Playlist
Show Playlist
Hide Playlist
Involved Proteins – Protein Movement and Cell Signaling
-
04 Basic ProteinMovement&CellSignaling.pdf
-
Biochemistry Free and Easy.pdf
-
Reference List Biochemistry.pdf
-
Download Lecture Overview
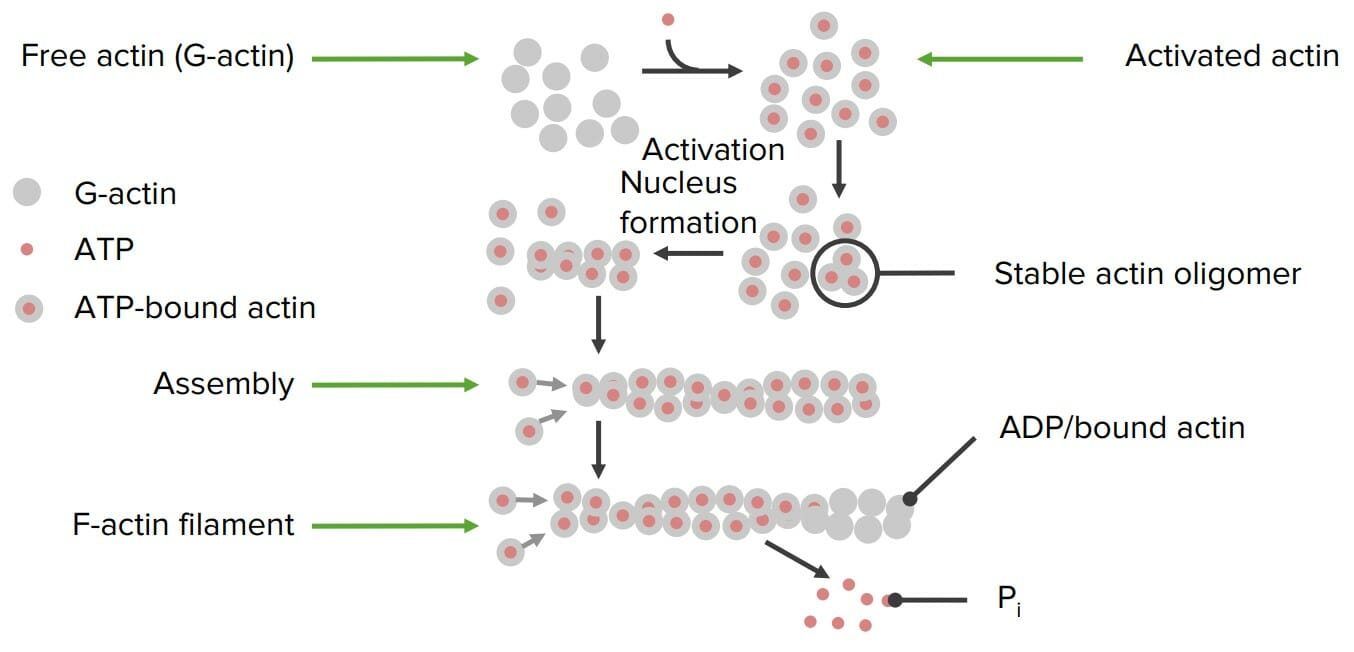
00:00 Another important function of proteins are those that are involved in movement. Now I talked previously about tubulin and actin, and how they were able to form either microfilaments or microtubules inside of cells. Here I want to talk about the proteins that actually move along these filaments. Now the mules that are involved in this process, at least the two most important ones as far as the microtubules are concerned, are called kinesin and dynein. What kinesin and dynein do is they use the energy of ATP, and with that energy of ATP, they grab cargo, which are things that they are moving literally like a mule, and they walk along the microtubules, step by step by step. It’s a remarkable process that this happens and so the movement of materials across the cell happens on this little microtubule highway that is existing inside of cells. 00:55 Another important protein involved in movement is known as myosin. In contrast to kinesin and dynein, which walk down microtubules, myosin walks down and does movement on top of actin filaments. Now myosin is involved in a variety of processes inside of a cell, and it's also the protein that's involved in muscular contraction. 01:14 Now, when we talk about movement, we really can't ignore what's happening with the assembly of the actin microfilaments. The polymerization act itself is a movement, as is the microtubule polymerization that happens with tubulin. One other movement that I want to mention in cells that is notable and very, very important is that of ATP synthase. ATP synthase is an enzyme found in the inner membrane of a mitochondrion, and it’s ATP synthase that is responsible for making probably 90% of all the ATP that's made inside of a cell. Interestingly this protein involves motion in order to make what it makes. Now this is shown schematically in a figure on the right, and in this figure on the right, we can see a portion of the ATP synthase. Specifically it's the portion of ATP synthase that makes the ATP. You can see on the screen a figure of what looks like a pie with three different colors on it. This is the functional part of the ATP synthase that actually makes the ATP. Now the three different colors correspond to three different proteins, but the three different proteins are actually identical in sequence to each other. They can each, however exist in a slightly different configuration and that configuration changes as a rotor turns within there. Now the rotor is shown in black, and the turning of the rotor causes the protein to change forms. Each protein here can have three different forms. One form for binding ADP and phosphate, one form for squeezing together ADP and phosphate to make ATP, and one form for letting go of the ATP that's been formed. During the turn of the rotor, all three of those processes will happen for these subunits as you can see here. Now watch on top first where the ADP and phosphate are going to come in and bind, you will see them be squeezed together and you will see them be released. Bound, squeezing and released. 03:24 Now, motion is fascinating inside of a cell. The motion that happens in the, what we describe as the 'walking along microtubules', is literally a walking process. Now this protein that I’m going to illustrate this for is a protein kinesin, and kinesin uses energy of ATP to actually do the walking process. And you can see the kinesin drawn schematically in a light grey above the microtubule below. You can see that the kinesin has associated with it to start on the left, adenosine diphosphate, as given by the D. The binding of ATP by the kinesin causes a swivel to actually happen, and you can see the swivel happen as a result of the binding of the T. The release of ADP causes another swivel to happen, as you can see here, and finally the release of a phosphate causes a third swivel. This swiveling back and forth allows the kinesin to literally take steps along this microtubule, a fascinating process. Dynein uses a very similar process, but dynein, in contrast to kinesin, works in the opposite direction. The tubulin has a polarity associated with it, kinesin walking one way and dynein walking the other way. 04:43 So we’ve seen in this module three important functions that proteins can perform. 04:49 First, structural function that makes for some very stable and durable proteins important for macromolecular organisms. And secondly we’ve seen two processes that actually mimic processes that happen in the macroscopic world that we see, that of communication and that of movement.
About the Lecture
The lecture Involved Proteins – Protein Movement and Cell Signaling by Kevin Ahern, PhD is from the course Biochemistry: Basics.
Included Quiz Questions
Which of the following is NOT true of intracellular motion?
- No energy is required.
- Kinesin and Dynein “walk” on cellular filaments.
- It can occur on tubulin and actin.
- It is a factor in muscular contraction.
- ATP synthase uses rotational movement to form ATP.
Which dimers must polymerize to create a microtubule?
- α- and β-tubulin dimers
- α- and β-kinesin dimers
- α- and β-dynein dimers
- Myosin dimers
- Actin dimers
Which of the following move the materials over the microtubule highways of the cell?
- Kinesin and Dynein
- Tubulin and Actin
- Actin and Myosin
- Myosin and Kinesin
- Dynein and Actin
Customer reviews
4,3 of 5 stars
| 5 Stars |
|
2 |
| 4 Stars |
|
0 |
| 3 Stars |
|
1 |
| 2 Stars |
|
0 |
| 1 Star |
|
0 |
I am appreciative that Dr. Ahern took the time to update his lectures. Shows he cares. Thank you!
The lecture breaks it down so simple that it’s easier to understand. There’s still more factors that I’m sure they’ll be discussed in other lectures but it’s perfect to help to create a strong foundation in your knowledge
ok. could use improvement please include that Kinesin and Dynein are ATPases that use energy. also that only Kinesin is displayed here. I was so busy looking for Dynein information, that Kinesin info went out the other ear. This could be better by improving the diagram.