the vast majority of patients do not have a classic profile of physical findings that will allow us clinicians to detect it early on. 04:27 That's why screening is so important. 04:29 Usually, there is no pain. 04:31 Maybe there is bleeding, maybe a large bowel obstruction. 04:36 That's why due to colonoscopy, early detection is the most important thing that we can offer to our patient. 04:42 By the time you wait for classic physical findings of bleeding and obstruction, it's likely that the colorectal cancer has already progressed into late stages. 04:52 We now consider colon surgery preventable due to screening. 04:56 And because of the predictable deterioration from a polyp to cancer in the polyp-cancer sequence. 05:04 At this point, it's worth mentioning the difference between left-sided lesions and right-sided lesions. 05:08 Left-sided lesions are more likely to present with symptoms of obstruction, such as constipation or decreased stool caliber. 05:15 On the other hand, right sided lesions are more commonly present with bleeding, macroscopic or microscopic, or iron deficiency anemia. 05:22 The reason that right-sided lesions do not cause obstruction is that the diameter of the right colon is greater than the left, and the stools have increased water content. 05:32 Again, no set of routine labs is indicative of colorectal cancer. 05:38 We may find a drop in the hemoglobin, hematocrit due to a slow bleed. 05:44 We oftentimes get a CEA or Carcinoembryonic Antigen, both before and after surgery. 05:52 We don't use that as a screening tool, however, but it is very useful as a follow up after treatment for detection or recurrences.
About the Lecture
The lecture Polyp-Cancer Sequence by Kevin Pei, MD is from the course General Surgery.
Included Quiz Questions
Which one of the following is the correct sequence of mutations which lead to colon cancer?
- APC --- K-ras --- TP53
- APC --- TP53 --- K-ras
- K-ras --- TP53 --- APC
- K-ras --- APC --- TP53
- TP53 --- APC --- K-ras
Which of the following gene mutations is correctly matched to its phenotype?
- APC gene mutation; tumor initiation
- K-ras mutation alone; colon cancer
- K-ras mutation following APC gene mutation; hyperplastic syndrome
- DDC gene deletion; tumor suppression
- P53 mutation; apoptosis of deleterious cells
Which of the following patients’ histories meets the Amsterdam Criteria I for diagnosis of hereditary non-polyposis colon cancer?
- A patient has three relatives with colon cancer over two successive generations, one of which was diagnosed at the age of 35.
- A patient has two relatives with colon cancer over two successive generations, both of whom were diagnosed before the age of 50.
- A patient has three relatives with colon cancer over three successive generations, all of whom were diagnosed after the age of 65.
- A patient has three relatives with colon cancer over three successive generations, all of whom were diagnosed after the age of 50.
- A patient has 1 relative with colon cancer over two generations, who was diagnosed at the age of 74.
Which of the following patients’ family histories meets the Amsterdam Criteria II for hereditary non-polyposis colon cancer?
- A patient with a family history of a mother with endometrial cancer, and two cousins with non-familial adenomatous polyposis colon cancer who were both diagnosed before the age of 50.
- A patient with a family history of a grandfather with colorectal cancer and a brother with renal cancer diagnosed at the age of 48.
- A patient with a family history of a mother, grandmother, and sister all affected by breast cancer before the age of 60.
- A patient with a family history of two brothers and a father all with familial adenomatous polyposis colon cancer.
- A patient with a family history of a grandfather with colorectal cancer diagnosed at the age of 68, a brother with small bowel cancer diagnosed at the age of 52, and a third cousin with lung cancer diagnosed at the age of 48.
All of the following are Lynch syndrome-associated cancers except?
- Prostate cancer
- Small bowel cancer
- Endometrial cancer
- Ureter cancer
- Renal pelvis cancer
An important tool to diagnose hereditary nonpolyposis colon cancer is the Bethesda criteria, which is described by all of the following EXCEPT?
- Colorectal carcinoma with high microsatellite instability astrologically diagnosed in a patient less than 50 years old
- Colorectal carcinoma diagnosed in a patient who is less than 50 years old
- The presence of synchronous or metachronous colorectal carcinoma or other Lynch syndrome-associated tumors
- Colorectal carcinoma diagnosed in one or more first-degree relatives with a Lynch syndrome-associated tumor
- Colorectal carcinoma diagnosed in two or more first-degree or second-degree relatives with Lynch syndrome-associated tumors, regardless of age
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |