Playlist
Show Playlist
Hide Playlist
Mineralocorticoids and Glucocorticoids – Adrenal Pharmacology
-
Slides Mineralocorticoids Glucocorticoids Adrenal Pharmacology.pdf
-
Reference List Pharmacology.pdf
-
Download Lecture Overview
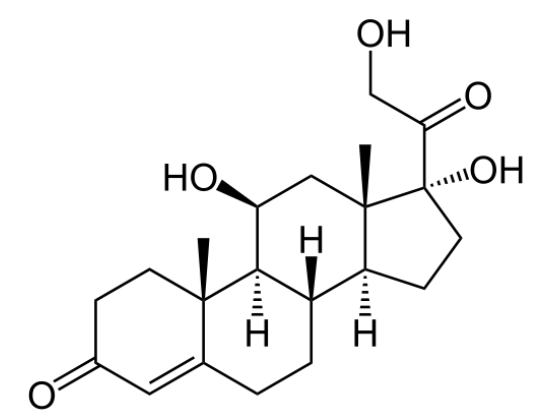
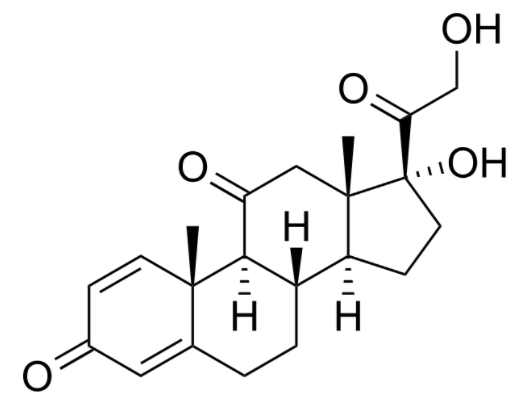
00:01 Let's move on to the zona glomerulosa and mineralocorticoid function. 00:07 Now remember that when we are talking about mineralocorticoids or even corticosteroids, we are talking about using a steroid receptor. 00:15 If you remember from your pharmacology lectures way back at the beginning, when we were looking at receptors, there are five major types of receptors. 00:24 Steroid receptors are intracellular receptors and so the drug, because it's lipophilic can cross the membrane easily and that's how the medications or the hormones work. 00:37 Now, if the steroid binds to the receptor inside the cell, it has to send a message into the nucleus. 00:45 It does so through something called an MRE or mineralocorticoid response element. 00:51 This MRE is part of the DNA molecule. 00:56 The MRE exerts its effects through DNA and you have production of certain enzymes. 01:02 Now the aldosterone is an endogenous mineralocorticoid that is part of the renin angiotensin system, and it has a very strong salt retaining activity by increasing sodium reabsorption from the distal renal tubules. This is coupled with the secretion of potassium and hydrogen ions into the urine, and will also encourage water reabsorption. 01:24 So, exogenous mineralocorticoids will mimic this effect. 01:27 A good example of a mineralocorticoid is Florinef or fludrocortisone. 01:30 Structurally it's very similar to cortisol. 01:36 It has moderate glucocorticoid activity and very strong mineralocorticoid activity. 01:42 It is involved and can be treating cerebral salt wasting syndrome. 01:48 It can be used in Addison's disease. 01:50 It can be used in this disease called POTS or paroxysmal orthostatic tachycardia syndrome. 01:56 And it's also sometimes used in geriatric orthostatic hypotensive syncope. 02:01 So, what we're doing here is we're retaining salt so that the blood pressures are a little bit higher and it prevents the drop in blood pressure that geriatric patients have when they stand up. 02:14 Spironolactone is a diuretic. 02:18 We discussed it already in the hypertension section of pharmacology, but I just want to bring it up here because it competes with aldosterone for receptors sites in the distal renal tubule, inhibiting aldosterone’s effects and resulting in sodium and water excretion while preserving potassium and hydrogen ions. 02:36 It also has anti-androgen activity, making it useful in the management of hirsutism. 02:42 Let's move on to the zona fasciculata. 02:45 This is where glucocorticoid production is occuring. 02:49 Now, glucocorticoids also enter the cell as a free molecule. 02:53 It doesn't need to be converted because it's already lipophilic. 02:57 It binds to the intracellular receptor or other proteins, and then enters into the nucleus as a complex. 03:05 Now, instead of an MRE, it binds to a GRE or the glucocorticoid response element which is still also a portion of the DNA. 03:15 The GRE regulates gene expression and transcription for its various functions. 03:21 Now, when we use synthetic glucocorticoids in patients, we have side effects. 03:26 That metabolic effect can be gluconeogenesis, lipolysis and reduced subdermal fat. 03:33 Morphologic changes can have lipid deposits in certain spots such as the face, the neck, and the shoulder. 03:39 So I want to go over that just again so it's clear. 03:43 Reduced subdermal fat refers to, say the back of the skin, so people will have parchment-like skin when they are given synthetic glucocorticoids for a long time. 03:53 People who are given synthetic glucocorticoids will have more fat in the face, shoulder and neck. 03:58 So, I say that they have a 'moon face', they have a round kind of full face, but they have thin parchment-like skin. 04:07 There are some catabolic effects as well. 04:10 Protein catabolism, and tissue wasting, and osteoporotic effects are all part and parcel of glucocorticoid excess activity. 04:19 So in other words, patients who are placed on synthetic glucocorticoids can have protein wasting and reduction of muscle mass. 04:27 And osteoporosis is a major concern. 04:30 And premature bone fracture is a major concern on people who are taking synthetic glucocorticoids for a long time. 04:38 Side effects can also be immunosuppressive as well. 04:41 Cell mediated immunity can be affected by long term glucocorticoid use. 04:47 For example, lymphocyte-dependent infections can become more rampant and so you have to be very careful of patients developing severe viral infections or increased susceptability to viral infections. 05:01 Renal function can be compromised. 05:03 Remember cortisol is normally required for water excretion. 05:06 Anti-inflammatory effects,or actually, I wouldn't call it necessarily a side effect, it's actually the effect we are looking for. 05:14 There is often a dramatic suppression of multiple markers. 05:17 That's often why we use synthetic glucocorticoids in high doses in the first place. 05:23 There is some neuropsychiatric issues. 05:25 Excess cortisol causes giddiness and euphoria, and synthetic cortisol derivatives can cause that same kind of behaviour change. 05:37 The prototypical synthetic glucocorticoid is prednisone. 05:42 It has a long half life, it has quite good membrane penetration, and it's often used in acute inflammation in most disease states. 05:51 It's often a steroid replacement therapy as well for people who have adrenal suppresion. 05:56 Dexamethasone is another synthetic glucocorticoid. 06:01 It has an even longer half life than the original prednisone. 06:05 It has even better membrane penetration. 06:08 It is often used in cancer chemotherapy as part of an adjunctive treatment regimen. 06:15 Triamcinolone, beclomethasone, budesonide are other glucocorticoids. 06:20 Beclomethasone is readily penetrating the respiratory mucosa so we often use it in pulmonary inhalers. 06:29 Let's take a look at glucocorticoid synthesis inhibitors. 06:34 Now this is a drug, or a set of drugs that we use to actually reduce excess endogenous cortisol production. 06:41 Now, interestingly enough, ketoconazole, you might recognize this as antifungal medication. 06:46 We do use it in nonresectable adrenal adenoma, breast cancer and prostate cancer. 06:51 And it is also used in hirsutism for the treatment of excess cortisol levels. 06:56 Metyrapone is another medication that we use in glucocoritcoid pharmacology. 07:02 It inhibits synthesis of cortisol. 07:05 We actually use it more for diagnositic testing rather than actual therapy. 07:10 Mitotane is an interesting agent which also inhibits enzymes necessary in the synthesis of steroids. 07:15 However, it also results in the production of metabolites that destroy mitochondria in the adrenal gland, resulting in adrenocortical cell death. 07:25 This latter effect is why it is primarily used in the management of adrenocortical carcinoma. 07:32 However, it may also be considered in the management of Cushing disease if other therapies are not effective or contraindicated. 07:40 The zona reticularis function and adrenal androgens has been covered in another lecture, so I won't go into too much detail here.
About the Lecture
The lecture Mineralocorticoids and Glucocorticoids – Adrenal Pharmacology by Pravin Shukle, MD is from the course Endocrine Pharmacology. It contains the following chapters:
- Steroid Binding is Intracellular
- Mineralcorticoids
- Glucocorticoids
Included Quiz Questions
What is the signaling pathway for mineralocorticoids and glucocorticoids?
- These steroid hormones bind to intracellular receptors.
- These steroid hormones bind directly to an intranuclear receptor.
- These steroid hormones bind to a membrane receptor on the outside of the cell called tyrosine kinase.
- These steroid hormones bind to a transmembrane receptor on the cell called JAK-STAT.
- These steroid hormones bind to a GPCR.
What best describes the side effect profile of glucocorticoids?
- Increased gluconeogenesis and lipolysis, abnormal lipid deposition
- Decreased blood pressure, a slight drop in sodium, increased urine output
- Immune dysfunction, primarily affecting humoral immunity
- COX-2 inhibition and decreased platelet activity
- Increased glycogenolysis in the liver and lipolysis in adipose tissue
What is the difference between prednisone and dexamethasone?
- Dexamethasone is more potent and has a longer half-life than prednisone.
- Dexamethasone is topical only while prednisone can be taken orally or via IV.
- Prednisone comes with multiple warnings associated with it while dexamethasone has a very mild side-effect profile.
- Prednisone binds to a steroid receptor in the cell while dexamethasone binds to a receptor on the cell membrane.
- Prednisone is not recommended for children under 9 years, while dexamethasone is not recommended for adults over 65 years.
Ketoconazole does NOT have a therapeutic role for which condition?
- Adrenal failure
- Fungal infection
- Hirsutism
- Adrenal adenoma
- Breast cancer
Which condition does NOT improve with induction of mineralocorticoid activity?
- Cushing syndrome
- Addison disease
- POTS (paroxysmal orthostatic tachycardia syndrome)
- Geriatric orthostatic hypotensive syncope
- Cerebral salt wasting syndrome
Customer reviews
5,0 of 5 stars
| 5 Stars |
|
5 |
| 4 Stars |
|
0 |
| 3 Stars |
|
0 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |